- Cart 0
- English
Reduction Mechanism of Dithiothreitol Solution and Biomolecule Protection Strategies
May 11, 2026
Clicks:69
In molecular biology and proteomics experiments, maintaining the reduced state of biological macromolecules is a fundamental step to ensure experimental success. As a highly efficient and low-toxicity reducing agent, Dithiothreitol (DTT) is widely used for cleaving protein disulfide bonds, protecting thiolated DNA modifications, and inhibiting oxidative stress. A thorough understanding of its chemical properties and application scenarios helps researchers optimize sample processing workflows and improve data reliability.
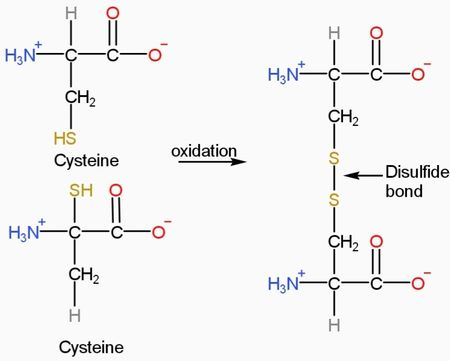
What is DTT?
DTT stands for Dithiothreitol, a small organic compound containing two thiol groups (-SH) (molecular formula C₄H₁₀O₂S₂), named for its structural similarity to threitol. Commercial DTT solution is typically supplied at a high concentration of 1 mol/L, with a solvent system containing stabilizers such as sodium acetate, and is filter-sterilized for direct use in cell culture or molecular biology experiments.
Chemically, the two thiol groups of DTT donate hydrogen atoms in redox reactions, reducing disulfide bonds (-S-S-) in proteins or nucleic acids to free thiols (-SH). Since DTT forms a stable six-membered cyclic disulfide (oxidized DTT) intramolecularly, this reaction is thermodynamically highly favorable, making it a potent reducing agent.
Why is DTT preferred over β-Mercaptoethanol?
In the selection of reducing agents, DTT is often regarded as a superior alternative to β-Mercaptoethanol (β-ME), mainly due to the following technical advantages:
Safety and Operational Friendliness: β-Mercaptoethanol has a strong pungent odor (like rotten onions) and high toxicity; long-term exposure may damage the respiratory system, and waste disposal requires special protection. In contrast, DTT has a significantly reduced pungent odor and lower toxicity, greatly improving the laboratory operating environment and reducing health risks to researchers.
Reducing Power and Stability: DTT exhibits strong reducing capacity at pH 6.5-7.0, with a standard redox potential of approximately -0.33 V, close to or slightly better than β-Mercaptoethanol. More importantly, DTT is more stable at physiological pH, resistant to air oxidation, and can maintain a reducing environment for an extended period.
Odor Control: In experiments requiring long-term incubation or open operations (e.g., protein dialysis, chromatographic purification), the low volatility of DTT reduces the accumulation of laboratory odors, complying with modern laboratory safety management standards.
In which experiments does DTT function?
DTT is applied across multiple key technical steps in biochemistry and molecular biology:
Protein Sample Preparation (Western Blot and SDS-PAGE): This is the most common application of DTT. In protein denaturation buffer (e.g., Laemmli buffer), DTT reduces intramolecular disulfide bonds of proteins, disrupting higher-order structures and completely denaturing proteins into linear polypeptide chains. This is critical for accurate determination of protein molecular weight (SDS-PAGE) and ensuring exposure of antigenic epitopes (Western Blot). Typically, during heating of samples at 95-100°C, the reducing action of DTT synergizes with the denaturing effect of SDS to achieve complete protein depolymerization.
DNA Synthesis and Oligonucleotide Deprotection: In solid-phase DNA synthesis, nucleotide monomers are often protected by thiol groups to prevent side reactions. As a deprotecting agent for thiolated DNA, DTT efficiently removes these protective groups to restore the native structure of DNA. Its rapid reaction kinetics and high conversion rate make it the reagent of choice for post-synthesis processing.
Protein Structure and Function Research: When studying the structure-function relationship of disulfide bond-containing proteins (e.g., antibodies, insulin, growth factors), DTT is used to reversibly break disulfide bonds and observe the effects of the reduced state on protein activity, folding, or aggregation. By controlling DTT concentration, partial reduction experiments can be performed to investigate the contribution of specific disulfide bonds to protein stability.
Cell Culture and Enzyme Activity Protection: In serum-free medium or specific enzyme reaction systems, adding low-concentration DTT (usually 0.1-1 mM) prevents oxidative inactivation of proteins and maintains the catalytic activity of thiol-containing enzymes (e.g., certain dehydrogenases, transferases). Its antioxidant effect also protects cells from oxidative damage induced by reactive oxygen species (ROS).
Protein Purification and Refolding: During the solubilization and refolding of inclusion body proteins, DTT is used for initial complete reduction, followed by gradual concentration reduction during dilution or dialysis refolding, allowing proteins to re-form correct disulfide bond pairings and restore native conformations.
How to properly store and use DTT?
The chemical properties of DTT dictate specific storage and handling protocols:
Avoid Repeated Freeze-Thaw Cycles: DTT solution is prone to oxidative degradation during repeated freeze-thaw cycles, resulting in reduced reducing power. Upon receiving the high-concentration (1 mol/L) stock solution, it is recommended to immediately aliquot into single-use small volumes (e.g., 50-100 μL/tube) and store frozen at -20°C. Take one tube for use upon thawing, and discard any remainder to avoid refreezing.
Sterile Handling: Although commercial DTT solutions are filter-sterilized, operations after opening should be performed in a biological safety cabinet or laminar flow hood to prevent microbial contamination. Especially in cell culture applications, contamination may cause protein degradation or cytotoxicity.
Concentration Optimization: Working concentrations should be adjusted according to experimental purposes:
- Protein Denaturation: Typically 10-100 mM (e.g., dilute 1M DTT stock 1:1 in 2×Loading Buffer)
- DNA Deprotection: 10-50 mM, incubate at 37°C for 30 minutes to several hours per synthesis protocol
- Antioxidant in Cell Culture: 0.1-1 mM, toxicity testing required for specific cell lines
Personal Protection: Although less toxic than β-Mercaptoethanol, lab coats, disposable gloves, and eye protection are still recommended to avoid direct skin contact and inhalation of aerosols. Handling large quantities of DTT should be performed in a fume hood.
Alternative and Combined Use: Under certain extreme conditions (e.g., requiring stronger reducing power), DTT can be used in combination with Tris(2-carboxyethyl)phosphine (TCEP), or replaced with odorless TCEP as needed. TCEP is more stable in aqueous solution and completely odorless but relatively costly.
Today, as proteomics and molecular biology techniques grow increasingly sophisticated, DTT remains an indispensable basic laboratory reagent due to its classic, reliable, low-toxic, high-efficiency, and easy-to-operate properties. Mastering its chemical principles and handling techniques provides solid assurance for the stable preservation and precise analysis of biological macromolecules.
Recommended Absin DTT Solution:
| Cat. No. | Product Name | Size |
|---|---|---|
| abs9226 | DTT Solution (1mol/L) | 5mL/5mL×2 |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
