- Cart 0
- English
Why is excessive BSA residue always detected in your biological products before their launch?
May 09, 2026
Clicks:72
What Exactly Is the BSA Quantitation ELISA Kit?
The BSA Quantitation ELISA Kit is a dedicated detection system based on the Sandwich Enzyme-Linked Immunosorbent Assay (Sandwich ELISA) technique, designed for the precise quantification of trace residual Bovine Serum Albumin (BSA) in biological products. This kit can detect BSA as low as 0.5 ng/mL in complex matrices, meeting the stringent requirements of the biopharmaceutical industry for trace impurity control.
Technically, a complete BSA detection kit contains the following core components:
Capture System: 96-well microplate pre-coated with anti-BSA antibody, forming a solid-phase immunosorbent interface
Detection Antibody: Horseradish Peroxidase (HRP)-conjugated anti-BSA antibody, forming a "sandwich" complex with captured BSA
Standard System: Serial diluted BSA standards (typically 0.5–32 ng/mL) for quantitative standard curve construction
Auxiliary Reagents: Sample Diluent/Wash Buffer (20× concentrate), Chromogenic Substrate (TMB Solution A & B), Stop Solution (usually sulfuric acid), and Plate Sealing Film
This dual-antibody sandwich structure ensures two key performances: first, high specificity—simultaneous recognition by two antibodies targeting distinct epitopes virtually eliminates cross-reactivity; second, high sensitivity—enzymatic signal amplification enables sub-nanogram detection limits.
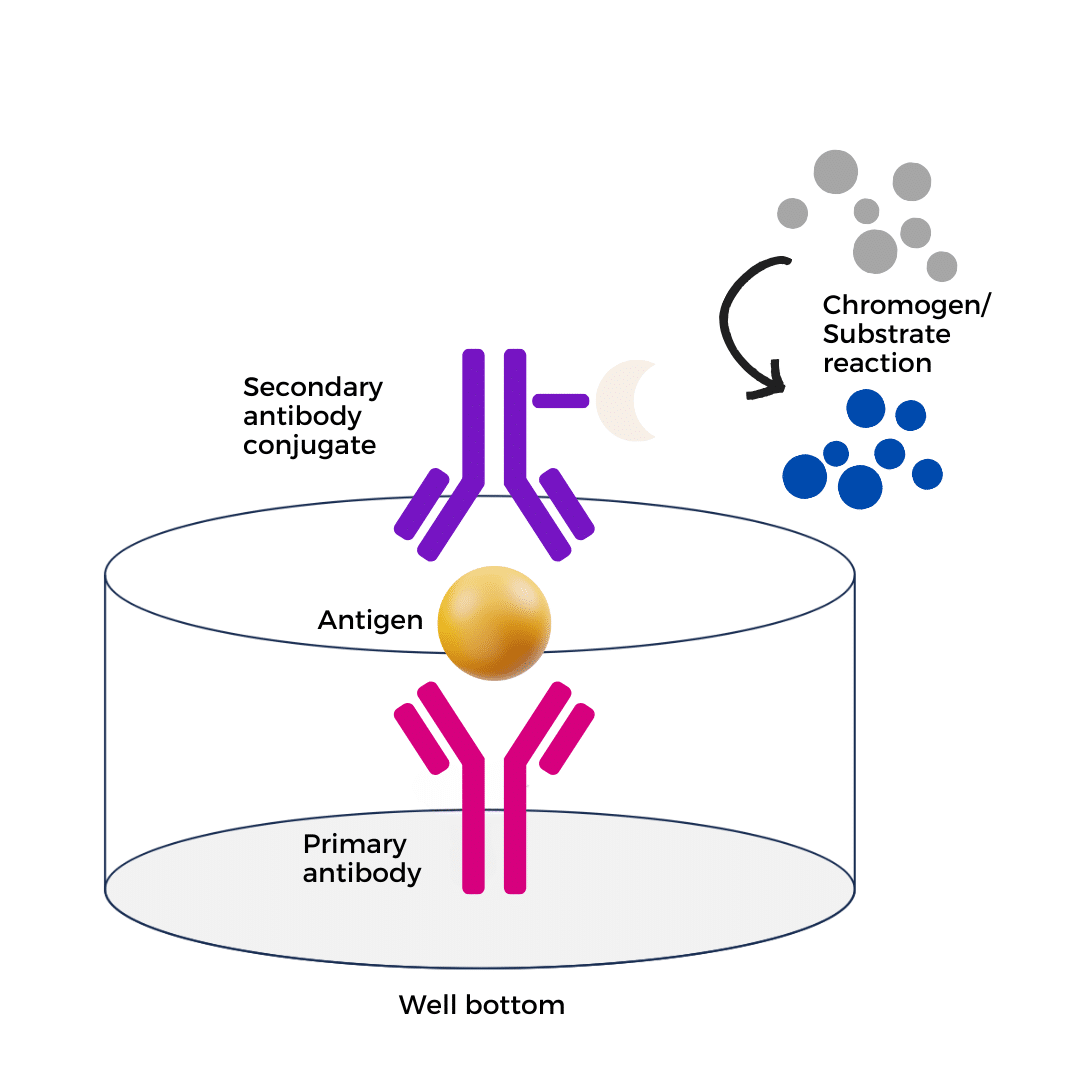
Figure 1: Principle of Sandwich ELISA. The capture antibody coated on the solid-phase carrier first binds BSA in the sample, followed by the addition of HRP-labeled detection antibody to form an "antigen-antibody-antibody" sandwich complex. After adding TMB substrate, HRP catalyzes the chromogenic reaction to produce a blue product, which turns yellow upon treatment with Stop Solution. Absorbance is measured at 450 nm, and signal intensity is positively correlated with BSA concentration.
Why Is BSA Residue Control So Critical?
BSA’s Dual Role: From Helper to Hazard
In cell culture and bioprocessing, BSA plays positive roles:
- Nutrient Carrier: Transports fatty acids, hormones, and small-molecule nutrients to support cell growth
- Surfactant: Reduces mechanical damage to cells from bubbles in culture media
- Stabilizer: Protects protein drugs from denaturation, aggregation, or surface adsorption
However, when these biological products enter the human body, BSA transforms from a "helper" into a "hazard":
- Immunogenicity Risk: As a heterologous protein, BSA may trigger allergic reactions or Anti-Drug Antibody (ADA) production
- Drug Efficacy Interference: High-concentration BSA may compete for targets with therapeutic proteins or affect pharmacokinetics
- Regulatory Mandate: Volume III of the Chinese Pharmacopoeia 2020 Edition clearly stipulates that BSA residues in human vaccines shall not exceed 50 ng/mL (bulk test) and 50 ng/dose (finished product test)
Complexity of Residue Sources
BSA residues can originate from multiple stages:
- Raw Materials: Fetal Bovine Serum (FBS), a key component of cell culture media, contains up to 30–50 mg/mL BSA
- Manufacturing Process: Incomplete media exchange, overlapping BSA elution peaks with therapeutic proteins during chromatographic purification
- Formulation: Some biological products use BSA as an excipient or stabilizer, requiring precise control of addition levels
Which Experimental Scenarios Require the BSA Quantitation ELISA Kit?
Biological Product Quality Control & Release Testing
This is the core application of BSA detection kits. Whether recombinant protein drugs, monoclonal antibodies, vaccines, or cell therapy products, BSA residue test reports must be provided before marketing registration.
Taking monoclonal antibody production as an example:
- Upstream Cell Culture: Monitor BSA clearance after serum-free media transition
- Downstream Purification: Test BSA residues after Protein A affinity chromatography and ion-exchange chromatography steps to optimize elution conditions
- Bulk Release: BSA content must be tested for each bulk batch to comply with pharmacopoeia standards (typically <100 ng/mL)
- Finished Product Testing: BSA residues in final formulations directly impact product safety evaluation
Serum-Free Media Process Development
As the biopharmaceutical industry transitions to serum-free, chemically defined media, BSA detection has become a key indicator for process validation. During media optimization:
- Serum Replacement Screening: Assess BSA contamination levels in alternative supplements (e.g., N-2, B-27, ITS)
- Media Transition Validation: Confirm continuous reduction of BSA residues in cell supernatants and products after switching from serum-containing to serum-free media
- Supplier Audit: Test BSA background in media from different batches and sources to establish supplier quality standards
Raw Material & Excipient Quality Control
BSA testing is a mandatory item in the biopharmaceutical raw material quality control system:
- Fetal Bovine Serum (FBS): As the highest-risk raw material, BSA content must be tested before each FBS batch is accepted, and batch-to-batch consistency evaluated
- Recombinant Protein Expression Hosts: Confirm no BSA introduction in fermentation media for E. coli, yeast, or mammalian cell expression systems
- Chromatography & Membrane Materials: Assess whether production consumables release BSA or adsorb BSA from products
Process Change & Comparability Studies
When manufacturing processes are modified (e.g., scale-up, equipment replacement, purification step adjustment), comparability studies are required. BSA residue detection is a critical indicator for evaluating process change impacts on product quality. Comparing BSA levels before and after changes confirms no increased impurity risk with the new process.
