- Cart 0
- English
Why is your intracellular lipid analysis always ambiguous and unclear?
May 09, 2026
Clicks:70
In laboratories focused on cellular metabolism research, there exists a subject that researchers both cherish and struggle with — Lipid Droplets (LDs). These intracellular reservoirs of neutral lipids play pivotal roles in energy metabolism, membrane synthesis, and signal transduction, yet they remain elusive like "ghosts": Oil Red O staining compromises cell viability; BODIPY labeling suffers from excessively high background fluorescence; mass spectrometry analysis disrupts cells and fails to distinguish lipid droplets from membrane lipids. Nile Red serves as the ideal "searchlight" to resolve this visual dilemma.
What Exactly is Nile Red?
Nile Red is a lipophilic fluorescent dye with the chemical name 9-Diethylamino-5H-benzo[a]phenoxazine-5-one and a molecular weight of 318.37 g/mol. It emits intense red fluorescence in hydrophobic environments while exhibiting negligible fluorescence in aqueous solutions. This environmental sensitivity makes it an ideal probe for detecting intracellular neutral lipids and lipid droplets.
Nile Red has several synonyms reflecting its chemical structure diversity: Nile Blue A oxazone, Phenoxazone 9. The pure product is typically supplied as a dark red to brown powder or green powder with a purity of 95%-98%.
As a fluorescent probe, the core advantages of Nile Red are:
- Dual-Color Emission: Displays distinct emission wavelengths in different polar environments — red fluorescence (~636 nm) in lipid droplets (hydrophobic) and green fluorescence (~580 nm) in membrane structures (polar lipids)
- High Signal-to-Noise Ratio: Nearly no background fluorescence in aqueous solutions, with fluorescence intensity increasing dozens of times upon partitioning into the lipid phase
- Live-Cell Compatibility: Low cytotoxicity, supporting live-cell real-time imaging and flow cytometric analysis
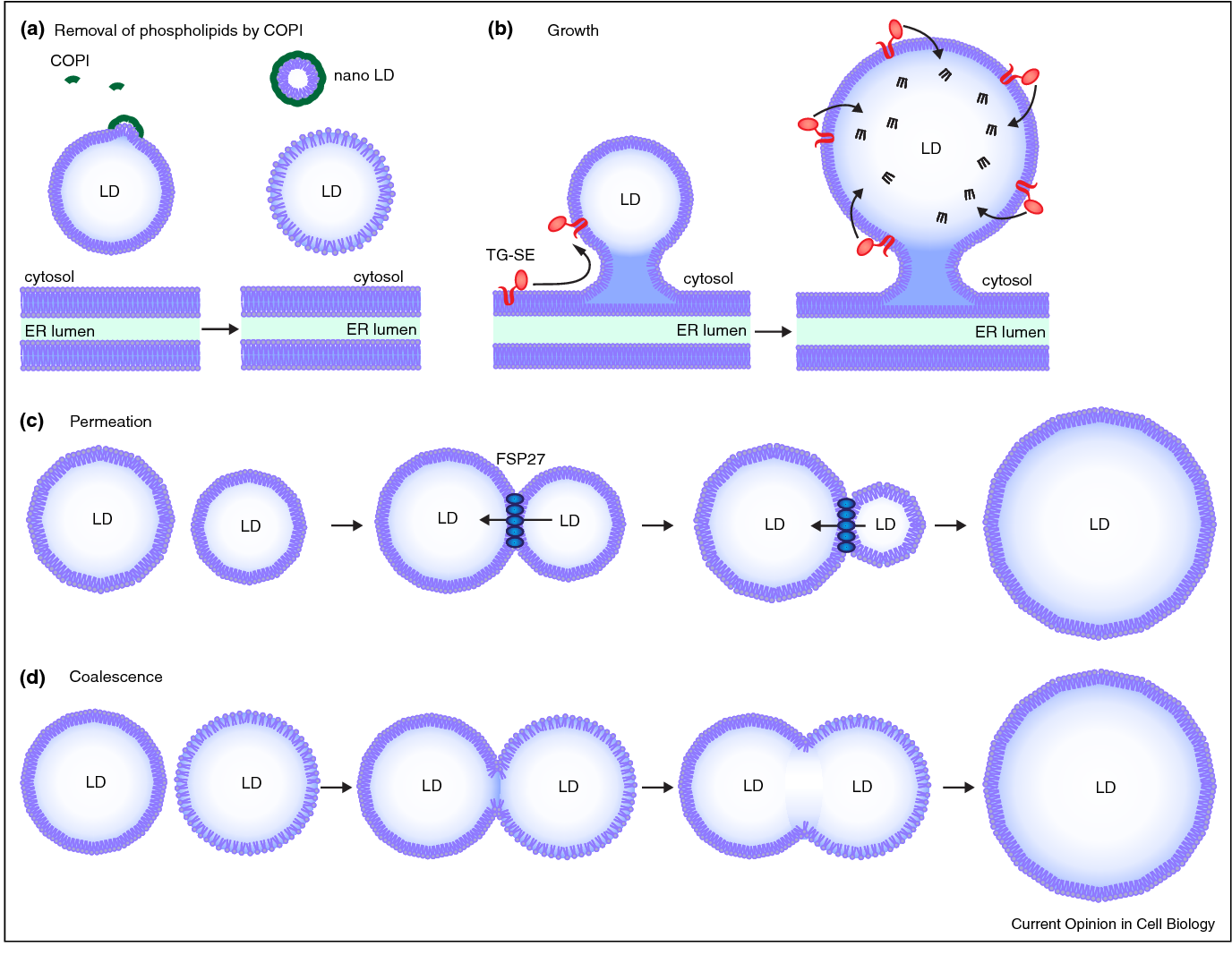
Figure 1: Biogenesis of lipid droplets and the staining principle of Nile Red. Lipid droplets bud from the endoplasmic reticulum and gradually fuse and grow. Nile Red inserts into the neutral lipid core of lipid droplets via its lipophilicity and emits intense red fluorescence in the hydrophobic environment. This staining method not only labels mature lipid droplets but also captures the dynamic process of lipid droplet formation.
Why Can Nile Red "Illuminate" the Secrets of Lipid Droplets?
Lipid droplets are essentially organelles composed of a neutral lipid (triglycerides and cholesterol esters) core, a phospholipid monolayer membrane, and surface proteins. This unique "oil droplet" structure provides a perfect platform for Nile Red.
Environmentally Sensitive Photophysical Properties
The fluorescence properties of Nile Red are highly dependent on environmental polarity:
- Hydrophobic Environment (lipid droplet core): Maximum excitation/emission wavelengths 552/636 nm, intense red fluorescence
- Polar Environment (aqueous solution): Fluorescence is almost quenched, extremely low background
- Medium-Polarity Environment (cell membrane): Emission wavelength blue-shifts to ~580 nm, yellow-green fluorescence
This "chameleon-like" property enables Nile Red to:
- Specifically Label Lipid Droplets: Emits strong red fluorescence only when entering the highly hydrophobic neutral lipid core
- Distinguish Lipid Types: Differentiate neutral lipids and polar lipids via emission spectra
- Real-Time Dynamic Tracking: Observe the formation, fusion, and degradation of lipid droplets in live-cell imaging
Sophisticated Design of Chemical Structure
The Nile Red molecule consists of:
- Large Conjugated Aromatic System: Ensures high fluorescence quantum yield
- Diethylamino Electron-Donating Group: Enhances charge-transfer characteristics and sensitivity to environmental polarity
- Phenoxazone Core: Provides photostability and chemical stability
This structure allows it to dissolve in organic solvents (DMSO, methanol) for easy preparation and remain stable in aqueous phases until encountering hydrophobic targets to "emit fluorescence".
