- Cart 0
- English
Why can type I collagen become the golden matrix in cell culture and tissue engineering?
April 24, 2026
Clicks:72
Collagen is the major component of the extracellular matrix (ECM) in connective tissues and internal organs, and is most widely distributed in the skin, tendons, and bones. As a core component of the ECM, collagen not only provides structural support but also interacts with cells through receptors such as integrins to regulate cell adhesion, proliferation, differentiation, and migration. In cell culture, Collagen Type I is widely used for coating culture vessel surfaces to enhance cell attachment or preparing three-dimensional (3D) gels to simulate the in vivo microenvironment due to its excellent biocompatibility and specific cell adhesion sites. Depending on the source and preparation process, Collagen Type I from rat tail and bovine Achilles tendon has distinct advantages in physical properties and application scenarios, providing diverse choices for researchers.
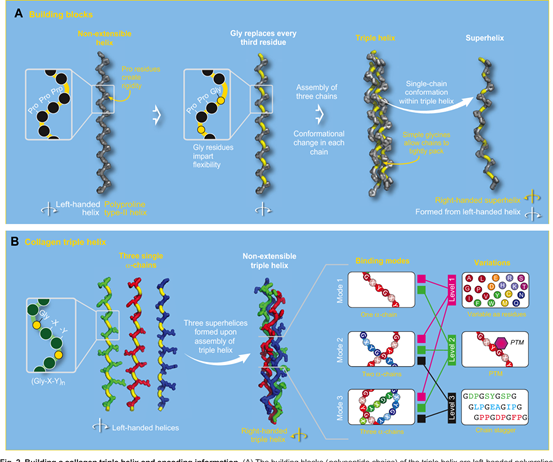
Triple Helix Structure of Collagen
What are the structural characteristics of Collagen Type I?
Structurally, Collagen Type I is a heterotrimer composed of two α1 chains and one α2 chain, which spontaneously forms a triple helix structure at 37°C and neutral pH. This unique triple helix conformation confers high mechanical strength and biological stability to collagen.
Molecular Properties:
- Self-assembly Capacity: Under appropriate conditions, triple helix molecules further assemble into collagen fibrils to form a network scaffold
- Cell Adhesion Sites: Contains cell adhesion sequences such as RGD (Arginine-Glycine-Aspartic acid), which can be recognized by integrin receptors on the cell membrane
- Biodegradability: Can be specifically degraded by collagenases, exhibiting excellent bioabsorbability in tissue engineering
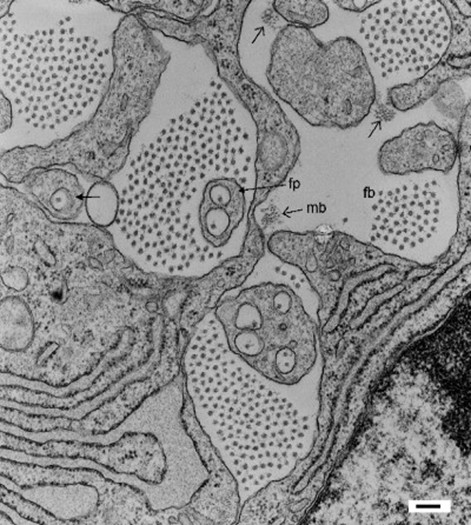
Electron Microscopic Structure of Collagen Fibrils
What is the difference between Rat Tail Collagen and Bovine Tendon Collagen?
Based on different sources and extraction processes, Collagen Type I commonly used in laboratories is mainly divided into two categories:
Rat Tail Collagen
- Source: Prepared from rat tail tendons via acetic acid extraction, sodium chloride precipitation, and disodium hydrogen phosphate precipitation
- Form: Sterile solution dissolved in 6 mM acetic acid (HAc), typically at a concentration of 5 mg/mL
- Features: Retains intact triple helix structure and gelling ability, can self-assemble into gels at neutral pH and 37°C
- Applications: Used for both culture surface coating and 3D collagen gel preparation
Bovine Tendon Collagen
- Source: Extracted from bovine Achilles tendon, usually atelocollagen (purified collagen with telopeptide regions removed by enzymatic cleavage)
- Form: White or pale yellow powder, BR grade, soluble in water
- Features: Thoroughly enzymolyzed, low molecular weight, no gelling ability, cannot form 3D gels
- Applications: Mainly used for 2D culture systems such as cell surface coating, cell adhesion assays, and drug screening
Summary of Key Differences:
How to establish a standardized surface coating protocol?
Collagen surface coating significantly improves cell adhesion and spreading morphology for cells that are difficult to attach to common plastic culture vessels (e.g., primary hepatocytes, fibroblasts, epithelial cells).
