- Cart 0
- English
How do pH-responsive fluorescent probes achieve precise visualization of lysosomes?
April 30, 2026
Clicks:72
As the cellular "digestive workshop", lysosomes undertake the core functions of degrading biological macromolecules, recycling nutrients, and maintaining cellular homeostasis. They maintain an acidic microenvironment with a pH of 4.5–5.5, which is not only essential for normal lysosomal function but also provides an ideal chemical target for specific labeling. Fluorescent probes based on acidic pH-activation mechanisms achieve highly selective, real-time dynamic imaging of lysosomes in living cells through protonation-induced fluorescence enhancement, providing a powerful visualization tool for cell biology and disease mechanism research.
Why is the acidic environment of lysosomes the key to targeted labeling?
Lysosomes are among the most acidic organelles in cells, with a luminal pH maintained at 4.5–5.5, while the cytoplasmic pH is approximately 7.2. This significant pH gradient arises from the continuous action of V-ATPase proton pumps on the lysosomal membrane, which pump H⁺ from the cytoplasm into the lysosomal lumen. This acidic environment is critical for the optimal activity of lysosomal hydrolases (e.g., proteases, nucleases, lipases).
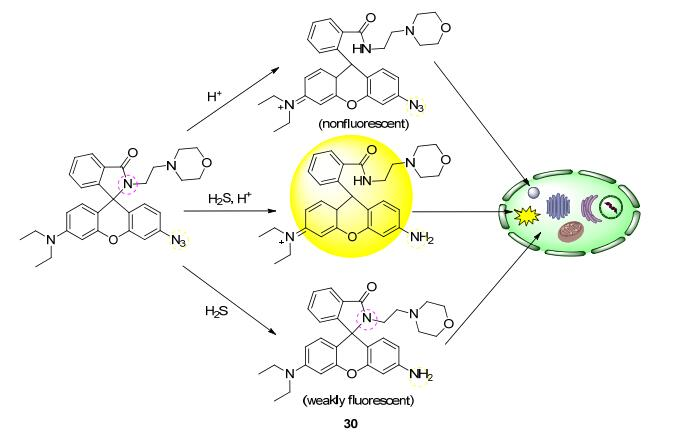
pH-responsive fluorescent probes achieve specific targeting by exploiting this biological feature. These probe molecules typically contain weakly basic side chains (e.g., morpholine groups) and are electrically neutral or weakly fluorescent at physiological pH. Upon entering the acidic lysosomal environment, the side chains become protonated, which not only enhances retention in lysosomes but, more importantly, relieves intramolecular fluorescence quenching, resulting in a significant increase in fluorescence signal. This "off-on" fluorescence activation mechanism ensures the probe emits light only in the target organelle, greatly reducing background interference.
How does the protonation mechanism drive fluorescence signal amplification?
The working principle of lysosomal fluorescent probes involves sophisticated molecular design. Taking a typical pH-indicator probe as an example, its core structure contains three functional modules: a fluorophore, a spacer, and a pH-responsive receptor.
pH Response and Fluorescence Regulation:
The weakly basic group in the probe (pKa usually 5.0–6.0) is deprotonated under near-neutral conditions, at which point the fluorophore may undergo fluorescence quenching via mechanisms such as photo-induced electron transfer (PET) or intramolecular charge transfer (ICT). When the probe accumulates in acidic lysosomes, protonation alters the molecular electron distribution, inhibits quenching pathways, and thereby enhances the fluorescence signal.
Selective Retention Mechanism:
Protonated probe molecules carry a positive charge, and the interior of the lysosomal membrane is positively charged relative to the cytoplasm. This electrochemical gradient further promotes probe accumulation in lysosomes. Meanwhile, protonation reduces membrane permeability, making it difficult for the probe to diffuse out freely, achieving selective retention characterized by "easy entry, difficult exit".
Spectral Property Optimization:
Different fluorophore designs offer diverse spectral options. Probes with near-infrared emission wavelengths (e.g., Ex/Em 649/665 nm or 577/590 nm) exhibit better tissue penetration and lower autofluorescence background in biological samples, suitable for deep-tissue imaging and multi-color labeling experiments.
Which experimental scenarios best reflect its technical advantages?
Morphology and Distribution Analysis of Lysosomes in Living Cells
Traditional immunofluorescence methods require cell fixation and permeabilization, which cannot reflect the true dynamics of lysosomes. pH-responsive fluorescent probes are specifically designed for live-cell imaging, enabling labeling of lysosomes under physiological conditions to observe their morphology, size, quantity, and distribution in real time. Using fluorescence microscopy, researchers can track lysosomal movement and analyze interactions with endosomes, autophagosomes, and other organelles.
Lysosomal Functional Status Assessment
Lysosomal pH is a key indicator of functional status. Abnormal lysosomal pH is commonly observed in tumor cells, neurodegenerative disease cells, or storage disorder cells. Using ratiometric pH probes (e.g., LysoSensor Yellow/Blue), quantitative measurement of lysosomal luminal pH can be achieved by detecting the ratio of fluorescence emission changes at different pH values, evaluating whether acidification function is normal.
