- Cart 0
- English
Why is thrombin both the core enzyme of the coagulation cascade and an important tool enzyme in protein engineering?
April 28, 2026
Clicks:73
Thrombin is a serine protease derived from bovine plasma with a molecular weight of approximately 37 kDa. It consists of one light chain (~6 kDa) and one heavy chain (~31 kDa) covalently linked via disulfide bonds. As the activated form of prothrombin (Coagulation Factor II) stimulated by Factor Xa, thrombin acts as an irreplaceable core regulator in the blood coagulation cascade. It specifically cleaves the Arg-Gly bond in fibrinogen molecules, releasing fibrinopeptide A (FPA) and fibrinopeptide B (FPB), which facilitates the polymerization of fibrin monomers to form stable blood clots. Beyond its inherent physiological functions, thrombin has become a critical tool enzyme in recombinant protein purification, platelet function assay and blood product quality control due to its highly specific recognition sequence and efficient hydrolytic activity.
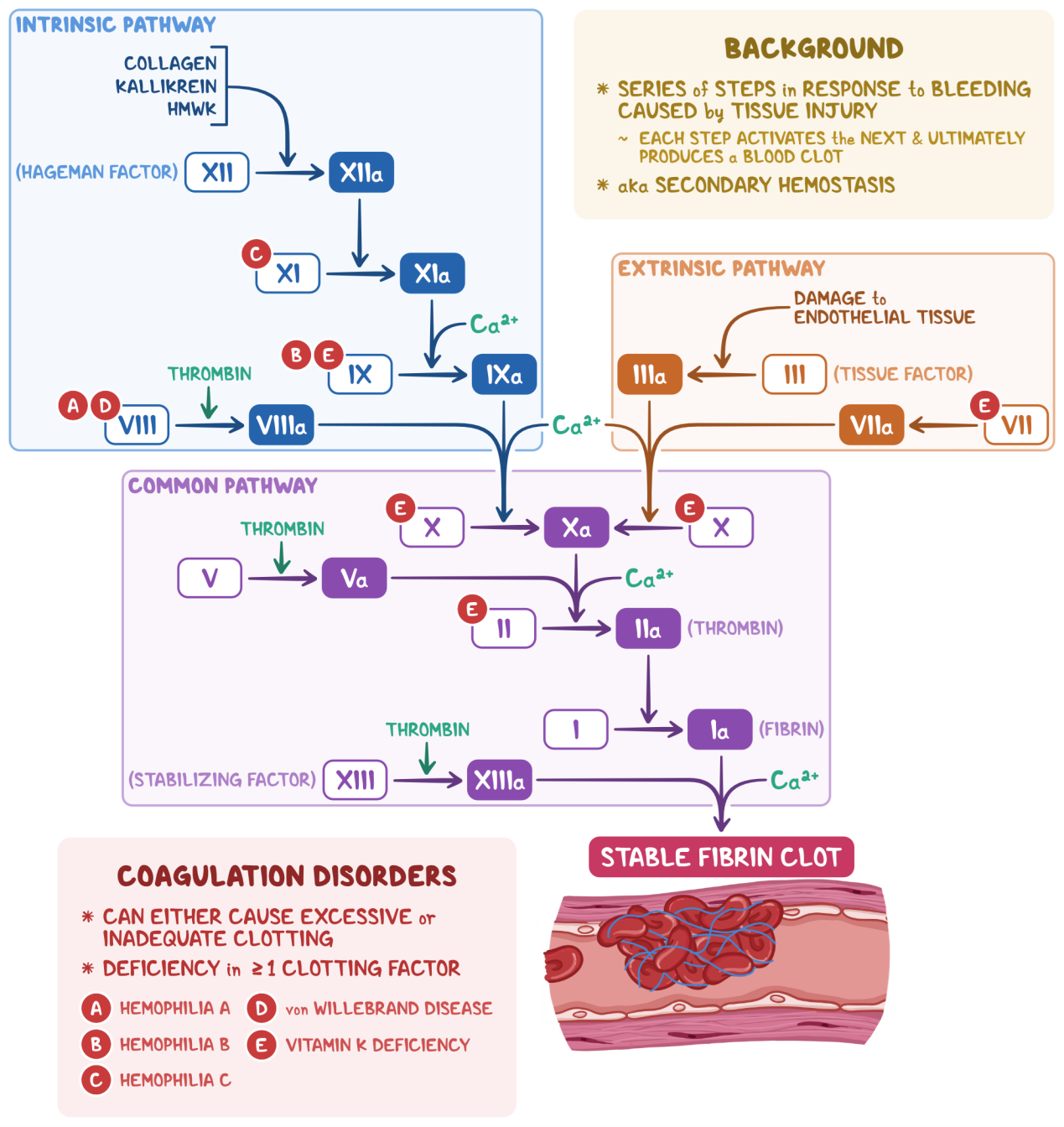
Schematic Diagram of Coagulation Cascade
How Does Thrombin Function as a Core Molecule in the Coagulation Cascade?
Thrombin is a key effector molecule in the common pathway of the coagulation cascade. After the endogenous pathway (contact activation) and exogenous pathway (tissue factor-initiated) jointly activate Factor X, activated Factor X (Xa) forms the prothrombinase complex together with Factor Va, phospholipids and calcium ions. This complex hydrolyzes inactive prothrombin (Factor II) into biologically active α-thrombin.
Multiple Biological Functions of Thrombin:
- Fibrinogen Conversion: Cleaves the Aα and Bβ chains of fibrinogen to release fibrinopeptides, enabling spontaneous assembly of fibrin monomers into fibrin polymers
- Factor XIII Activation: Activates Coagulation Factor XIII to promote fibrin cross-linking and form stable, lytic-resistant fibrin clots
- Positive Feedback Regulation: Activates Factor V and Factor VIII to amplify coagulation signals; meanwhile triggers the protein C pathway to participate in anticoagulant regulation

Fibrinogen Structure and Thrombin Cleavage
Why Is Thrombin the Preferred Tool Enzyme for Fusion Protein Cleavage?
In genetic engineering, target proteins are usually fused with affinity tags (e.g., His-tag, GST-tag) to facilitate recombinant protein expression and purification. Removal of these tags is essential to obtain native target proteins, and thrombin serves as an ideal tool for this process.
Technical Advantages:
- High Specificity of Recognition Sequence: The canonical recognition motif of thrombin is X4-X3-P-R[K]-X1'-X2', in which X4 and X3 represent hydrophobic amino acids, while X1' and X2' are non-acidic amino acids
- Efficient Cleavage Capacity: Effectively hydrolyzes protein substrates at a mass ratio of 1:2000; complete cleavage can be achieved within 0.5–16 hours under 20–37°C and pH 8.0
- Easy Removal: Can be specifically eliminated from cleavage products via affinity chromatography such as p-aminobenzamidine agarose columns or benzamidine agarose resins
Common Recognition Sites:
- L-V-P-R-G-S (Leu-Val-Pro-Arg-Gly-Ser)
- L-V-P-R-G-F (Leu-Val-Pro-Arg-Gly-Phe)
- M-Y-P-R-G-N (Met-Tyr-Pro-Arg-Gly-Asn)
Optimization Strategies:
Insertion of five glycine residues (glycine linker) between the thrombin cleavage site and N-terminal tag can significantly improve cleavage efficiency, reduce non-specific digestion, and achieve complete cleavage with a lower enzyme dosage.
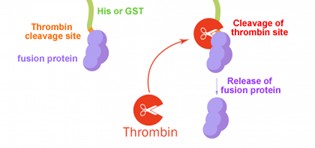
Principle of Fusion Protein Cleavage
Applications of Thrombin in Platelet Function Research
Thrombin is a potent agonist for platelet aggregation. It binds to protease-activated receptor 1 (PAR-1) on the platelet surface, triggering platelet activation, morphological transformation, granule release and cell aggregation. This property makes thrombin a standard agonist for clinical and laboratory platelet function detection.
Standard Protocol for Platelet Aggregation Assay:
- Blood Collection: Collect whole blood in plastic tubes and add anticoagulant (e.g., 86 mmol/L sodium citrate mixed with blood at a volume ratio of 0.15)
- PRP Preparation: Centrifuge at 200 g for 10 minutes to obtain supernatant platelet-rich plasma (PRP)
- Platelet Counting: Adjust platelet concentration to (3–4)×10¹¹/L
- Aggregation Detection: Pre-incubate 0.2 mL platelet suspension with test agents for 1 minute, then add 500 U/L thrombin and incubate for 5 minutes
- Result Measurement: Detect optical density changes via platelet aggregometer and calculate platelet aggregation rate
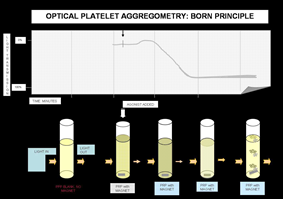
Principle of Platelet Aggregation Assay
Clinical and Scientific Research Value:
- Evaluate the therapeutic efficacy of antiplatelet drugs (e.g., Aspirin, Clopidogrel)
- Diagnose congenital platelet dysfunction such as Glanzmann thrombasthenia
- Explore the regulatory mechanism of thrombin receptor signaling pathways
Roles of Thrombin in Serum Preparation and Quality Control
In clinical laboratory testing and blood product manufacturing, thrombin is widely applied in defibrination, which converts fibrinogen in plasma or whole blood into insoluble fibrin for removal, so as to obtain fibrin-free serum or plasma samples.
Application Scenarios:
- Serum Quality Control Material Preparation: Remove fibrinogen via thrombin treatment to prevent fibrin clot formation during long-term storage
- Detection of Biochemical Indicators: Eliminate fibrin interference to ensure accurate quantification of serum proteins, electrolytes and other biomarkers
- Cell Culture Supplement Production: Prepare defibrinated serum to reduce experimental variables in culture systems

Differences between Plasma & Serum and Thrombin Function
Strategies to Maintain High Activity and Stability of Thrombin
Key Quality Indicators:
- Purity: No miscellaneous protein bands detected by SDS-PAGE to guarantee single enzymatic activity
- Specific Activity: ≥2000 U/mg protein for high catalytic efficiency
- Biochemical Specificity: Free of miscellaneous protease activity to avoid non-specific cleavage
- Biosafety: Double virus inactivation via S/D treatment and dry heat sterilization to meet biological safety standards
Storage & Handling Recommendations:
Lyophilized Powder:
- Stable for 12 months at room temperature (25°C) with no significant loss of enzyme activity
- 36-month shelf life under refrigeration (2–8°C)
- White lyophilized powder or block, soluble in deionized water (10 mg/mL)
Solution Preparation:
- Stock Solution: Prepare at 100 U/mL in 0.1% (w/v) BSA solution to reduce protein adsorption loss
- Short-term Storage: Maintain stable activity for one week at 0–5°C
- Long-term Storage: Aliquot into plastic tubes to avoid glass adsorption and store at -20°C
Reaction Condition Optimization:
- Buffer System: 20 mM Tris-HCl, 150 mM NaCl, pH 8.0
- Reaction Temperature: 20–37°C (room temperature to physiological temperature)
- Reaction Duration: 0.5–16 hours, adjustable according to substrate dosage and cleavage efficiency
Key Notes for Experimental Operation
Q1: How to Prevent Non-specific Cleavage of Thrombin?
Ensure no contamination of exogenous proteases in the reaction system, and strictly control reaction duration and enzyme dosage. For refractory fusion proteins, appropriately prolong incubation time or increase enzyme dosage; meanwhile monitor the cleavage process by SDS-PAGE to prevent over-digestion.
Q2: How to Remove Thrombin after Cleavage?
Affinity chromatography is the recommended method: p-aminobenzamidine agarose or benzamidine agarose resins can specifically bind thrombin while target proteins are collected in flow-through fractions. Commercial thrombin removal resins (Thrombin CleanCleave Resin) are also available for one-step elimination.
Q3: Key Factors Affecting Thrombin Activity
- Adsorption Loss: Thrombin tends to adhere to glass surfaces; plastic tubes are required for storage and reaction setup
- Temperature Variation: Avoid repeated freeze-thaw cycles; dissolve and aliquot lyophilized products in one-time operation
- pH Value: The optimal pH is 8.0; extreme acidic or alkaline conditions will sharply reduce enzymatic activity
Q4: How to Verify Thrombin Cleavage Efficiency?
Monitor via SDS-PAGE electrophoresis: the band intensity of fusion proteins gradually declines, accompanied by the emergence of independent tag and target protein bands. Incomplete cleavage can be improved by adjusting buffer pH, reaction temperature or eliminating steric hindrance.
With high substrate specificity, potent catalytic activity and convenient removal properties, thrombin has become an indispensable tool enzyme for basic biological research and biopharmaceutical production. Rational application of thrombin in recombinant protein purification, platelet functional research and blood product processing can greatly improve experimental efficiency and product quality.
Absin Thrombin Product Recommendation
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
