- Cart 0
- English
How does the hydrolysis of β-1,4 glycosidic bonds become a fatal weakness of bacterial cells?
April 30, 2026
Clicks:76
During the long evolutionary process in nature, organisms have developed diverse defense mechanisms against microbial invasion. In 1922, Alexander Fleming discovered an enzyme capable of dissolving bacterial cell walls in chicken egg white and named it "Lysozyme". As a basic enzyme, it precisely recognizes specific chemical bonds in peptidoglycan of bacterial cell walls, making it a vital component of the body's innate immune defense. Lysozyme extracted from egg white, with its unique ability to hydrolyze β-1,4 glycosidic bonds, not only exerts antibacterial effects in organisms but also demonstrates extensive application value in molecular biology experiments, food industry, and bioengineering fields.
How does lysozyme recognize and destroy bacterial cell walls?
Lysozyme is a low-molecular-weight basic glycosidase with a molecular weight of approximately 14.4-14.8 kDa and an isoelectric point between pH 10.5-11.0, belonging to basic proteins. The core function of this enzyme is to catalyze the hydrolysis of β-1,4 glycosidic bonds between N-acetylmuramic acid (NAM) and N-acetylglucosamine (NAG) in the peptidoglycan backbone.
Substrate Recognition Mechanism:
The active center of lysozyme contains a cleft structure that specifically recognizes the hexasaccharide unit of the peptidoglycan backbone. The active site of the enzyme binds to the substrate through hydrogen bonds and hydrophobic interactions, precisely recognizing the spatial conformation of NAM and NAG residues.
Catalytic Hydrolysis Process:
- Binding Stage: Lysozyme binds to the peptidoglycan chain, and the Glutamic acid 35 (Glu35) and Aspartic acid 52 (Asp52) residues in the active center are positioned on both sides of the glycosidic bond to be hydrolyzed
- Protonation Stage: Glu35 donates a proton to protonate the oxygen atom of the glycosidic bond, forming an oxonium ion intermediate
- Hydrolytic Cleavage: The carboxylate anion of Asp52 stabilizes the carbocation intermediate, and a water molecule attacks the glycosidic bond, resulting in the cleavage of the β-1,4 glycosidic bond
- Product Release: Hydrolyzed soluble glycopeptide fragments are released from the cell wall, leading to the loss of structural integrity of the cell wall
Consequence of Cell Lysis:
The cell wall is a critical structure for bacteria to maintain osmotic pressure stability. After the peptidoglycan layer is hydrolyzed by lysozyme, the cell wall cannot resist internal osmotic pressure, and a large amount of water enters the cell, causing cell swelling and rupture (lysis), leakage of cellular contents, and bacterial death.
Why are Gram-negative bacteria naturally resistant to lysozyme?
Lysozyme exhibits obvious selectivity in its antibacterial spectrum, with significant effects on Gram-positive bacteria (such as Micrococcus lysodeikticus, Bacillus megaterium, Sarcina flava) and limited effects on Gram-negative bacteria. This difference stems from structural variations in bacterial cell walls:
Vulnerability of Gram-positive Bacteria:
- Cell wall thickness reaches 20-80 nm, and the peptidoglycan layer accounts for 50-90% of the cell wall dry weight
- The peptidoglycan layer is directly exposed to the external environment without outer membrane protection
- Lysozyme can easily access and hydrolyze the peptidoglycan backbone
Defense Mechanisms of Gram-negative Bacteria:
- Thin cell wall (2-7 nm), with peptidoglycan accounting for only 5-10%
- Outer membrane covers the outside of the peptidoglycan layer, composed of lipopolysaccharide (LPS), phospholipids, and lipoproteins
- Outer membrane forms an osmotic barrier that prevents macromolecular lysozyme from contacting the peptidoglycan layer
- Periplasmic space contains proteases that can degrade foreign proteins
Strategies to Enhance Sensitivity of Gram-negative Bacteria:
- Add EDTA to chelate Mg²⁺ in the outer membrane and disrupt outer membrane stability
- Use osmotic shock or freeze-thaw treatment to damage outer membrane integrity
- Combine with surfactants (such as Triton X-100, SDS)
Antiviral Mechanism: Electrostatic Binding and Double Salt Formation
In addition to antibacterial activity, lysozyme exhibits broad-spectrum antiviral capacity, and its mechanism of action is independent of enzymatic activity but relies on the electrostatic properties of the protein:
Electrostatic Binding:
Lysozyme is positively charged at physiological pH (basic protein) and can directly bind to negatively charged viral proteins (such as viral envelope proteins or capsid proteins).
Double Salt Formation:
Lysozyme forms insoluble complexes (double salts) with viral nucleic acids (DNA or RNA) and apoproteins, interfering with viral assembly, release, or infection processes and leading to viral inactivation.
Antiviral Spectrum:
This mechanism is effective against a variety of viruses, including DNA viruses and RNA viruses, providing a theoretical basis for the application of lysozyme in antiviral research and biomedicine fields.
Which experimental scenarios best reflect its technical advantages?
Bacterial Cell Lysis and Protein Extraction
In molecular biology experiments, lysozyme is the preferred tool for lysing Gram-positive bacteria:
- Prokaryotic Protein Expression: Escherichia coli (Gram-negative) expression systems usually require lysozyme-assisted lysis, especially when preparing periplasmic proteins or inclusion bodies. Although E. coli has outer membrane protection, lysozyme can effectively hydrolyze the peptidoglycan layer in combination with freeze-thaw or sonication treatment.
- Gram-positive Bacteria Research: For Gram-positive bacteria such as Staphylococcus aureus and Bacillus subtilis, lysozyme can be used alone to achieve gentle lysis and maintain protein activity.
- Cell Wall Preparation: Lysozyme can be used to prepare protoplasts (Gram-positive bacteria) or spheroplasts (Gram-negative bacteria) for cell wall structure research or protoplast fusion experiments.
Nucleic Acid Extraction and Purification
Lysozyme plays an auxiliary role in nucleic acid extraction:
- Bacterial DNA/RNA Extraction: Lyses bacterial cell walls to release nucleic acids, and improves extraction efficiency when used with proteinase K and SDS.
- Plasmid Extraction: In alkaline lysis method, lysozyme pretreatment can enhance lysis effect and improve plasmid yield.
Food Preservation and Biological Antisepsis
Lysozyme is widely used as a natural preservative in the food industry:
- Dairy Preservation: Inhibits Gram-positive contaminating bacteria (such as Listeria) in cheese and yogurt to extend shelf life.
- Meat and Seafood: Controls the growth of spoilage and pathogenic bacteria to maintain food freshness.
- Baby Food: As a natural antibacterial agent, it has high safety and is suitable for infant formula food.
Virology Research and Antiviral Development
Based on the antiviral properties of lysozyme:
- Viral Inactivation: Lysozyme can be used as a viral inactivator in the production of biological products (such as vaccines, blood products).
- Antiviral Drug Development: Studies the interaction mechanism between lysozyme and viruses to develop novel antiviral strategies.
Cell Culture and Tissue Processing
- Mucolysis: Lysozyme can degrade glycosaminoglycans in the extracellular matrix and is used to dissociate mucin-rich tissues.
- Cell Dispersion: In some tissue digestion protocols, lysozyme assists collagenase/trypsin to improve cell yield.
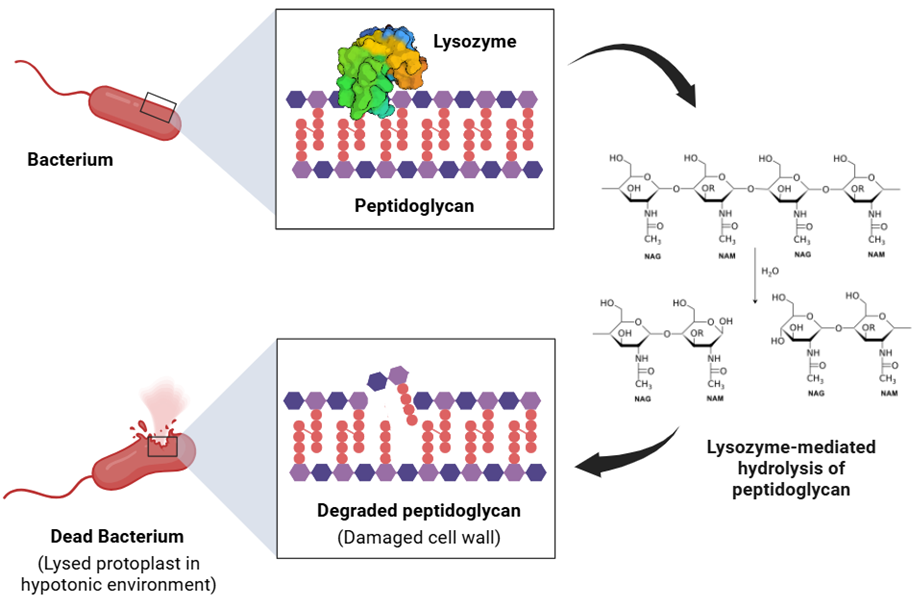
Figure: Schematic diagram of the mechanism by which lysozyme hydrolyzes bacterial cell walls. The top-left panel shows the binding of lysozyme to the peptidoglycan layer of the bacterial cell wall; the top-right panel displays the peptidoglycan structure, composed of alternating N-acetylmuramic acid (NAM, purple) and N-acetylglucosamine (NAG, red); the bottom panel shows that after lysozyme hydrolyzes the β-1,4 glycosidic bond, the peptidoglycan structure is destroyed, and bacteria lyse and die under osmotic pressure
How to Optimize the Lysis Efficiency of Lysozyme?
Optimization of Reaction Conditions
- pH Value: The optimal pH of lysozyme is 5.3-6.4 (acidic to weakly acidic), with the highest activity in the range of pH 6.0-7.0. Although lysozyme is a basic protein (isoelectric point pH 10.5-11.0), its enzymatic activity is optimal under slightly acidic conditions.
- Temperature: The optimal reaction temperature is 25-37°C, and it still has activity at 4°C but with reduced efficiency. Avoid high temperatures (>60°C) that cause protein denaturation and inactivation.
- Substrate Concentration: Ensure sufficient contact between lysozyme and the substrate. For high-density bacterial suspensions, appropriately increase the enzyme dosage or extend the reaction time.
Sample Pretreatment
Treatment of Gram-negative Bacteria:
- Add EDTA (1-2 mM) to chelate Mg²⁺ and disrupt outer membrane stability
- Freeze-thaw cycles (-80°C/37°C) to damage cell membrane integrity
- Osmotic shock (treatment with hypotonic buffer)
Reaction System Preparation:
- Common concentration: 100-500 μg/mL (for bacterial suspension)
- Buffer: 20-50 mM Tris-HCl (pH 7.0-8.0) or phosphate buffer (pH 6.0-7.0)
- Auxiliary additives: 0.1-1% Triton X-100 (enhances membrane permeability)
Enzyme Activity Protection
- Avoid Repeated Freeze-thaw Cycles: Lysozyme is a protein, and repeated freeze-thaw cycles lead to reduced activity. Aliquot storage is recommended.
- Storage Conditions: Lyophilized powder can maintain activity for several years at -20°C; after dissolution, short-term storage at 4°C (1-2 weeks) and several months at -20°C.
- Inhibitor Notes: Avoid long-term coexistence with metal ion chelators (such as EDTA at high concentrations), and some metal ions (such as Zn²⁺) may affect enzyme activity.
Considerations for Technical Selection
- Strain Type: Gram-positive bacteria can use lysozyme alone; Gram-negative bacteria need to be combined with EDTA or physical methods to enhance permeability.
- Experimental Purpose: Protein extraction requires gentle lysis (low concentration, short time); nucleic acid extraction can be combined with strong denaturants; food antisepsis needs to consider pH and temperature adaptability.
- Enzyme Activity Unit: Calculate the dosage according to the activity unit (U/mg or IU/mg) marked on the product, and adjust the usage amount for products of different purities (BR grade vs >95%).
- Cost-effectiveness: Egg white-derived lysozyme has low cost and is suitable for large-scale use; recombinant human lysozyme has higher purity and is suitable for biomedical applications.
Conclusion
As a natural "bacterial killer" endowed by nature, lysozyme plays a crucial role in the body's immune defense by precisely hydrolyzing peptidoglycan β-1,4 glycosidic bonds. This basic enzyme extracted from chicken egg white is not only a powerful tool for bacterial lysis and protein extraction in the laboratory but also shows broad application prospects in food preservation, antiviral research, and biomedicine fields. Understanding its catalytic mechanism and antibacterial spectrum characteristics helps researchers optimize usage conditions in molecular biology experiments, microbiology research, and bioengineering applications, and give full play to the technical advantages of this classic enzyme preparation.
Absin Lysozyme Recommendation
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
