- Cart 0
- English
Why is pertussis toxin a key molecular tool in neuroimmunology research?
April 28, 2026
Clicks:71
Pertussis Toxin (PTX), a bacterial exotoxin derived from Bordetella pertussis, has become an essential core reagent for the establishment of the Experimental Autoimmune Encephalomyelitis (EAE) model due to its unique enzymatic activity. Through specific post-translational modification mechanisms, this toxin profoundly regulates the migratory capacity and differentiation direction of immune cells, providing a standardized animal model foundation for the research of central nervous system demyelinating diseases such as Multiple Sclerosis (MS).

Figure: ADP-ribosylation mechanism of PTX
How Does Pertussis Toxin Regulate Cellular Signaling via Molecular Mechanisms?
PTX is a multi-subunit protein complex with core enzymatic activity of ADP-ribosyltransferase. Specifically, PTX recognizes and modifies cysteine residues on the α-subunit of Gi proteins (GTP-binding inhibitory regulatory proteins). It permanently inactivates Gi proteins by covalently conjugating ADP-ribose moieties.
This modification exerts two critical biological effects:
- Blockade of inhibitory signal transduction: After Gi protein inactivation, GTP hydrolysis to GDP is inhibited, leading to persistent activation of downstream adenylyl cyclase and abnormal elevation of intracellular cAMP levels
- Disruption of GPCR regulation: PTX suppresses and modulates various Gi protein-coupled chemokine receptors, impairing the normal response of immune cells to chemotactic signals
Relying on its capacity to block Gi protein-coupled receptor-mediated signaling pathways, PTX alters the directional migration of immune cells and enables their penetration across the blood-brain barrier into the central nervous system.
Why Is PTX an Essential Adjuvant for EAE Model Establishment?
The EAE model is the most classic animal model for MS research, and PTX plays multiple pivotal roles in this model:
1. Promotion of Central Nervous System Infiltration
By inhibiting Gi protein-coupled chemokine receptors on the surface of T cells, PTX disrupts normal immune surveillance mechanisms, allowing antigen-specific T cells (primarily Th1 and Th17 cells) to infiltrate the central nervous system. This immunomodulatory function is critical for breaking self-antigen tolerance and establishing stable autoimmune responses.
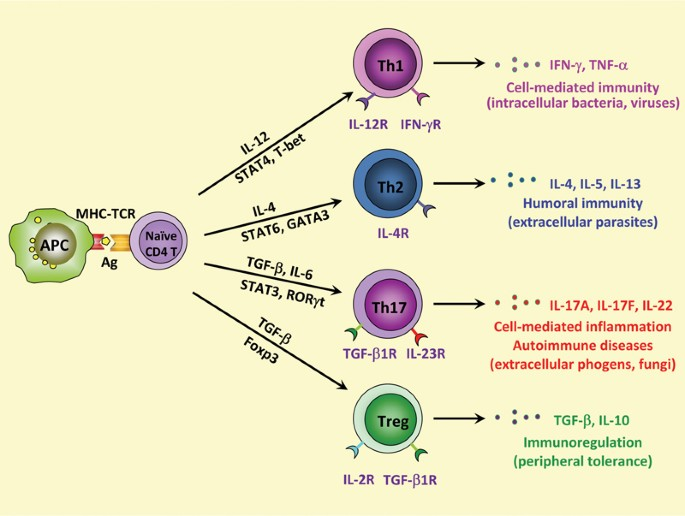
Figure: Th1/Th17 cell differentiation and EAE pathogenesis
2. Modulation of T Cell Differentiation
Studies have demonstrated that PTX induces IL-6 production in Th cells, a prerequisite for the differentiation of naive T cells into pathogenic Th1/Th17 cells. By regulating the IL-6 signaling pathway, PTX affects subsequent disease severity and disease progression.
3. Breakdown of Immune Tolerance
In EAE models induced by MOG (Myelin Oligodendrocyte Glycoprotein) or PLP (Proteolipid Protein), PTX synergizes with antigen emulsions to markedly break immune tolerance to endogenous myelin components and improve the success rate of model induction.
How to Design a Standardized PTX Administration Protocol?
Successful EAE modeling relies on precise PTX dosage control and administration timing. The standard operating procedure is as follows:
Key Points for Solution Preparation
- Stock Solution Preparation: Reconstitute lyophilized PTX with sterile double-distilled water to prepare a high-concentration stock solution of 100 μg/mL (100 ng/μL), aliquot and store at -20°C
- Working Solution Dilution: Dilute the stock solution to a working concentration of 2 ng/μL with sterile PBS before use
- Stability Notes: Avoid repeated freeze-thaw cycles; reconstituted toxin should be used in one single dose or stored in strictly divided aliquots
Immunization Protocol (C57BL/6 Mice Induced by MOG35-55 as an Example)
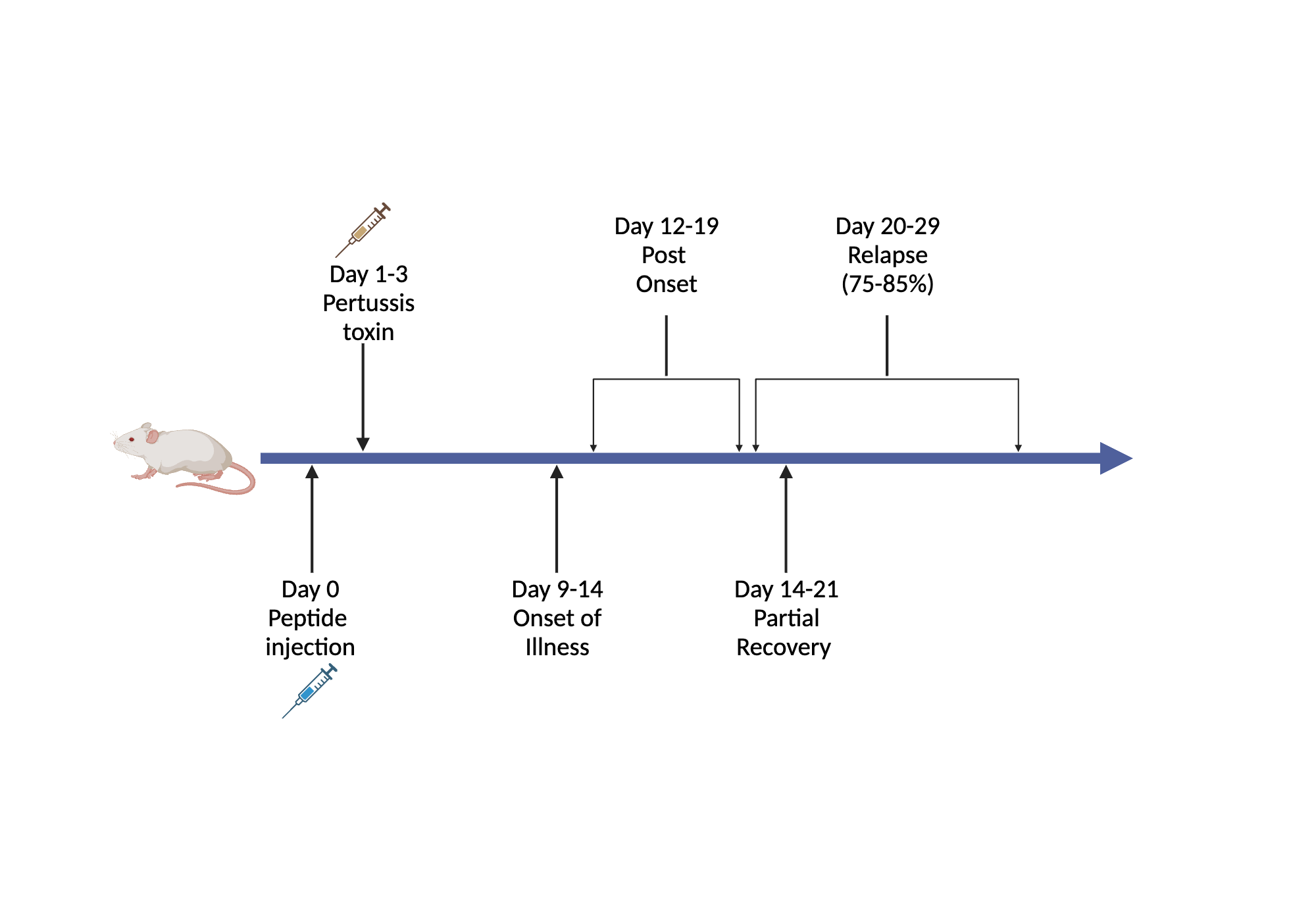
Figure: EAE model construction timeline
- Day 0 (D0): Subcutaneous injection of antigen emulsion (complete Freund's adjuvant containing 200 μg MOG35-55 and heat-killed Mycobacterium tuberculosis H37 Ra)
- D0 Same Day: Intraperitoneal injection of PTX (200 ng per mouse, approximately 100 μL working solution)
- D2 (48 Hours Later): Second intraperitoneal injection of PTX (200 ng per mouse)
This classic protocol of "antigen sensitization plus PTX challenge" can induce obvious clinical EAE symptoms within 10–14 days after immunization.
Why Does Emulsion Quality Directly Affect Modeling Success Rate?
The emulsification quality of antigens and adjuvants is a prerequisite for successful EAE modeling. Substandard emulsions lead to excessive antigen release, insufficient immune responses or local tissue necrosis.
Emulsification Quality Evaluation Criteria:
Drop the prepared emulsion onto the water surface. A qualified emulsion should remain non-diffusible and non-dispersible with an intact droplet morphology. Rapid spreading on the water surface indicates an incomplete water-in-oil structure, and re-emulsification is required.