Which Experimental Scenarios Rely on Nile Red?
Adipocyte Differentiation and Metabolism Research
This is the most classic application scenario of Nile Red. During the differentiation of 3T3-L1 preadipocytes into mature adipocytes:
- Differentiation Monitoring: The number and size of lipid droplets increase sharply, and Nile Red fluorescence intensity serves as a quantitative indicator of differentiation degree
- Lipolysis Assays: After isoproterenol stimulation, lipid droplets shrink and fluorescence weakens, reflecting lipolysis in real time
- Drug Screening: Evaluate the promoting or inhibitory effects of compounds on adipogenesis
Compared with other methods, Nile Red allows dynamic observation of live cells, whereas Oil Red O requires cell fixation and organic solvent treatment.
Non-Alcoholic Fatty Liver Disease (NAFLD) Models
Abnormal accumulation of lipid droplets in hepatocytes is the core pathological feature of NAFLD:
- Cell Models: Quantify lipid droplet accumulation in HepG2 or primary hepatocytes treated with oleic acid/palmitic acid using Nile Red
- Drug Intervention: Assess the efficacy of candidate drugs (e.g., PPAR agonists, FASN inhibitors) in reducing hepatic steatosis
- Mechanistic Studies: Observe the spatial relationship between lipid droplets and the endoplasmic reticulum/mitochondria via confocal microscopy
Cancer Lipid Metabolism Research
Tumor cells often exhibit a "lipid addiction" phenotype:
- Lipid Synthesis: Detect nascent lipid droplets in tumor cells with Nile Red to reflect fatty acid synthase (FASN) activity
- Lipophagy Research: Observe the co-localization of lipid droplets and autophagosomes to study the role of lipophagy in tumor survival
- Chemotherapy Resistance: Increased lipid droplet accumulation in certain drug-resistant cell lines can be analyzed by Nile Red for resistance mechanism studies
Atherosclerosis Models
Macrophage-derived foam cells are a hallmark of atherosclerosis:
- oxLDL Uptake: Label lipid droplets formed by oxidized low-density lipoprotein (oxLDL) with Nile Red
- Cholesterol Efflux: Evaluate the efficiency of cholesterol efflux mediated by transporters such as ABCA1 and ABCG1
- Plaque Analysis: Localize lipid cores within plaques via Nile Red staining of tissue sections
Neurodegenerative Disease Research
Abnormal lipid droplets in neurons and glial cells are associated with various neurological disorders:
- Alzheimer's Disease: Lipid droplet accumulation in APOE4 astrocytes
- Parkinson's Disease: Relationship between neuronal lipid metabolism disorder and α-synuclein aggregation
- Amyotrophic Lateral Sclerosis (ALS): Lipid droplet pathology in motor neurons
Single-Cell Lipidomics
Combined with flow cytometry sorting and mass spectrometry analysis:
- Cell Sorting: Separate cell populations with high (lipid-rich) and low Nile Red expression
- Metabolic Heterogeneity: Reveal individual differences in lipid metabolism within the same cell population
- Rare Cell Analysis: Identify rare cell subsets with unique lipid phenotypes
How to Use Nile Red Correctly?
