- Cart 0
- English
Technical Principles and Experimental Applications of Exosome Extraction Kits
May 15, 2026
Clicks:71
What is an Exosome Extraction Kit?
The exosome extraction kit adopts functional modified magnetic bead capture technology to selectively isolate exosomes from complex biological samples through interactions between specific surface ligands and vesicle membrane proteins. The kit generally contains core components including magnetic bead suspension, incubation buffer, washing buffer and elution buffer, forming a complete extraction workflow.
Compared with traditional ultracentrifugation, the magnetic bead capture method shortens the extraction time from 4‑16 hours to approximately 2 hours, and avoids physical damage to vesicle structures caused by high centrifugal force. Instead of relying on vesicle density, this method identifies surface markers, theoretically yielding a more complete vesicle population.
Principle:

Why Choose Magnetic Bead‑Capture Method for Exosome Extraction?
Operational Convenience: No expensive and complex ultracentrifuge is required; all operations can be completed with a conventional centrifuge, magnetic rack and pipette. Ordinary researchers can master it with simple training, featuring high experimental throughput for simultaneous processing of multiple samples.
Wide Sample Compatibility: The kit can process various biological fluids including cell culture supernatant, urine, saliva, cerebrospinal fluid, etc. Differentiated operational protocols are provided for different sample types to meet the demands of diverse matrix characteristics.
Structural Integrity Preservation: Mild incubation and elution conditions maximally maintain the morphological structure and cargo integrity of exosomes, and isolated vesicles are suitable for direct electron microscopy observation and functional research.
In Which Experiments Is the Exosome Extraction Kit Indispensable?
Exosomal Proteomics Research: Extracted exosomes can be directly used for Western Blot verification of marker expression (e.g., CD63, CD9, TSG101) to confirm vesicle identity. After lysis, mass spectrometry can be applied to analyze intra‑vesicular protein composition and discover disease‑related biomarkers.
Exosomal Nucleic Acid Analysis: Purified exosomes can be used for RNA extraction (including mRNA, miRNA, lncRNA). RT‑qPCR or RNA‑seq can be adopted to analyze characteristics of nucleic acid cargo and explore their regulatory roles in intercellular communication.
Exosome Functional Research: Extracted exosomes are added into target cell culture systems to study their effects on functions such as proliferation, migration, apoptosis or differentiation of recipient cells. Owing to mild extraction processes, the biological activity of vesicles is retained, suitable for functional validation experiments.
Exosome Morphological Characterization: Isolated vesicle samples can be directly used for transmission electron microscopy (TEM) observation of morphology and particle size distribution, or nanoparticle tracking analysis (NTA) for concentration and particle size spectrum determination to comprehensively evaluate extraction quality.
Disease Biomarker Screening: Exosomes are extracted from patient urine or serum, and disease‑specific molecular biomarkers are identified via proteomic or transcriptomic analysis, providing technical support for liquid biopsy and early diagnosis.
What Are the Differences in Extraction Strategies for Different Sample Types?
Cell Culture Supernatant: Exosome concentration in supernatant is relatively high; only 5 μL magnetic beads per 1 mL supernatant enable efficient capture. Pre‑treatment steps are relatively simple, and direct extraction can be performed after removing cells and debris by centrifugation at 500‑2000 g.
Urine Samples: Urine contains numerous metabolites and salt interferences. A ratio of 20 μL magnetic beads per 1 mL urine is adopted to improve capture efficiency. Mid‑stream morning urine is recommended. Centrifuge at 2500 g for 10 min to remove cell debris; repeat centrifugation if necessary to obtain purer supernatant.
Saliva and Cerebrospinal Fluid: Such samples have high protein content and viscosity; magnetic bead dosage is increased to 50 μL per 1 mL sample to enhance capture capacity. Incubation time can be appropriately extended to 1 h to ensure sufficient binding.
Sample Storage and Pre‑treatment: Fresh samples should be processed immediately or stored at −80 °C. Thaw rapidly in a 37 °C water bath and avoid repeated freeze‑thaw cycles that cause vesicle rupture. All centrifugation steps are performed at 4 °C to prevent protein degradation.
How to Achieve High‑Quality Exosome Extraction?
Precise Control of Magnetic Bead Incubation: Incubate processed samples with magnetic beads at room temperature for 0.5‑1 h, and gently invert and mix 2‑3 times during incubation to promote contact between beads and vesicles. Insufficient incubation time leads to incomplete capture, while excessive time may increase non‑specific adsorption.
Key Details of Washing Steps: Invert up and down 20‑30 times during washing to disperse magnetic bead pellets with gentle movements to avoid shedding of captured exosomes. Fully perform magnetic separation after each wash to completely remove supernatant. Pre‑cool washing buffer to 4 °C to reduce non‑specific vesicle dissociation.
Optimization of Elution Conditions: Use special elution buffer to release exosomes via competitive binding or pH alteration. Vortex for 10 min in the standard protocol to ensure sufficient elution; one repeated elution can improve recovery rate by 20‑30%. Combine two eluates for adequate sample recovery.