Figure: Schematic diagram of subcutaneous injection
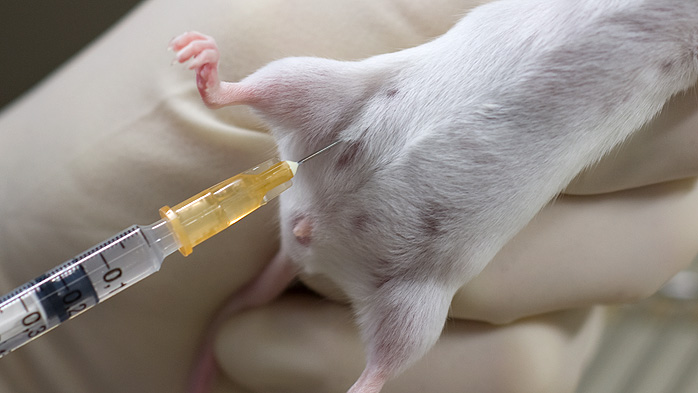
Figure: Intraperitoneal injection technique in mice
Operational Precautions:
- Perform subcutaneous injection bilaterally along the dorsal spinal column (avoid intramuscular injection), generally four-point injection (50 μL per site, 200 μL emulsion in total)
- Avoid visceral organs during intraperitoneal PTX injection; adopt the Trendelenburg position to ensure injection into the lower abdominal cavity
How to Scientifically Evaluate the Establishment of EAE Models?
After successful modeling, mice usually exhibit progressive neurological symptoms, which require multi-dimensional verification:
Clinical Scoring System (0–5 Scale)
Auxiliary Verification Indicators:
- Body Weight Monitoring: The body weight of diseased mice usually decreases by more than 10%. Continuous monitoring can objectively reflect disease progression
- Histopathology: Spinal cord sections stained with HE show inflammatory cell infiltration; LFB (Luxol Fast Blue) staining reveals demyelination
- Molecular Biology Detection: Flow cytometry for the proportion of Th1 (CD4+IFN-γ+) and Th17 (CD4+IL-17+) cells; ELISA for detection of serum/spinal cord cytokines (TNF-α, IL-6, IL-17, IL-10, etc.)
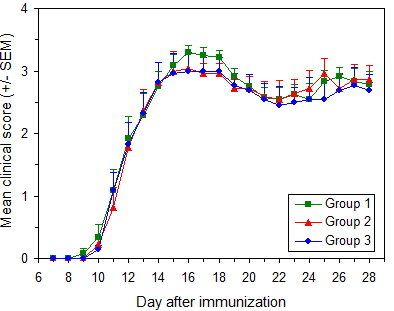
Figure: EAE model disease course curve
Frequently Asked Questions in PTX Application
Q1: Is slight turbidity after PTX reconstitution normal with incomplete dissolution?
Slight turbidity after reconstitution is normal and does not affect subsequent use. To ensure full dissolution, recommendations are as follows:
- Gently pipette up and down for mixing, avoid vigorous shaking or vortexing
- Do not filter-sterilize PTX solution (will cause protein loss)
- In case of obvious precipitation, perform brief centrifugation and collect the supernatant for use
Q2: Why do mice show no obvious symptoms after modeling?
The success rate of EAE modeling is affected by multiple factors:
- Insufficient antigen emulsification: A stable water-in-oil emulsion must be formed
- Insufficient PTX dosage: 200 ng per mouse per administration with two injections is recommended
- Individual animal differences: Female C57BL/6 mice (8–10 weeks old) generally have a higher modeling success rate
- Adjuvant activity: Ensure the use of freshly prepared complete Freund's adjuvant containing heat-inactivated mycobacteria
Q3: Large-area ulceration or necrosis at the injection site?
This condition is usually caused by overly shallow subcutaneous injection (intradermal injection) or poor emulsion quality. Ensure the needle is accurately placed in the subcutaneous loose connective tissue layer, and confirm the formation of a subcutaneous vesicle-like bulge after injection.
Q4: How to reduce high mortality?
Potential causes include excessive PTX dosage (no more than 500 ng per mouse per time) or secondary infection. Strict aseptic operation is required. Strengthen nursing care during the peak disease period (D14–18) and provide accessible drinking water and soft feed.
Technical Parameters and Storage Guidelines
Product Properties:
- Purity: >99% (verified by high-performance liquid chromatography)
- Appearance: White to off-white lyophilized powder
- Molecular Mechanism: ADP-ribosylation of Gi protein α-subunit and inhibition of GPCR signal transduction
- Alternative Names: Adenylate Cyclase Toxin, PT, Ptx
Safety Precautions for Use:
PTX is a bioactive substance. Wear protective gloves, masks and goggles during operation to avoid inhalation or skin contact. Experimental waste shall be disposed of in accordance with biosafety regulations.
Through precise control of PTX dosage and administration timing combined with standardized immunization procedures, researchers can establish stable and reproducible EAE models, providing a reliable experimental basis for exploring MS pathogenesis, evaluating immunomodulatory therapeutic strategies and drug screening.
Absin Pertussis Toxin Recommendation
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