Lysosomal Dynamics and Organelle Interactions
The development of super-resolution microscopy imposes higher requirements on the photostability of fluorescent probes. New-generation lysosomal probes feature excellent photostability and low cytotoxicity, supporting long-term continuous imaging (up to tens of minutes) to capture lysosomal fission, fusion, and contact events with mitochondria, endoplasmic reticulum, and other organelles. This is critical for cutting-edge research including mitochondrial-lysosomal contact (MLC) and mitophagy.
Drug Screening and Toxicity Evaluation
Many drugs and nanomaterials enter cells via the lysosomal pathway or induce lysosomal dysfunction. Lysosomal fluorescent probes can be used for high-throughput screening of compounds affecting lysosomal pH or integrity, evaluating lysosomal accumulation and potential toxicity of drugs. In drug delivery system research, probe labeling helps track intracellular trafficking and lysosomal escape of nanocarriers.
How to optimize live-cell staining experimental conditions?
Working Solution Preparation and Pre-treatment
Probes are typically supplied as 1 mM DMSO stock solutions and should be diluted to working concentrations (recommended 50–100 nM) with cell culture medium or PBS before use. High dilution ratios (1:10,000 to 1:20,000) help reduce background fluorescence and non-specific binding. Working solutions can be pre-warmed at 37°C to minimize temperature stress on cells.
Cell Staining and Incubation
Adherent Cell Protocol:
Remove cell culture medium, gently wash once with PBS, add pre-warmed probe working solution. Incubate at 37°C for 30 minutes to 2 hours, with the exact time optimized based on cell type and probe permeability. After incubation, wash three times with PBS to remove unbound probe before imaging.
Suspension Cell Protocol:
Collect cells by centrifugation, discard supernatant, resuspend cells in pre-warmed probe working solution, and incubate at 37°C for 30 minutes to 2 hours. After centrifugation and washing, resuspend in fresh medium for fluorescence microscopy or flow cytometry analysis.
Multi-color Labeling and Nuclear Staining
For multi-color imaging, lysosomal probes can be combined with nuclear dyes (e.g., Hoechst 33342, recommended concentration 10 μg/mL). Perform lysosomal staining first, wash with PBS, add nuclear dye, incubate at 37°C for 5 minutes, wash again, and observe lysosomes and nuclei simultaneously.
Imaging Notes
Photostability Protection:
Fluorescent dyes exhibit photobleaching; avoid prolonged intense light exposure during imaging. Use anti-fade reagents or select highly photostable probes to extend observation time.
Rapid Imaging:
Some lysosomal probes may undergo photochromism or signal attenuation under light; complete imaging promptly after staining to minimize light exposure.
Concentration Optimization:
If staining is insufficient, increase probe concentration or extend incubation time; if background is high, reduce concentration or shorten time to find the optimal signal-to-noise ratio balance.
Probe molecules selectively accumulate in acidic lysosomes via targeting groups such as morpholine, and fluorescence is enhanced upon protonation, enabling specific lysosomal labeling. The protonation state of the probe under different pH conditions determines its fluorescence emission properties
Considerations for Technology Selection
When selecting lysosomal fluorescent probes, comprehensively evaluate the following factors:
Spectral Compatibility:
Select matching excitation/emission wavelengths based on available laboratory equipment (lasers, filter sets). Common options include blue (Ex ~400 nm, Em ~425 nm), green (Ex ~500 nm, Em ~511 nm), yellow (Ex ~577 nm, Em ~590 nm), and red/near-infrared (Ex ~649 nm, Em ~665 nm) channels.
pH Sensitivity:
The probe’s pKa should be close to the physiological lysosomal pH (4.5–5.5) to ensure maximum fluorescence change sensitivity in this range. Ratiometric probes enable quantitative pH measurement, while non-ratiometric probes are more suitable for qualitative localization studies.
Cell Permeability:
Probes should exhibit excellent cell membrane permeability, freely entering living cells and being trapped by lysosomes with low cytotoxicity.
Photostability:
For long-term live-cell imaging or super-resolution microscopy, high photostability probes reduce photobleaching and provide more reliable dynamic data.
Conclusion
The development of lysosomal fluorescent probe technology provides a precise visualization tool for subcellular structure research in living cells. Probe designs based on the pH-response mechanism ingeniously utilize the inherent acidic microenvironment of lysosomes to achieve high-selectivity, high signal-to-noise ratio specific labeling. From basic lysosomal morphological observation to complex organelle interaction research, from drug screening to disease mechanism exploration, such probes demonstrate extensive application value in life science research. With continuous advances in fluorescent dye chemistry and imaging technology, new-generation lysosomal probes will achieve greater breakthroughs in sensitivity, photostability, and versatility, driving cell biology research toward deeper subcellular dynamic analysis.
Absin Lysosomal Fluorescent Probe Recommendation:
| Cat. No. | Product Name | Size |
|---|---|---|
| abs47038872 | Lysosome Deep Red Fluorescent Probe (FluoLyso Deep Red) | 50uL/50uL×10 |
| abs47038871 | Lysosome Red Fluorescent Probe (FluoLyso Red) | 50uL/50uL×10 |
| abs47038870 | Lysosome Green Fluorescent Probe (FluoLyso Green) | 50uL/50uL×10 |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |