- Cart 0
- English
Analysis of the Principles and Application Scenarios of High‑Purity Plasmid Extraction Technology in Large‑Scale Quantities
May 11, 2026
Clicks:70
In molecular biology workflows, the quality and yield of plasmid DNA directly determine the success or failure of downstream experiments. For applications such as cell transfection, gene‑therapy research, high‑throughput screening and recombinant protein expression, high‑purity plasmid DNA at microgram‑to‑milligram levels is an indispensable experimental material. Midi‑to‑Maxi‑scale plasmid preparation is specially designed for bacterial cultures of 50 mL to over 100 mL. By optimizing lysis conditions, membrane‑based purification and endotoxin‑removal strategies, it enables efficient isolation of high‑concentration, low‑endotoxin plasmid DNA from bacterial cultures.
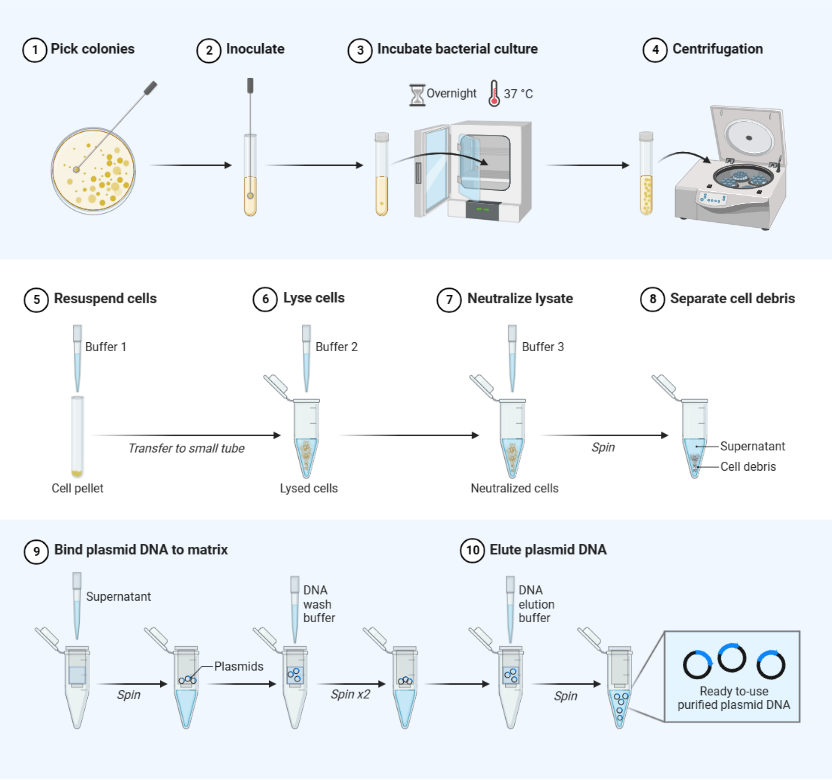
What Are the Fundamental Differences Between Midi‑/Maxiprep and Miniprep?
Conventional miniprep (1–5 mL bacterial culture) is mainly used for clone validation and PCR screening. For transfection‑grade or digestion‑grade plasmids, midi‑/maxiprep delivers technical upgrades in multiple dimensions:
Challenges of linear scale‑up: When culture volume increases from 1 mL to 50 mL or even 100 mL, uniform lysis, effective precipitate separation and spin‑column binding capacity become technical bottlenecks. Specialized kits increase lysis‑buffer volume ratios and adopt large‑volume silica‑membrane columns (binding capacity ≥150 μg) to ensure thorough processing of large‑volume cultures without column clogging.
Critical endotoxin control: Gram‑negative bacterial cell walls contain abundant lipopolysaccharides (LPS, endotoxins). Background contamination is relatively low in miniprep but rises sharply in large‑scale preparations. Endotoxins impair eukaryotic transfection efficiency, trigger inflammatory responses in immune cells, and inhibit in‑vitro transcription/translation systems. Midi‑/maxiprep kits reduce endotoxin levels to transfection‑acceptable standards (typically <0.1 EU/μg DNA) via specialized wash buffers and membrane‑purification technology.
Dual guarantees for concentration and purity: By optimizing elution conditions (e.g., pre‑warmed elution buffer, controlled elution volume), midi‑/maxiprep yields plasmid solutions with concentrations above 2 μg/μL, eliminating extra concentration steps, while maintaining A260/A280 ratios of 1.8–2.0 with minimal RNA and genomic DNA contamination.
Key Technical Points of Alkaline Lysis for Midi‑/Maxiprep
Modified alkaline lysis is widely used for large‑scale plasmid extraction, based on pH‑dependent differences in DNA denaturation and renaturation:
Functional analysis of buffer systems:
- Resuspension Buffer (Solution A/PI): Contains Tris‑HCl and EDTA to maintain osmotic pressure, chelate Mg²⁺ and inhibit DNase activity, ensuring homogeneous bacterial resuspension without clumps;
- Lysis Buffer (Solution B/PII): Contains SDS and NaOH (pH 12.0–12.5) to disrupt cell membranes and denature DNA. Lysis time must be strictly controlled (typically 2–5 min) in large‑scale systems; over‑lysis fragments genomic DNA and hinders separation from plasmids;
- Neutralization Buffer (Solution C/PIII): Contains high‑concentration potassium acetate or guanidine hydrochloride to rapidly neutralize pH and precipitate proteins, genomic DNA and SDS‑protein complexes into flocculent pellets.
Operational highlights: Perform high‑speed centrifugation (10,000×g, 15 min) at 4 °C after neutralization to form tight pellets and obtain clear supernatants. For cultures larger than 75 mL, process in batches or parallel tubes to avoid thick precipitates that reduce clarification efficiency.
How Do Spin‑Column Silica Membranes Achieve High‑Purity Purification?
Modern midi‑/maxiprep kits widely adopt high‑efficiency spin‑column silica‑membrane technology (silica or anion‑exchange membranes), replacing traditional phenol‑chloroform extraction and ethanol precipitation:
Binding mechanism: Under high‑salt conditions (usually with isopropanol or chaotropic salts), plasmid DNA specifically binds to silica matrices via hydrogen bonding and electrostatic interactions, while proteins, polysaccharides and small‑molecule metabolites flow through. Large‑diameter columns (compatible with 15 mL or 50 mL centrifuge tubes) increase membrane area and prevent supernatant overflow from large‑volume samples.
Washing strategy: Two sequential washes are normally performed — the first with ethanol‑containing buffer to remove salts and residual proteins, and the second with high‑concentration ethanol (70%–80%) for further purification and stable DNA‑membrane binding. Thorough centrifugation (10,000×g, 2 min) to eliminate residual ethanol is critical, as leftover ethanol inhibits downstream enzymatic reactions.
Elution optimization: Pre‑warmed (65 °C) low‑salt elution buffer (TE or Elution Buffer) significantly improves elution efficiency. Elution volumes of 40–100 μL can be selected according to target concentrations. For example, 40 μL elution buffer yields 2 μg/μL high‑concentration plasmid from an expected 80‑μg yield.
Which Experiments Require Such High Plasmid Purity?
Applications of high‑purity midi‑/maxiprep span modern molecular biology and biotechnology:
Cell transfection and gene therapy: Whether for transient transfection of 293T cells for viral packaging or primary‑cell electroporation for gene editing, high‑purity, low‑endotoxin plasmids are prerequisites for high transfection efficiency and cell viability. Endotoxin contamination alters membrane permeability and induces inflammatory‑factor release, severely disrupting experimental results.
Restriction digestion and clone validation: While routine digestion tolerates lower purity, high‑purity plasmids ensure complete cleavage and avoid star activity or non‑specific cutting for multi‑enzyme digestion, large‑fragment (>10 kb) plasmids or DNA‑methylation analysis.
DNA sequencing: Sanger sequencing or NGS library construction requires plasmid templates free of RNA contamination (interfering with fluorescence signals) and salt interference (affecting capillary electrophoresis or cluster generation).
In‑vitro transcription and translation: Cell‑free protein‑expression systems (e.g., rabbit reticulocyte lysate) are extremely sensitive to endotoxins and RNases; high‑purity plasmids guarantee high yields of correctly folded recombinant proteins.
When Should Extraction Strategies Be Adjusted?
Low‑copy‑number plasmids: Low‑copy plasmids (e.g., BAC, PAC or stringent plasmids) may produce poor yields even from 50‑mL cultures. Increase culture volume to ≥100 mL and proportionally scale all buffers (lysis, neutralization, isopropanol). Respect column binding limits (≤150 μg per column), as overloading causes DNA breakthrough.
Large plasmids (>10 kb): Large plasmids are prone to mechanical shearing during lysis. Avoid vigorous vortexing; mix by gentle inversion; extend pre‑resuspension time in Solution I for full bacterial dispersion; prolong post‑neutralization incubation for complete precipitate formation.
Gram‑positive bacteria: Standard kits may incompletely lyse thick‑cell‑wall Gram‑positive bacteria (e.g., Bacillus subtilis). Add lysozyme pre‑treatment or use specialized lysis formulations.
How to Ensure Batch‑to‑Batch Consistency?
Uniform bacterial culture status: Overnight cultures (12–16 h) should reach late‑logarithmic phase with OD600 = 2.0–4.0. Over‑dense cultures (OD > 5) cause incomplete lysis, while dilute cultures reduce yields.
Buffer condition monitoring: Precipitates may form in resuspension and neutralization buffers at low temperatures; heat at 37 °C before use for homogeneity. Lysis buffer (NaOH‑containing) absorbs CO₂ and undergoes pH drop, reducing lysis efficiency; store sealed and protected from light.
Cross‑contamination prevention: Large‑volume centrifuge tubes and columns increase cross‑contamination risks. Set negative controls (plasmid‑free bacterial cultures) per batch and use filter‑tip pipettes.
With advancing gene‑function research, synthetic biology and biopharmaceutical development, midi‑/maxiprep technology has evolved from a simple scaled‑up miniprep into a refined biomolecule‑purification process. Mastering its technical points improves experimental efficiency and guarantees reliable data for downstream applications, especially cell‑level functional studies.
Recommended Absin Midi‑/Maxi‑Scale High‑Purity Plasmid Extraction Kit
| Cat. No. | Product Name | Size |
|---|---|---|
| abs60297 | Midi‑/Maxi‑Scale High‑Purity Plasmid Extraction Kit | 20 T/40 T |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