Biosimilar & Reference Product Comparison
When developing biosimilars, consistency in impurity profiles is a key regulatory focus besides demonstrating similar efficacy. As a critical process-related impurity, comparative analysis of BSA residue levels provides important evidence for proving quality similarity between biosimilars and reference products.
How to Use the BSA Quantitation ELISA Kit Correctly?
Critical Pre-Experiment Preparations
- Strictly prohibit BSA powder weighing or storage in the same area
- Thoroughly clean lab benches; dedicated testing stations are recommended
- Dedicate pipettes, centrifuges, and equipment to avoid cross-contamination
Reagent Preparation:
- All reagents and samples must equilibrate to room temperature (20–25°C) before use
- Dilute 20× concentrated Buffer to 1× working solution with deionized water; warm at 37°C to dissolve crystals if present
- Mix equal volumes of Chromogenic Substrate A and B, prepare fresh (10 minutes before use); discard if blue after mixing
- Dilute HRP-conjugated antibody (100× concentrate) 500-fold with 1× Buffer to working concentration, prepare fresh, and keep cold during handling
Precise Standard Curve Construction
Standard curve quality directly determines quantitative accuracy. An 8-point curve (0.5, 1, 2, 4, 8, 16, 32 ng/mL plus blank control) is recommended:
- 1 High-Concentration Stock Preparation: Prepare 2-fold serial dilutions from 5 μg/mL standard
- 2 Duplicate Testing: At least 2 replicates per concentration to reduce random error
- 3 4-Parameter Logistic Fit: Use professional software (e.g., GraphPad Prism) for curve fitting, ensuring R² > 0.99
Sample Handling & Dilution Strategy
Complex sample matrices are the main challenge in BSA detection:
- High-Concentration Samples: If expected BSA exceeds the curve upper limit (32 ng/mL), dilute appropriately with 1× Buffer to bring OD values within the linear range
- Viscous Samples: For cell culture supernatants or high-concentration protein formulations, centrifuge at 3000 rpm for 5 minutes to remove insoluble matter before testing supernatants
- Colored Samples: For naturally colored samples (e.g., phenol red-containing media), set sample blanks and subtract background during calculation
Key Operational Details
Pipetting Techniques:
- Add standards in sequential order (low to high or high to low) to avoid cross-contamination
- Complete sample addition within 10 minutes to prevent reaction variability from time differences
- Avoid dispensing onto well walls or generating bubbles
Incubation Conditions:
- Incubate at 37°C with shaking at 400–600 rpm for 1 hour to ensure full antigen-antibody binding
- Seal tightly with plate film to prevent evaporation-induced edge effects
Washing Guidelines:
- Add 250 μL 1× Buffer per well, wash 3 times
- Pat dry residual liquid after each wash, but avoid over-drying that causes protein denaturation
- If using a washer, let stand 1 minute or gently shake 5 seconds after washing to ensure thoroughness
Color Development & Termination:
- Incubate at 37°C in the dark for 20 minutes after adding substrate
- Complete reading within 20 minutes after adding stop solution to avoid color fading
- Dual-wavelength reading: 450 nm primary, 630 nm reference; OD = OD₄₅₀ – OD₆₃₀ to effectively deduct plate background
From Detection to Control: Quality Management Thinking for BSA Testing
Completeness of Method Validation
In accordance with ICH Q2(R1) and Chinese Pharmacopoeia Guideline 9101, BSA detection methods require systematic validation:
- Specificity: Confirm cross-reactivity <1% with potential interferents (horse serum, rabbit serum, collagenase, etc.)
- Accuracy: Spike recovery between 70%–130%
- Precision: Repeatability (intra-assay) <10%, intermediate precision <15%
- Linearity: Correlation coefficient r ≥ 0.99 within 0.5–32 ng/mL
- LOQ & LOD: LOQ typically set at 0.5 ng/mL, LOD <0.5 ng/mL
Laboratory Quality Control System
- Positive Control: Include known-concentration QC on each plate to monitor batch consistency
- Blank Control: Monitor reagent background and environmental contamination
- Matrix Effect Assessment: Spike known BSA into sample matrix to evaluate recovery
Conclusion
The BSA Quantitation ELISA Kit is an indispensable foundational tool in biopharmaceutical quality control systems. From raw material release to finished product testing, process development to change validation, sensitive, specific, and reproducible BSA detection directly impacts product safety, efficacy, and regulatory compliance. Amid increasingly stringent bioproduct regulation, mastering this technology and establishing a robust BSA residue control strategy is essential expertise for every biopharmaceutical professional.
Recommended Absin BSA Residue Detection Kit
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
