- Cart 0
- English
How to effectively inhibit fluorescence quenching to achieve high-quality microscopic imaging and long-term sample preservation?
April 28, 2026
Clicks:72
In experiments such as immunofluorescence, immunocytochemistry and immunohistochemistry, fluorescent labeling technology enables researchers to visualize specific target proteins at the cellular and subcellular levels. However, fluorescent substances are highly susceptible to photobleaching and fluorescence quenching under excitation light irradiation, resulting in rapid signal attenuation. This not only impairs the image quality of real-time observation, but also restricts the long-term preservation and repeated observation of precious samples. Anti-fade mounting medium forms a protective microenvironment by introducing anti-quenching components during the mounting process to significantly delay fluorescence decay, serving as a critical reagent for ensuring high-quality fluorescence microscopic imaging and sample archiving.
Mechanism and Protection of Fluorescence Quenching
How Does Fluorescence Quenching Occur?
Fluorescence quenching refers to the process in which excited-state fluorescent molecules return to the ground state by dissipating energy through various pathways without photon emission. The main mechanisms include:
- Photochemical Damage: Excited fluorophores react with molecular oxygen to generate reactive oxygen species such as singlet oxygen, which disrupt fluorophore structures
- Molecular Collision: Collisions between fluorescent molecules and solvent or solute molecules lead to energy dissipation
- pH Sensitivity: Certain fluorescent dyes are sensitive to pH fluctuations, and deviation from the optimal pH causes a decline in fluorescence intensity
- Aggregation-Caused Quenching: Fluorescent molecules form aggregates at high concentrations, inducing non-radiative energy transfer
Anti-fade mounting medium functions through the following mechanisms:
- Antioxidant Protection: Antioxidants are supplemented to neutralize free radicals and block photochemical reaction cascades
- Physical Isolation: Glycerol or polymer matrices reduce oxygen diffusion and alleviate oxidative stress
- Chemical Stabilization: Maintain an optimal pH microenvironment to preserve the structural integrity of fluorophores
Differences Between Various Types of Anti-fade Mounting Media
Based on compositional characteristics and application requirements, anti-fade mounting media are mainly classified into the following categories:
1. Glycerol-Based Anti-Fade Mounting Medium
Formulated with high-purity glycerol as the basal matrix and proprietary anti-fluorescence decay additives. Glycerol possesses a refractive index (~1.47) similar to biological tissues, which reduces light scattering and improves imaging resolution.
Characteristics:
- Potent anti-fluorescence decay effect, markedly prolonging the observation time window
- Good fluidity without permanent bonding between coverslips and slides (facilitating re-sampling)
- Warm up in a 37°C water bath before use to reduce viscosity
- Store slides at 4°C or -20°C away from light after mounting
Application Scenarios:
- Conventional immunofluorescence assays
- Adherent cell samples requiring short-term observation (hours to days)
- Real-time observation and imaging of tissue sections
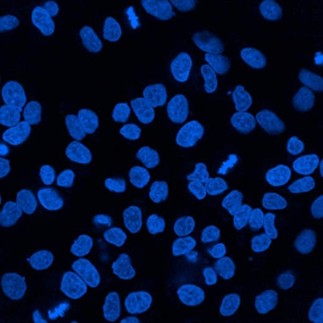
DAPI-Stained Cell Nuclei
2. Mounting Medium with Nuclear Dyes (DAPI/PI)
Nuclear dyes are pre-mixed into the basic anti-fade formula to realize one-step mounting and nuclear staining.
DAPI-Containing Mounting Medium:
- DAPI (4',6-diamidino-2-phenylindole) binds tightly to DNA, with a maximum excitation wavelength of 360 nm and emission wavelength of 460 nm, emitting blue fluorescence
- Permeable to intact cell membranes, suitable for both live and fixed cells
- Incubate for 30 minutes in the dark after mounting for sufficient dye binding
- Eliminate independent nuclear staining steps and simplify experimental workflows
PI (Propidium Iodide)-Containing Mounting Medium:
- The routine working concentration of PI is 6.7 μg/mL; it cannot penetrate intact cell membranes and only stains dead cells
- Excitation at 488 nm and emission at 630 nm with red fluorescence signals
- Applied in cell viability detection and cell membrane integrity assessment
- Use with cell staining buffer and incubate for 20-30 minutes at room temperature in darkness
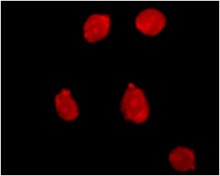
PI-Stained Dead Cells
3. Water-Soluble Semi-Permanent Mounting Medium
Formulated with non-fluorescent water-soluble polymers to provide semi-permanent immobilization and greatly extend the storage period of glass slide samples.
Characteristics:
- Compatible with aqueous systems without dehydration procedures
- Stable storage at room temperature and ready-to-use by direct dropping
- Contains preservatives (e.g., 0.1% sodium azide or natural essential oils)
- Ready for observation after air-drying for 5 minutes at room temperature in darkness
- Suitable for medium-to-long-term preservation at 2-8°C
Application Scenarios:
- Mounting of cell smears and coverslip cultures
- Immunohistochemistry of tissue sections
- Confocal microscopy detection with optimal refractive index matching
4. Crystal Mounting Medium (Curable Type)
A water-soluble gel mounting medium (pH 7.5, refractive index: 1.38-1.41) with rapid drying and thermosetting properties.
