- Cart 0
- English
How do 583 amino acid residues construct a universal protein barrier in the laboratory?
April 29, 2026
Clicks:71
In life science research, one protein resides on the reagent shelf of almost every molecular biology laboratory—Bovine Serum Albumin (BSA). A globular protein purified from bovine serum via the Cohn cold ethanol fractionation method (Fraction V), BSA serves as a core reagent for Western Blot blocking, cell culture, protein quantification standards, and immunoassay support due to its unique molecular structure, excellent stability, and versatility. Composed of a single polypeptide chain folded by 583 amino acid residues and precisely cross-linked by 17 pairs of disulfide bonds, it forms a stable protein structure with a molecular weight of approximately 66 kDa and an isoelectric point of 4.7, playing an irreplaceable role in multiple fields of modern biotechnology.
Why Does BSA Simultaneously Achieve Stability, Binding, and Blocking Functions?
The versatility of BSA stems from its sophisticated molecular architecture and physicochemical properties:
Structural Stability:
The secondary structure of BSA is rich in α-helices, folded into a characteristic heart-shaped three-dimensional conformation. Seventeen pairs of disulfide bonds act as molecular "rivets", locking the polypeptide chain in a stable state, conferring excellent thermal and chemical stability. It maintains solubility and functional activity over a wide pH range (6.5-7.2) and temperature range.
Ligand Binding Capacity:
The BSA molecule possesses multiple ligand-binding sites on its surface, capable of reversibly binding hydrophobic or charged molecules such as fatty acids, bilirubin, calcium ions, and copper ions. This "molecular carrier" property not only maintains its physiological transport function but also provides a basis for stabilizing hydrophobic reagents in the laboratory.
High Solubility and Compatibility:
BSA is highly soluble in water and can be prepared into high-concentration solutions (up to 50 mg/mL or more) with low viscosity, facilitating precise pipetting. Its acidic nature (pI 4.7) renders it negatively charged at physiological pH (7.4), favoring the regulation of interactions with positively charged surfaces.
How Does the Protein Barrier Block Non-Specific Signal Interference?
In immunoassay techniques, BSA constructs a "protein barrier" through multiple mechanisms:
Steric Hindrance Effect:
BSA molecules (66 kDa) form a dense protein monolayer on the surface of solid-phase carriers (nitrocellulose membranes, PVDF membranes, or microplates), physically occupying the hydrophobic sites and pores of the carrier, preventing subsequent antibodies or detection proteins from contacting these sites.
Charge Neutralization:
With an isoelectric point of 4.7, BSA carries a negative charge in physiological pH (7.4) buffers, neutralizing positively charged regions on the carrier surface and reducing non-specific binding caused by electrostatic adsorption.
Hydrophobic Interaction Blocking:
Hydrophilic amino acid residues on the BSA surface form a hydration layer, masking hydrophobic regions on the carrier surface, thereby inhibiting non-specific adsorption mediated by hydrophobic interactions.
Antibody Dilution Stabilization:
Adding BSA (0.5-2%) to antibody diluents prevents non-specific aggregation of low-concentration antibodies or adsorption to container walls, maintaining the effective concentration and activity of antibodies.
Which Experimental Scenarios Best Demonstrate Its Technical Advantages?
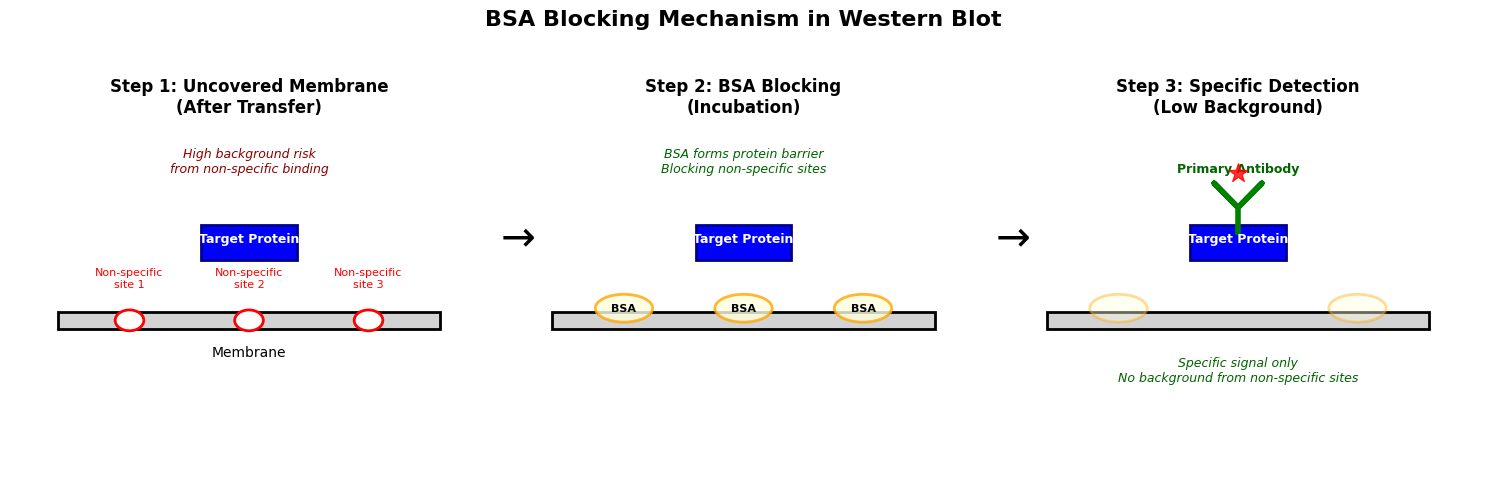
Figure: Schematic diagram of the BSA blocking mechanism in Western Blot. The left panel shows non-specific binding sites (red circles) on the membrane after transfer, posing a high background risk; the middle panel shows BSA molecules (yellow ovals) adsorbed on the membrane surface via incubation, forming a protein barrier to block non-specific sites; the right panel shows that after blocking, the primary antibody (green Y-shape) binds only to the specific target protein (blue rectangle), producing clear specific signals with significantly reduced background.
