- Cart 0
- English
Why are your phosphorylated protein experiments always unstable? You may have overlooked this crucial step!
May 08, 2026
Clicks:72
Among various techniques in protein research, phosphorylation modification analysis is undoubtedly one of the most challenging and informative directions. However, many researchers often encounter a confusing problem during experiments: phosphorylation signals of the target protein are clearly detected in cells or tissues, but the signals inexplicably weaken or even disappear after sample preparation. Behind this phenomenon lies a key factor that is often overlooked —— the activity of endogenous phosphatases.
What is a Broad-Spectrum Phosphatase Inhibitor Cocktail?
A Broad-Spectrum Phosphatase Inhibitor Cocktail is an optimized biochemical reagent combination specifically designed to maintain protein phosphorylation status during cell or tissue lysis. This cocktail typically contains four or more specific inhibitors targeting different types of phosphatases, which can block multiple dephosphorylation pathways in a synergistic manner.
Technically, this cocktail mainly targets the following classes of phosphatases:
- ● Serine/Threonine Phosphatases: Including members of protein phosphatase families such as PP1, PP2A, and PP2B
- ● Protein Tyrosine Phosphatases (PTPs): Responsible for removing phosphate groups from tyrosine residues
- ● Alkaline Phosphatases: Phosphomonoester hydrolases active under alkaline pH conditions
- ● Acid Phosphatases: Phosphatases functioning in acidic environments
These reagents are usually supplied as 100× concentrated aqueous solutions, and can be diluted to working concentration at a ratio of 1:99 (e.g., 10 μL inhibitor added to 990 μL lysis buffer) before use.
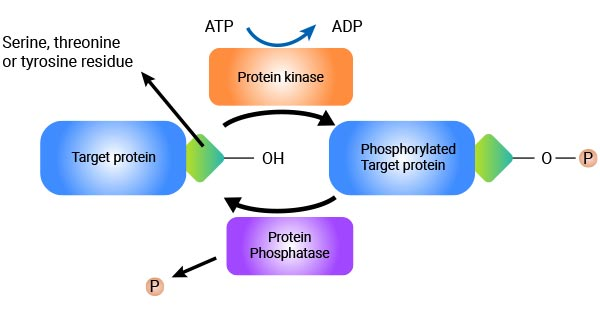
Figure 1: Schematic diagram of the protein phosphorylation-dephosphorylation cycle. Protein kinases catalyze the transfer of phosphate groups from ATP to serine, threonine, or tyrosine residues of target proteins to form phosphorylated proteins; while protein phosphatases are responsible for removing phosphate groups to dephosphorylate proteins. During sample preparation, the release of endogenous phosphatases leads to the loss of phosphorylation signals.
Why is Phosphatase Inhibition So Critical?
To understand the importance of phosphatase inhibitors, we first need to recognize the dynamic nature of phosphorylation modifications in cells. Protein phosphorylation is a reversible post-translational modification co-regulated by protein kinases and phosphatases, forming a precise "molecular switch" system. Under normal physiological conditions, this dynamic balance is crucial for cell signal transduction, metabolic regulation, and gene expression.
However, when cells or tissues are lysed, this precise regulatory mechanism is completely disrupted:
- Disruption of Compartmentalization: In intact cells, kinases and phosphatases are often localized in different subcellular regions. After lysis, these enzymes mix freely in solution, leading to uncontrolled acceleration of dephosphorylation reactions.
- Dilution or Inactivation of Inhibitors: Endogenous small-molecule inhibitors (natural counterparts of Microcystin-LR) in cell lysates are diluted, while pH changes and altered redox environments may enhance the activity of some phosphatases instead.
- Temperature Effects: Routine sample processing is usually performed on ice or at 4°C, but some phosphatases still maintain significant activity at low temperatures and continuously catalyze dephosphorylation reactions.
Studies have shown that in lysates without phosphatase inhibitors, some highly active phosphatases can significantly reduce the abundance of phosphorylated proteins within minutes, which may lead to false-negative results for target proteins with inherently low phosphorylation levels.
Which Experiments Cannot Do Without Phosphatase Inhibitors?
Based on their functional properties, the application scenarios of broad-spectrum phosphatase inhibitor cocktails are mainly focused on experiments requiring accurate retention or detection of protein phosphorylation status.
Western Blot
This is the core technique for validation of phosphorylation-specific antibodies. Whether detecting dual phosphorylation of ERK1/2 at Thr202/Tyr204 or phosphorylation of Akt at Ser473, phosphatase inhibitor cocktail must be added to the lysis buffer. Otherwise, even if cells are fully stimulated with growth factors before lysis, the final band intensity may not reflect the true phosphorylation level.
Co-Immunoprecipitation (Co-IP) & Kinase Activity Assays
Phosphatase inhibitors are particularly prominent when studying protein-protein interactions or kinase activity in immune complexes. For example, after precipitating a signaling molecule with a specific antibody, the presence of inhibitors ensures that the phosphorylation status of downstream effector molecules bound to it is not lost during precipitation.
In addition, for in vitro kinase activity assays using cell lysates as the kinase source, adding phosphatase inhibitors prevents dephosphorylation of phosphorylated substrates or the kinase itself, ensuring the accuracy of enzyme activity measurements.
Phosphoproteomics
In mass spectrometry-based phosphoproteomics research, the quality of sample preparation directly affects identification depth and quantitative accuracy. Since complex protease digestion and peptide enrichment steps are required before mass spectrometry analysis with a long time span, the use of phosphatase inhibitors has become an essential part of standard operating procedures.
Other Application Scenarios
- Enzyme-Linked Immunosorbent Assay (ELISA): Quantitative detection of phosphorylated protein content in cell lysates
- Immunohistochemistry/Immunofluorescence: When preparing tissue lysates in advance for protein concentration standardization
- Protein Microarray Technology: Analysis of expression profile changes of multiple phosphorylated proteins
How to Use It Correctly for Optimal Results?
Timing and Method of Use
The golden principle for using phosphatase inhibitor cocktail is "the earlier, the better". It is recommended to pre-cool and add the inhibitor to the lysis buffer at the recommended ratio (usually 1:100) before cell lysis, ensuring thorough mixing of the inhibitor and lysis buffer before contacting the sample. For tissue samples, due to the usually higher content of endogenous phosphatases, it may be necessary to appropriately increase the inhibitor concentration or use it together with protease inhibitor cocktail.
Operational Precautions
Conclusion
In protein phosphorylation research, quality control during sample preparation often determines the success or failure of experiments. As a basic but indispensable reagent, the value of broad-spectrum phosphatase inhibitor cocktail lies in providing researchers with a "time window" to truly retain phosphorylation modification status during post-lysis operations. For any experimental protocol involving phosphorylated protein detection, incorporating phosphatase inhibitors into standard operating procedures is a necessary guarantee to obtain reproducible and reliable data.
Absin Broad-Spectrum Phosphatase Inhibitor Cocktail Recommendation
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