Recommended Coating Parameters:
- Concentration Range: 1–5 μg/cm², initial optimization at 5 μg/cm²
- Diluent: 6 mM acetic acid (for rat tail collagen stock solution)
Protocol (using rat tail collagen as an example):
- Dilution: Dilute 5 mg/mL stock to working concentration with 6 mM HAc (e.g., 50 μg/mL for 5 μg/cm² coating)
- Addition: Add calculated volume to each well of dishes/plates to fully cover the surface
- 96-well plate (0.3 cm²/well): ~30 μL (5 μg/cm²)
- 6-well plate (9.5 cm²/well): ~950 μL
- Incubation: Incubate at room temperature for 1 hour, or air-dry uncovered overnight in a biosafety cabinet
- Washing: Aspirate excess liquid, wash 3–4 times with sterile PBS
- Usage: Use directly, or store at 4–25°C (coated vessels stable for 3 months)
Key Note: Bovine tendon collagen powder must first be dissolved in sterile water, then diluted and coated following the same protocol. Due to the lack of gelling ability, it is more suitable for experiments requiring only enhanced adhesion without 3D structure.
How to prepare and use 3D collagen gels?
3D collagen culture mimics the physical and biochemical properties of the in vivo ECM, ideal for studying cell-matrix interactions, tissue morphogenesis, and drug screening.
Gelling Mechanism:
Rat tail Collagen Type I forms strong 3D gels at concentrations >1 mg/mL and pH ~7.0. The gelation involves a sol-to-gel phase transition, typically completed within 20 minutes at 37°C.
Preparation (1 mL of 1 mg/mL 3D gel):
- Ice Bath Pre-cooling: Chill all reagents and tubes on ice to prevent premature gelation
- Mixing:
- Take 200 μL collagen stock (5 mg/mL)
- Add 12 μL 0.1 M NaOH to neutralize acetic acid, add NaOH to collagen, NOT reverse
- Mix immediately
- Dilution: Add 100 μL 10×PBS or 10×medium, mix
- Volume Adjustment: Add 688 μL sterile water, mix (final pH ~7.0)
- Loading: Add to culture vessels immediately
- Solidification: Incubate at 25°C for 20 minutes until gelation, then transfer to 37°C incubator
Preparation of cell-laden 3D gels:
After step 3 above, add 760 μL cell suspension (instead of water), mix well and add to dishes immediately. Cells will be uniformly embedded in the gel to simulate the in vivo 3D growth environment.
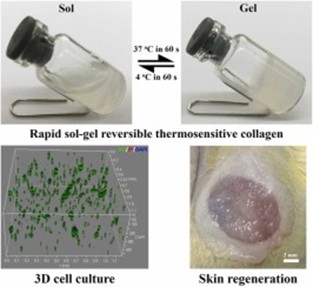
Cell Culture in 3D Collagen Gel
Which cells are most suitable for collagen culture systems?
As a culture substrate, Collagen Type I is particularly suitable for the following cell types:
Strong Adhesion-Dependent Cells:
- Primary Hepatocytes: Maintain polarity and metabolic function for drug metabolism studies
- Fibroblasts: Promote spreading and proliferation for ECM synthesis research
- Epithelial Cells: Enhance tight junction formation and maintain barrier function
- Endothelial Cells: Promote angiogenesis for vascularization assays
Neuronal Cells:
- Dorsal Root Ganglion (DRG) Neurons: Promote neurite outgrowth
- Schwann Cells: Maintain myelination capacity
- Spinal Cord Neurons: Support long-term survival and differentiation
Other Specialized Cells:
- Muscle Cells: Promote myotube formation
- Embryonic Lung Cells: Support primary culture establishment
Cell Adhesion on Collagen Surface
Key Precautions for Usage
Operating Environment Control:
- Low-Temperature Operation: Perform 3D collagen preparation entirely on ice; room temperature accelerates gelation and causes inhomogeneity
- Sterile Operation: Complete all steps in a biosafety cabinet to avoid contamination affecting cell growth
pH Regulation:
- Neutralization Order: Always add NaOH to collagen solution, NOT reverse; high local pH causes collagen denaturation and aggregation
- pH Testing: For first-time preparation, check final mixture pH with test strips to ensure ~7.0 (pink with phenol red indicator)
Gelation Judgment:
- Tilt the dish; the gel should not flow
- Gently shake; the gel should be elastic with no liquid movement on the surface
Storage and Stability:
- Stock Solution: Store at 4°C, avoid repeated freeze-thaw cycles
- Coated Vessels: Stable for 3 months at 4–25°C; check sterility before use
- 3D Gels: Use immediately after preparation; long-term storage causes dehydration or protein degradation
Selection Advice for Different Collagen Sources:
- 3D Culture Required: Must use rat tail collagen (gelling capacity)
- Enhanced Adhesion Only: Choose bovine tendon collagen (easy operation, no neutralization)
- Large-Scale Coating: Bovine tendon powder is easier for bulk working solution preparation
By selecting the appropriate collagen type and optimizing coating or gelation conditions, researchers can provide an in vivo-like growth environment for precious and difficult-to-culture primary cells, or establish standardized 3D tissue models, offering a reliable technical platform for cell biology, pharmacology, and regenerative medicine research.
Absin Recommended Collagen Products
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