Lyophilization Concentration Strategy: Eluates are usually large‑volume (200‑400 μL) with low protein concentration. Lyophilization concentrates samples by 10‑20 times for convenient downstream operations. Lyophilized powder can be stored long‑term at −80 °C with higher stability than liquid form.
What Precautions Should Be Taken During Experimental Operations?
Magnetic Bead Storage Conditions: Magnetic bead components are stored at 2‑8 °C and must not be frozen to prevent bead aggregation and failure. Other buffers can be stored at room temperature, but equilibrate to room temperature or pre‑cool as instructed before use.
Magnetic Rack Selection: High‑magnetic‑force racks should be used to ensure rapid and complete magnetic separation in 1.5‑2 mL centrifuge tubes. Insufficient magnetic force causes bead residue or reduced recovery rate.
Avoid Vigorous Vortexing: Do not vigorously vortex samples at any step, which causes magnetic bead fragmentation and exosome structural damage. Up‑and‑down inversion mixing is recommended.
Prevention of Cross‑Contamination: Replace pipette tips when handling different samples. Avoid repeated pipetting of magnetic bead suspension to generate aerosols. Clearly label extracted samples with source and date.
How to Evaluate Extraction Quality and Result Reliability?
Extraction Efficiency Verification: Determine total protein content of extracted exosomes via the BCA method and calculate extraction efficiency based on initial sample volume. High‑quality extraction yields 1‑5 μg protein per mL urine or 10‑50 μg protein per mL cell supernatant.
Purity Evaluation: Exosome markers should be positive via Western Blot, while organelle markers such as Calnexin (endoplasmic reticulum) should be negative. Purity can also be assessed by detecting contamination levels of non‑vesicular proteins such as albumin.
Morphological Integrity: Typical saucer‑shaped vesicles with a diameter of 50‑150 nm should be observed under transmission electron microscopy. NTA analysis should show a single particle size peak; excessively low concentration or broad particle size distribution indicates insufficient capture efficiency or aggregation.
Functional Verification: Co‑culture extracted exosomes with target cells. Expected biological effects (e.g., promoting cell migration or altering gene expression) confirm that extraction processes preserve vesicle biological activity.
Through magnetic bead capture technology, the exosome extraction kit provides researchers with an efficient and standardized exosome isolation solution. Mastering sample processing key points, optimizing extraction parameters and implementing strict quality control can obtain high‑purity and high‑integrity exosome samples, laying a solid technical foundation for downstream multi‑omics analysis and functional research.
Absin Exosome Extraction Kit Result Images:

WB detection results of exosomes extracted by EVtrap
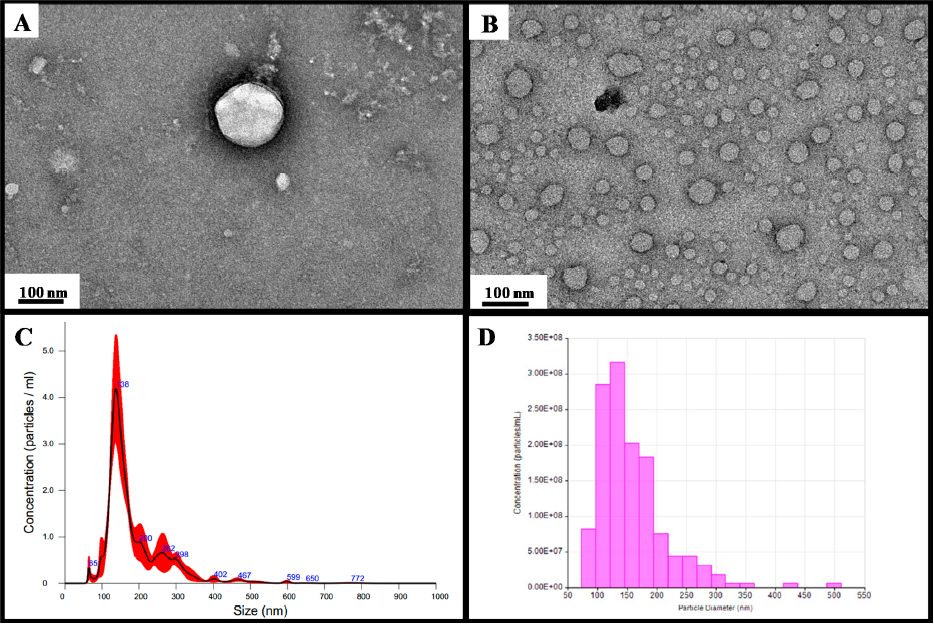
Transmission electron microscopy (TEM) images of (A) single EV and (B) multiple EVs captured from the plasma by EVtrap. TEM imaging of EVs was carried out on a Hitachi H‑8100 electron microscope with an accelerating applied potential of 200 kV. (C) Nanoparticle analysis using NTA after elution off EVtrap beads. (D) Nanoparticle analysis using TRPS after the elution of EVtrap beads.
Absin Exosome Extraction Kit Recommendation:
| Cat. No. | Product Name | Size |
|---|---|---|
| abs9775 | Exosome Extraction Kit | 1mL |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