Working Solution Preparation
Stock Solution Preparation:
- Dissolve Nile Red powder in anhydrous DMSO to prepare a 1 mM stock solution
- Aliquot into single-use volumes (e.g., 20-50 μL/tube) and store at -20°C protected from light
- Avoid repeated freeze-thaw cycles and prevent DMSO hygroscopicity from reducing solubility
Working Solution Dilution:
- Dilute with HHBS (Hanks' Balanced Salt Solution) or physiological buffer (pH 7.0-7.4)
- Typical working concentration: 1 μM (1 mM stock solution diluted 1:1000)
- Adjust concentration according to the experimental system — 0.5-2 μM for live-cell staining, slightly higher for fixed cells
Key Notes:
- Prepare fresh before use; Nile Red has limited stability in aqueous solutions
- Operate protected from light to prevent photobleaching
- If precipitation occurs, sonicate at 37°C to assist dissolution or prepare a new solution
Staining Protocol
Live-Cell Staining:
- Seed cells in culture dishes or slides and culture to appropriate density
- Treat cells with compounds (e.g., fatty acids, drugs) to induce lipid droplet changes
- Remove the culture medium and wash with PBS 1-2 times
- Add Nile Red working solution (500 μL/tube or sufficient to cover the cell surface)
- Incubate at room temperature or 37°C for 5-10 minutes protected from light
- Wash with PBS 2-3 times to remove unbound dye
- Add pre-warmed HHBS or medium for imaging
Fixed-Cell Staining (Optional):
- Fix with 4% paraformaldehyde for 15 minutes and wash with PBS
- Stain with Nile Red working solution (extend incubation time to 15-20 minutes for fixed cells)
- Note: Some fixatives may affect lipid droplet structure; live-cell staining is preferred
Detection and Analysis
Fluorescence Microscopy:
- Excitation filter: 535-555 nm (green excitation)
- Emission filter: >590 nm (red emission for specific lipid droplet detection)
- For membrane-bound Nile Red (green emission), use a 530-560 nm band-pass filter
Flow Cytometer:
- 488 nm laser excitation
- Detection channels: FL2 (585 nm) or FL3 (>670 nm)
- Collect data from both channels to distinguish lipid droplet (high FL3) and cell membrane (high FL2) signals
Quantitative Analysis:
- Fluorescence intensity reflects total lipid content of lipid droplets
- High-content imaging analyzes parameters such as lipid droplet number, size distribution, and circularity
- Time-lapse imaging tracks dynamic changes of lipid droplets
Common Problems and Optimization
Excessive Background:
- Reduce staining concentration or shorten incubation time
- Increase washing times to ensure removal of unbound dye
- Check cell density; over-confluent cells cause non-specific dye adsorption
Weak Signal:
- Confirm sufficient lipid droplets in cells (positive control: oleic acid-treated cells)
- Extend incubation time to 15-20 minutes
- Check stock solution for degradation (should be dark red; discoloration indicates inactivation)
Cytotoxicity:
- Nile Red has low intrinsic toxicity, but final DMSO concentration should be <0.1%
- Observe promptly after staining to avoid prolonged incubation
- For sensitive cells, reduce working concentration to 0.2-0.5 μM
From "Staining" to "Insight": The Technical Philosophy of Nile Red
The value of Nile Red lies not only in "making lipid droplets visible" but also in providing a non-invasive, dynamic, and quantitative lipid analysis tool. In fields such as metabolic diseases, cancer biology, and neuroscience, lipid droplets are no longer static "fat storage depots" but active metabolic hubs and signaling platforms.
Mastering the art of using Nile Red enables researchers to:
- Capture Moments: The formation and disappearance of lipid droplets are dynamic processes, and real-time imaging reveals spatiotemporal patterns
- Quantitative Comparison: Standardize fluorescence intensity to compare lipid accumulation across different treatments and cell lines
- Multidimensional Correlation: Combine with other probes (e.g., MitoTracker, ER-Tracker) to analyze the interaction network between lipid droplets and organelles
Conclusion
As a classic lipophilic fluorescent dye, Nile Red remains timeless in life science research. From adipocyte differentiation to tumor metabolism, from fatty liver models to neurodegenerative diseases, this "environmentally sensitive" probe continues to reveal the mysteries of the intracellular lipid world. Amid the booming development of metabolism research, the integration of Nile Red technology with high-resolution imaging, flow cytometry, and single-cell analysis is driving lipid biology from descriptive studies to mechanistic exploration.
Absin Nile Red Recommendation
|
Cat. No. |
Product Name |
Size |
|
Nile Red, Purity ≥98% |
50mg/500mg |
|
|
Nile Red, Purity 95%, BR Grade |
500mg/1g |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