Advantages & Properties:
- Ultra-Long Storage Lifespan: TSA multiplex fluorescent samples can be stored for 6 months to 5 years under dark conditions
- Semi-Permanent Immobilization: Form stable solid mounting layers after baking at 37°C for over 30 minutes
- Natural Preservation: Adopts lavender, lily of the valley or green tea essential oils as preservatives, free of highly toxic components such as sodium azide
- Chemical Inertness: Does not alter the spectral characteristics of fluorescence signals, ideal for multicolor fluorescence analysis
Operation Key Points:
- Add 1-2 drops onto the sample and slowly cover with a coverslip to avoid air bubbles
- Dry in a 37°C incubator (moisture boxes are not recommended)
- Store at room temperature in darkness for 1 month, and preserve long-term at -20°C
- Coverslips can be detached by water bath incubation at 37-50°C for 1-12 hours for subsequent staining

Multicolor Fluorescence Imaging
Which Experiments Require Anti-Fade Mounting Medium?
Fluorescence Immunomicroscopy:
- Cellular Immunofluorescence (IF): Detection of intracellular protein localization and expression levels
- Tissue Immunohistochemistry (IHC): Observation of antigen distribution in tissue sections
- Immunocytochemistry (ICC): Immunofluorescence labeling of cell coverslips and smears
Specialized Imaging Technologies:
- Confocal Laser Scanning Microscopy (CLSM): Long-time scanning for multilayer image acquisition
- Super-Resolution Microscopy (STED/PALM/STORM): High-intensity laser irradiation easily induces fluorophore photobleaching
- Long-term live cell imaging: Reduce fluorescence decay caused by phototoxicity
Sample Archiving:
- Semi-permanent preservation of valuable clinical specimens
- Teaching slides requiring repeated observation
- Standardized sample preparation for multicenter research projects
Long-Term Preservation of Fluorescent Samples
Standard Mounting Operation Protocols
Protocol for Adherent Cell Samples:
- Complete Staining: Finish all staining procedures including primary antibody, secondary antibody and fluorescent dye incubation
- Remove Excess Liquid: Gently aspirate residual liquid on culture wells or slides with pipettes or absorbent paper and avoid over-drying
- Add Mounting Medium: Pipette 20-50 μL anti-fade mounting medium onto the slide
- Coverslip Sealing: Place the cell-side coverslip downward slowly to fully contact with mounting medium and prevent bubble formation
- Dark Incubation (If Needed): Incubate for 30 minutes in darkness for sufficient dye binding for DAPI-containing medium
- Microscopic Observation: Observe under a fluorescence microscope, and seal coverslip edges with nail polish for long-term storage if necessary
Protocol for Tissue Sections:
- Complete Staining: Complete immunohistochemical staining of tissue sections
- Discard Waste Liquid: Shake or aspirate redundant staining solution (additional washing is not required, especially for nuclear dye-containing medium)
- Apply Mounting Medium: Add 1-2 drops of mounting medium to the tissue section
- Coverslip Coverage: Gently place the coverslip to ensure full contact between sections and mounting liquid
- Remove Overflow Liquid: Blot excess liquid from the coverslip edge with absorbent paper (avoid pressing)
- Curing & Storage: Air-dry at room temperature or bake at 37°C according to medium types
Bubble Removal Tips:: If excessive bubbles interfere with observation, aspirate the mounting medium into a centrifuge tube, centrifuge at over 10,000 g for 10 minutes to eliminate bubbles, and re-mount the sample.
Long-Term Storage Strategies for Fluorescent Samples
Short-Term Storage (Within 1 Month):
- Store at room temperature or 4°C away from light (with preservative-containing mounting medium)
- Place in slide boxes to avoid extrusion and damage
- Return samples to dark conditions timely after observation
Long-Term Storage (Several Months to Years):
- Temperature Control: Store at -20°C in darkness to greatly enhance fluorescence stability
- Humidity Management: Ensure complete sample drying before -20°C preservation to prevent condensate damage caused by repeated freeze-thaw cycles
- Light Avoidance: Wrap with aluminum foil or store in light-tight boxes to prevent cumulative photodamage from ambient light
- Stability Difference: Curable crystal mounting medium enables 6-month to 5-year preservation of multicolor fluorescent samples at -20°C, while conventional glycerol-based medium is recommended for observation within 3-6 months
Important Reminder: Fluorophores still undergo slow attenuation even with anti-fade mounting medium. It is recommended to capture high-quality original images during the first observation and record exposure parameters for subsequent comparative analysis.
Key Precautions for Application
Safety Operation:
- Mounting medium containing sodium azide is toxic; wear protective goggles and gloves during operation to prevent skin and eye contact
- Natural essential oil-based medium features low toxicity, yet basic personal protection is still required
- Operate in a well-ventilated environment to avoid inhalation of volatile components
Technical Key Points:
- Dark Operation: Keep samples protected from light throughout the whole process from staining to imaging (use dark boxes or aluminum foil wrapping)
- Temperature Adjustment: Warm glycerol-containing medium in a 37°C water bath before use to reduce viscosity and improve spreadability
- Anti-Extrusion: Never press the coverslip after mounting to avoid cell deformation and structural damage
- Edge Sealing: Seal coverslip peripheries with nail polish for long-term storage or anti-volatilization
Compatibility Verification:
When using new types of mounting medium for the first time, conduct preliminary tests with 1-2 samples to confirm: spectral compatibility with existing fluorescent dyes (no autofluorescence interference), impacts on target protein antigenicity, and whether the pH value of the medium is suitable for specific fluorescent probes.
By rationally selecting anti-fade mounting medium formulations and implementing standardized operating procedures, researchers can significantly extend the fluorescence signal observation window, achieve high-definition microscopic imaging, and establish a long-term storable precious sample library, providing reliable support for in-depth mechanistic analysis and repeated experimental verification.
Absin Anti-Fade Mounting Medium Recommendation
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