Western Blot Blocking
This is the most classic application of BSA. After protein electrophoresis and membrane transfer, incubate with 3-5% BSA solution (dissolved in TBST or PBST) at room temperature for 1-2 hours (or overnight at 4°C), which can effectively block unoccupied hydrophobic sites on the membrane, significantly reduce non-specific binding of primary and secondary antibodies, and improve the signal-to-noise ratio.
Enzyme-Linked Immunosorbent Assay (ELISA)
In ELISA, BSA is used to block unbound antigen/antibody sites on microplates and as an additive in antibody and antigen diluents to prevent protein adsorption to tube walls or denaturation, while reducing non-specific signals.
Cell and Tissue Culture
BSA is a critical additive in serum-free media, acting as a carrier protein for transporting hydrophobic molecules such as fatty acids and hormones; adding BSA to cell cryopreservation media protects cell membranes and reduces ice crystal damage; differentiation culture of certain cell lines also requires specific concentrations of BSA support.
Protein Quantification Standard
Due to high purity and stability, BSA serves as the standard protein for protein quantification methods such as Bradford, BCA, and Lowry assays, accurately determining protein concentrations in unknown samples by plotting standard curves.
Immunofluorescence and Flow Cytometry
Used for cell and antibody preparation blocking to reduce non-specific fluorescent signals and improve detection specificity and accuracy.
How to Select the Appropriate Grade Based on Experimental Needs?
Purity requirements for BSA vary significantly across different experiments:
Standard Grade (≥96% purity):
Suitable for general experiments such as Western Blot blocking, routine ELISA, and antibody dilution. Cost-effective and ideal for large-scale use. May contain trace amounts of fatty acids, globulins, and proteases.
High Purity Grade (≥98% purity):
Suitable for cell culture and protein standard preparation. Low endotoxin levels reduce cytotoxicity, and protease-free activity protects sensitive proteins.
Special Purified Grade (≥99% purity):
- Fatty Acid-Free BSA: Endogenous fatty acids removed, suitable for lipid metabolism research, fatty acid-sensitive cell culture, hormone or cholesterol analysis.
- Protease-Free BSA: Protease activity removed, suitable for enzyme activity assays and protease-sensitive experiments.
- IgG-Free BSA: Immunoglobulins removed, suitable for immunoprecipitation, co-immunoprecipitation, avoiding endogenous antibody interference.
- Low Endotoxin/Virus-Free BSA: Suitable for high-standard applications such as vaccine production, stem cell culture, and in vivo experiments.
What Are the Key Control Points for Optimizing Usage Conditions?
Preparation and Dissolution:
- Working concentrations: 3-5% for blocking, 0.5-2% for antibody dilution, 0.5-4 mg/mL for cell culture.
- Dissolution buffers: TBST (Western Blot), PBST (ELISA), PBS (cell culture).
- Dissolution method: Gently stir at room temperature to dissolve, avoid vigorous shaking to prevent foaming; for rapid dissolution, gently heat at 37°C, but avoid prolonged high temperatures.
Filter Sterilization:
- For cell experiments, dissolved BSA solution must be filter-sterilized through a 0.22 μm syringe filter.
- Autoclaving is strictly prohibited, as high temperatures cause protein denaturation and aggregation precipitation.
Storage Conditions:
- Lyophilized powder: Store dry at 4°C or -20°C to avoid moisture absorption and caking.
- Stock solution: Short-term storage at 4°C (1-2 weeks), long-term storage at -20°C (6-12 months). Aliquot before storage to avoid activity loss from repeated freeze-thaw cycles.
Contamination Control:
- BSA solution is nutrient-rich and prone to bacterial growth; prepare fresh before use or add preservatives (e.g., 0.02% sodium azide) for non-cell experiments.
- Maintain aseptic technique during preparation and use to avoid microbial contamination.
Conclusion
With its stable molecular structure (583 amino acids, 17 pairs of disulfide bonds), excellent solubility, and versatility, Bovine Serum Albumin has become an indispensable "universal reagent" in life science research. From the classic Cohn Fraction V to modern high-purity special grades, the quality of BSA continues to improve and its application fields expand. Understanding the characteristic differences of different grades, selecting precisely according to experimental needs, and mastering correct preparation and usage methods are the keys to fully leveraging BSA's technical advantages. In proteomics, immunology, and cell biology research, this 66 kDa globular protein will continue to play a fundamental and important role.
Absin Bovine Serum Albumin Products
| Cat. No. | Product Name | Size |
|---|---|---|
| abs9157 | Bovine Serum Albumin | 100g |
| abs9156 | Bovine Serum Albumin (Cell Culture Grade) | 50g/100g/500g |
| abs9262 | Bovine Serum Albumin (Fatty Acid-Free) | 100g |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
