- Cart 0
- English
How does papain play multiple roles in biological research?
May 06, 2026
Clicks:74
In biochemistry and cell biology research, the selection of enzymes directly determines experimental success. As a classic cysteine protease, papain plays an irreplaceable role in cell isolation, antibody engineering, and protein structure studies due to its unique hydrolysis properties and broad substrate adaptability. This article explores the molecular characteristics, activation mechanism, and application value of papain in various experiments.
What is Papain?
Papain is a cysteine protease composed of a single polypeptide chain containing three disulfide bonds and one thiol group essential for enzymatic activity. It has a molecular weight of approximately 23 kDa and an optimal working pH range of 6.0–7.0. Derived from unripe fruits of the *Carica papaya* plant, this natural biological enzyme has an activity of ≥2000 units/mg.
Papain exhibits endopeptidase activity, cleaving peptide bonds of basic amino acids, leucine, or glycine, and also possesses esterase and amidase activities. Uniquely, it preferentially recognizes and cleaves amino acids with large hydrophobic side chains (P2 position), giving it broader proteolytic activity than trypsin and enabling cleavage of most protein substrates.
Why Must Papain Be Activated Before Use?
The active site of papain contains a critical cysteine residue. In its native state, this thiol group may be oxidized or bound to other molecules, rendering the enzyme inactive or weakly active. Therefore, activation is required before use to restore full enzymatic activity.
Activation Mechanism:
Papain is typically diluted in buffer containing ~5 mM L-cysteine. Cysteine acts as a reducing agent to break disulfide bonds at the active site, exposing free thiol groups. Meanwhile, EDTA (a metal chelator) removes heavy metal ions that inhibit enzyme activity. Dimercaprol may also be used as a stabilizer.
Common Activation Buffer Recipe:
Buffer containing 1.1 mM EDTA, 0.067 mM β-mercaptoethanol, and 5.5 mM cysteine hydrochloride. Complete activation occurs after 30 minutes of incubation. Sterilization by filtration through a 0.22 μm membrane is recommended before use.
Stability Characteristics:
While papain solutions exhibit excellent thermal stability, their stability is pH-dependent, with significant activity loss under acidic conditions (pH < 2.8). The enzyme retains 100% activity after recrystallization in denaturants such as 70% methanol or 8 M urea but loses substantial activity in 10% trichloroacetic acid or 6 M guanidine hydrochloride.
Which Experimental Scenarios Can Be Applied?
Mild Cell Isolation:
This is the most classic application of papain. Compared with trypsin, papain provides gentler dissociation for certain tissues, making it ideal for isolating cells sensitive to mechanical and chemical damage, such as cortical neurons, retinal cells, and smooth muscle cells. Its mild hydrolysis effectively dissociates cell-cell junctions while preserving cell viability.
Antibody Fragment Preparation:
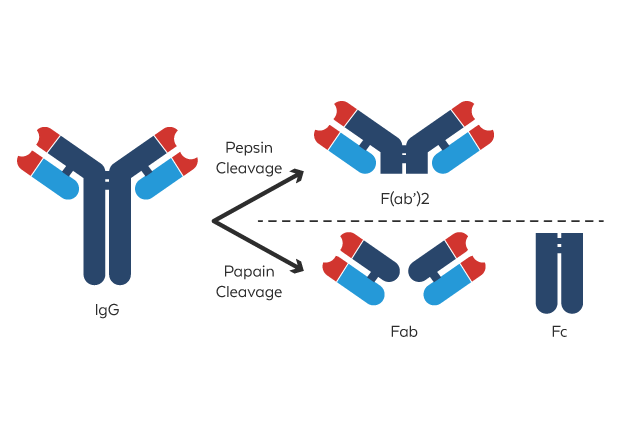
A key immunological application: papain cleaves IgG and IgM antibodies at the hinge region, generating two Fab fragments (antigen-binding) and one Fc fragment (crystallizable). Fab fragments retain full antigen-binding capacity but have half the molecular weight (~50 kDa), offering advantages including superior tissue penetration, reduced immunogenicity, and absence of Fc-mediated effector functions. This technique is widely used in therapeutic antibody development, antibody-drug conjugate (ADC) research, and diagnostic reagent production.
Protein Structure Analysis:
Papain is used for structural studies and peptide mapping of enzymes and other proteins. Controlled proteolysis yields specific peptide fragments for mass spectrometry and sequencing.
Red Blood Cell Serology:
Papain modifies red blood cell surfaces to enhance or abolish antigen reactivity, serving as an auxiliary method for blood typing. It is also used for antibody screening, identification, and platelet serology studies.
Enzymatic Synthesis:
Papain catalyzes the enzymatic synthesis of amino acids, peptides, and other molecules. Its reversible hydrolysis enables peptide bond formation under specific conditions, valuable in organic synthesis.
Integral Membrane Protein Solubilization:
Papain facilitates solubilization of integral membrane proteins, aiding extraction and purification.
Proteoglycan Degradation:
Papain digests purified proteoglycans to produce glycopeptides for glycobiology research.
Enzymatic Wound Debridement:
Papain removes necrotic tissue to promote wound healing.
How to Use Papain Correctly?
Stock Solution Preparation:
Dissolve an appropriate amount of papain in water to prepare a 10 mg/mL stock solution. Papain is soluble in sodium chloride solution and remains stable for months at 2–8°C with minimal activity loss.
Enzyme Activation Steps:
Activate before use to achieve full activity. Dilute the papain stock solution in buffer containing ~5 mM L-cysteine (standard buffer: 1.1 mM EDTA, 0.067 mM β-mercaptoethanol, 5.5 mM cysteine hydrochloride). Incubate for 30 minutes for complete activation. Sterilize by filtration through a 0.22 μm membrane before use.
Reaction Condition Control:
Optimal pH: 6.0–7.0; temperature: typically 37°C. Reaction time is optimized based on substrate and purpose, ranging from minutes to hours.
Key Precautions for Use
- pH-dependent stability. Papain stability is pH-dependent, with significant activity loss at pH < 2.8. Avoid strongly acidic conditions during preparation and use.
- Avoid oxidative inactivation. Activity depends on free thiols; avoid oxidants. Stabilize with EDTA, cysteine, or dimercaprol during storage.
- Denaturant tolerance. Retains full activity in 70% methanol or 8 M urea but loses activity in 10% trichloroacetic acid or 6 M guanidine hydrochloride. Account for sample denaturants in experimental design.
- Temperature control. Despite good thermal stability, prolonged exposure to >50°C causes irreversible inactivation. Maintain reaction temperature around 37°C.
- Sterile operation. Filter-sterilize through a 0.22 μm membrane before use to prevent microbial contamination and enzyme degradation.
Schematic Diagram of Antibody Enzymatic Fragmentation
Conclusion
As a classic cysteine protease, papain holds a vital position in life science research due to its broad substrate specificity, mild digestion conditions, and versatile applications. From primary cell isolation to antibody engineering, protein structure analysis to preclinical research, papain demonstrates unique value as a "biological scissors." Mastering activation methods, condition optimization, and stability control maximizes experimental efficiency. With rapid advances in antibody drugs and cell therapy, papain’s prospects in biopharmaceuticals are increasingly promising.
Recommended Absin Papain:
| Cat. No. | Product Name | Size |
|---|---|---|
| abs47014927 | Papain | 25g/100g |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |