- Cart 0
- English
How do aminoglycoside antibiotics achieve precise selection of eukaryotic cells?
April 30, 2026
Clicks:73
In the fields of molecular biology and cell engineering, the stable integration of exogenous genes into the host cell genome and the establishment of stably expressing cell lines are key steps for gene function research, recombinant protein production, and gene therapy studies. However, successfully transfected cells account for only a small minority, making the screening of positive clones from a large number of untransfected cells a technical bottleneck. G-418 Sulfate (Geneticin), an aminoglycoside antibiotic, blocks protein synthesis by interfering with eukaryotic cell ribosome function. Combined with the bacterial-derived resistance gene (neo), it has established a classic screening system for stable transfection of eukaryotic cells. This "toxicity-resistance" screening strategy has become a standard tool for genetic manipulation of eukaryotic cells including mammalian, plant, and yeast cells.
How does G-418 block protein synthesis through ribosome interference?
G-418 Sulfate (chemical name: Geneticin) belongs to the aminoglycoside antibiotic family, with a molecular structure consisting of amino sugars and aminocyclitols linked by glycosidic bonds. Unlike commonly used bacterial aminoglycoside antibiotics (e.g., gentamicin, kanamycin), G-418 exhibits a broader spectrum of biological activity and potent toxicity against eukaryotic cells.
Target of Action:
The primary target of G-418 is the 80S ribosome in eukaryotic cells. Upon entering cells, G-418 specifically binds to the decoding region of 18S rRNA and interferes with the function of the ribosomal A-site. This binding leads to two severe consequences:
- Translational Misreading: G-418 reduces translational fidelity, causing erroneous recognition between mRNA codons and tRNA anticodons, resulting in the incorporation of incorrect amino acids
- Translational Inhibition: At high concentrations, G-418 directly blocks polypeptide chain elongation, leading to the accumulation of immature polypeptide chains and ribosomal stalling
Cytotoxicity:
Due to the blockade of protein synthesis, cells cannot maintain normal metabolic activities, ultimately leading to cell death. G-418 is far more toxic to eukaryotic cells than prokaryotic cells, making it an ideal choice for eukaryotic cell screening.
Why can bacterial resistance genes function in eukaryotic cells?
The sophistication of the G-418 screening system lies in the functional expression of the bacterial-derived resistance gene (neo or aphA1) in eukaryotic cells. This "cross-kingdom" functionality is based on the following molecular mechanisms:
- Enzymatic Modification and Inactivation: The neo gene encodes aminoglycoside phosphotransferase (APT), which catalyzes the phosphorylation of specific amino groups in the G-418 molecule. The modified G-418 loses its ability to bind to ribosomes, thereby failing to interfere with protein synthesis.
- Dominant Selectable Marker: The neo gene is dominantly expressed in eukaryotic cells, and a single copy is sufficient to confer cellular resistance. This gene can be driven by various promoters (e.g., CMV, SV40, EF-1α) and is applicable to different cell types.
- Origin from Tn5 Transposon: The original neo gene is derived from the bacterial Tn5 transposon and can be efficiently expressed in eukaryotic cells including mammalian, plant, and yeast cells after codon optimization.
Which experimental scenarios best reflect its technical advantages?
Establishment of Stably Transfected Cell Lines
This is the most classic application scenario of G-418. After co-transfecting the target gene with the neo gene (usually on the same plasmid or different plasmids), under G-418 selection pressure, only cells that have successfully integrated and expressed the neo gene can survive and form colonies.
Operational Procedure:
- 48 hours after transfection, replace with medium containing G-418
- Replace fresh selection medium every 3-4 days
- Observe resistant colony formation after 7-10 days
- Pick single colonies for expansion culture
Gene Knockout and Functional Research
In CRISPR-Cas9 gene editing, G-418 is commonly used to screen cells with successful target gene knockout. By constructing knockout vectors containing the neo gene, or co-transfecting donor DNA containing neo, successfully edited cells can be enriched under G-418 pressure.
Generation of Transgenic Animals
In the generation of animal models such as transgenic mice and rats, G-418 is used to screen transfected embryonic stem cells or embryos after pronuclear microinjection. Positive embryos carrying transgenes can be rapidly identified through G-418 resistance screening.
Cross-Species Cell Screening
The broad-spectrum activity of G-418 makes it applicable to a variety of biological systems:
- Mammalian Cells: HEK293, CHO, HeLa, etc. (400-1000 μg/mL for screening, 200 μg/mL for maintenance)
- Plant Cells: Tobacco, Arabidopsis, etc. (25-50 μg/mL for screening, 10 μg/mL for maintenance)
- Yeast Cells: Saccharomyces cerevisiae, Pichia pastoris, etc. (500 μg/mL for screening, 125-200 μg/mL for maintenance)
- Protists: Dictyostelium, etc. (10-30 μg/mL)
How to establish a kill curve to determine the optimal screening concentration?
Different cell types vary significantly in sensitivity to G-418, and a kill curve must be established to determine the minimum lethal concentration before first use:
- Day 1: Seed untransfected cells at 20-25% confluence in a 24-well plate
- Day 2: Replace with medium containing gradient concentrations of G-418 (e.g., 0, 50, 100, 200, 400, 800, 1000 μg/mL), with 3 replicate wells per concentration
- Days 3-10: Replace drug-containing medium every 2 days and observe cell death
- Result Determination: Select the lowest concentration that kills the vast majority of cells within 7-10 days as the screening concentration
Concentration Reference Range:
- Mammalian cells: 200-2000 μg/mL (commonly 400-1000 μg/mL)
- Plant cells: 10-100 μg/mL
- Yeast cells: 500-1000 μg/mL
- Bacteria: 8-16 μg/mL
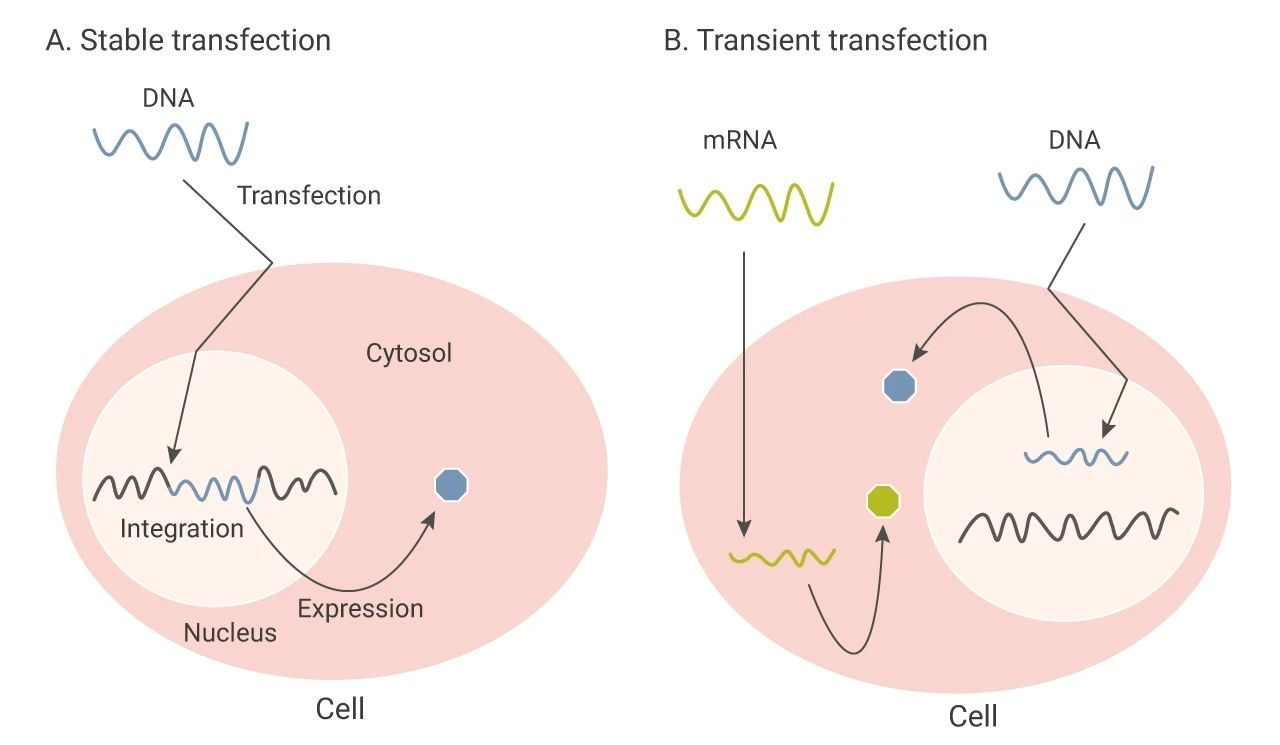
Figure: Schematic diagram of G-418 resistance screening mechanism and transfection types. The left panel shows stable transfection: exogenous DNA (containing resistance gene) integrates into the cell genome, and resistant cells survive and form colonies under G-418 selection pressure; the right panel shows transient transfection: exogenous DNA does not integrate and is only transiently expressed, and cells die under G-418 screening.
Key Control Points for Stable Transfected Cell Screening
- Cell Density Control: Cell density during screening should not be too high (recommended <25%) to ensure G-418 effectively kills untransfected cells. Over-confluent cells reduce screening efficiency and lead to false-positive colonies.
- Screening and Maintenance Concentrations: Usually use a high concentration (e.g., 800 μg/mL) for initial screening, and switch to a lower concentration (e.g., 200 μg/mL) for maintenance culture after obtaining resistant colonies to reduce cellular metabolic burden.
- Medium Replacement Frequency: Replace fresh G-418-containing medium every 3-4 days to maintain stable drug concentration and avoid metabolite accumulation.
- Colony Picking: After 7 days of screening, mark positive colonies under a microscope, pick them using cloning rings or limiting dilution method, and transfer to 35mm culture dishes for further expansion.
Considerations for Technical Selection
- Cell Type Differences: The sensitivity of different species and cell lines to G-418 can vary by 10-100 folds, and optimal concentrations must be determined through preliminary experiments.
- Plasmid Design: The neo gene can be located on the same plasmid as the target gene (cis) or different plasmids (trans), with cis co-transfection having higher efficiency.
- Screening Time: Rapidly dividing cells usually develop resistant colonies in 7-10 days, while slow-growing cells may take 2-3 weeks.
- Alternative Solutions: For G-418-resistant cell lines, other selection markers such as Puromycin, Hygromycin B, or Blasticidin can be considered.
Storage and Usage Precautions
- Stock Solution Preparation: Commonly prepared as 50 mg/mL stock solution, dissolved in 100 mM HEPES (pH 7.3) or PBS, filter-sterilized with 0.22 μm filter, aliquoted and stored at -20°C (valid for 12 months) or 4°C for short-term storage (1 month).
- Working Solution Stability: G-418 solution is thermally stable, but it is recommended to prepare fresh and avoid repeated freeze-thaw cycles.
- Autoclaving: G-418 cannot be autoclaved and must be added to the medium after filter sterilization.
- Safety Protection: G-418 is a toxic substance. Wear lab coats and gloves during operation to avoid inhaling dust or skin contact.
Conclusion
The combination of G-418 Sulfate and the neo resistance gene constitutes the most classic and reliable screening system in eukaryotic cell genetic manipulation. Through the toxicity mechanism of blocking protein synthesis by interfering with ribosome function, combined with the bacterial-derived phosphotransferase resistance mechanism, precise screening of transfected cells is achieved. From mammalian cell culture to transgenic animal generation, from gene function research to recombinant protein production, G-418 screening technology has become a basic tool in modern molecular biology laboratories. Mastering its mechanism of action, concentration optimization, and screening strategies will help researchers efficiently establish stable cell lines and promote the in-depth development of genetic engineering and cell biology research.
Recommended Absin G-418 Sulfate Products
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
