- Cart 0
- English
Why is a broad-spectrum protease inhibitor cocktail an essential safeguard for protein sample protection?
April 23, 2026
Clicks:63
Within cells, the synthesis and degradation of proteins maintain a precise dynamic equilibrium. However, once cells are lysed for in vitro studies, this balance is immediately disrupted: protein synthesis ceases, while abundant endogenous proteases released by cell lysis remain active and continue to degrade target proteins. These proteases are derived from primary cells, mammalian cells, animal and plant tissues, bacteria, yeast, etc., and encompass diverse catalytic types, including serine proteases, cysteine proteases, aspartic proteases, and aminopeptidases. Without the addition of effective protease inhibitors to the lysis buffer, precious target proteins may be rapidly degraded during extraction, leading to blurred Western Blot bands, loss of immunoprecipitation products, distorted enzyme activity assays, and ultimately compromising the accuracy and reproducibility of experimental data.
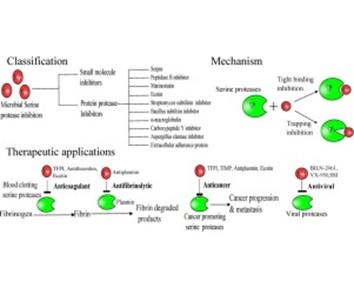
Schematic diagram of protease inhibition mechanism
What are the key components of a broad-spectrum protease inhibitor cocktail?
Compared with single inhibitors, broad-spectrum protease inhibitor cocktails achieve comprehensive inhibition of various proteases through synergistic mechanisms. A typical formulation contains the following six potent inhibitors:
| Inhibitor Name | Target | Inhibition Mechanism | Features |
|---|---|---|---|
| AEBSF (4-(2-Aminoethyl)benzenesulfonyl fluoride) | Serine proteases | Irreversible inhibition | Safe alternative to PMSF, 14-fold less toxic |
| Aprotinin | Serine proteases | Reversible inhibition | Inhibits trypsin, kallikrein, etc. |
| Bestatin | Aminopeptidases | Reversible inhibition | Blocks N-terminal amino acid exolysis |
| Leupeptin | Serine and cysteine proteases | Reversible inhibition | Dual inhibitory capacity |
| Pepstatin | Aspartic proteases | Reversible inhibition | Specifically inhibits pepsin-like enzymes |
| E-64 | Cysteine proteases | Irreversible inhibition | Specifically inhibits thiol proteases |
This combination ensures comprehensive coverage of common proteases in samples ranging from animal tissues to microbial lysates, including serine proteases, cysteine proteases, metalloproteases (aminopeptidases), and acid proteases.
What are the advantages over the single inhibitor PMSF?
Phenylmethylsulfonyl fluoride (PMSF) is traditionally used in laboratories as a serine protease inhibitor, but broad-spectrum cocktails are significantly superior to PMSF in multiple dimensions:
Greatly enhanced safety:
The LD₅₀ of PMSF is approximately 200 mg/kg (oral in mice), whereas AEBSF, its primary replacement component, has an LD₅₀ as high as 2834 mg/kg, representing a ~14-fold reduction in toxicity. This is critical for protecting the health of researchers exposed to reagents long-term.
Faster and more durable inhibition:
PMSF has an extremely short half-life in aqueous solution (only ~30 minutes) and is prone to rapid hydrolysis and inactivation during cell lysis. In contrast, the multiple inhibitors in the cocktail exhibit superior chemical stability and provide longer-lasting inhibition against different protease classes.
Broader inhibition spectrum:
PMSF targets only serine proteases and fails to inhibit cysteine proteases, aspartic proteases, etc. Broad-spectrum cocktails exert multi-target synergistic effects to ensure effective inhibition of all protease types, making them particularly suitable for tissue samples rich in diverse proteases such as liver and spleen.
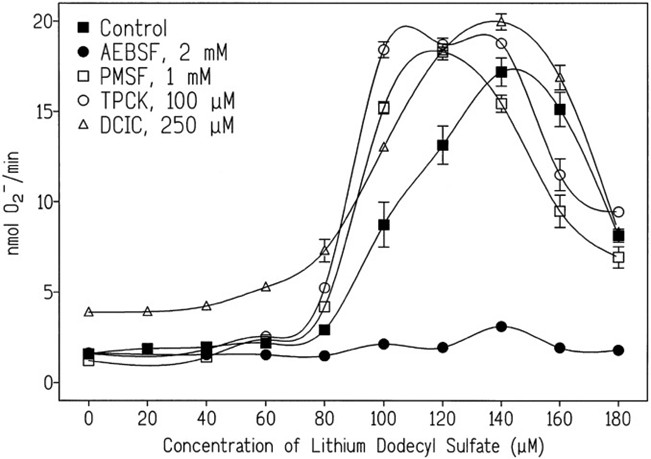
Comparison of PMSF and AEBSF efficacy
In which experimental scenarios is it indispensable?
Western Blot:
Ensures clear and intact target protein bands, avoiding smearing or diffusion. Especially critical for low-abundance transcription factors and signaling pathway proteins.
Immunoprecipitation (IP/Co-IP) and Pull-down assays:
Prevents degradation of bait or prey proteins during incubation in protein-protein interaction studies, guaranteeing reliable detection of interactions.
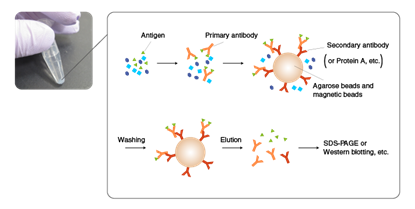
Principle of immunoprecipitation
Preparation of cell and tissue lysates:
Used for protein purification, kinase activity assays, subcellular fractionation, etc. A 1:100 dilution ratio (e.g., 1 mL cocktail treats 20 g wet weight cells) provides sufficient protective concentration.
Immunofluorescence (IF) and Immunohistochemistry (IHC):
Protects protein antigens from degradation by endogenous proteases during tissue section preparation and antigen retrieval.
Compatibility with Immobilized Metal Affinity Chromatography (IMAC):
Notably, high-quality broad-spectrum cocktails are EDTA-free (ethylenediaminetetraacetic acid, a metalloprotease inhibitor). This ensures perfect compatibility with downstream applications such as IMAC (e.g., Ni-NTA purification of His-tagged proteins) and 2D gel electrophoresis, eliminating concerns about EDTA stripping metal ions from chromatographic columns.
Application of protease inhibitors in Western Blot
How to use correctly for optimal protection?
Storage and dissolution:
Products are typically supplied as 100× concentrated DMSO stock solutions. Equilibrate to room temperature (recommended 25°C) before use to ensure complete dissolution, as DMSO melts slowly at low temperatures. Avoid repeated freeze-thaw cycles; aliquot for storage.
Timing and ratio of addition:
- Standard dilution: Add to cell or tissue lysis buffer at a 1:100 volume ratio (e.g., 10 μL cocktail into 1 mL lysis buffer)
- Add in advance: Add immediately when preparing lysis buffer; do not add after cell lysis is complete
- Prepare fresh: Use lysis buffer containing inhibitors immediately or aliquot and freeze-store; avoid prolonged storage at 4°C
Optimization for different samples:
- Protease-rich samples (e.g., liver, pancreas): Appropriately increase concentration to 1:50
- Mild lysis (e.g., non-denaturing conditions): Increase inhibitor concentration to compensate for residual proteases from incomplete lysis
- Bacterial lysates: May require additional supplementation with specific inhibitors targeting bacterial proteases
Key precautions during use
Special requirements for phosphorylated protein research:
Broad-spectrum protease inhibitor cocktails lack phosphatase inhibitory activity. For studies on phosphorylated proteins (e.g., signaling pathway analysis), a phosphatase inhibitor cocktail (e.g., NaF, Na₃VO₄, etc.) must be added separately, otherwise phosphorylation modifications will be rapidly lost during lysis.
Limitations in inhibiting deubiquitinases (DUBs):
Conventional protease inhibitors (including AEBSF, Leupeptin, etc.) do not inhibit certain deubiquitinases (e.g., ATAXIN-3). To maintain protein ubiquitination status for research, specific DUB inhibitors (e.g., PR-619, P22077, etc.) must be added separately.
Assessment of DMSO effects:
The final concentration of DMSO in the stock solution is approximately 1% (when diluted 1:100), which generally does not affect most enzyme activities or protein interactions. However, for certain DMSO-sensitive kinases or enzymatic assays, preliminary validation experiments are recommended.
Real-time monitoring of protein degradation:
A negative control without inhibitors is recommended to verify inhibitor efficacy by observing intact target protein bands via Western Blot. If multiple degraded bands or blurred bands appear, check for inhibitor inactivation or insufficient concentration.
By systematically using broad-spectrum protease inhibitor cocktails, researchers can maximize the integrity of protein samples, ensuring target proteins remain stable throughout the entire process from cell lysis to downstream analysis, thereby obtaining reliable and reproducible experimental data.
Absin Broad-Spectrum Protease Inhibitor Cocktail Recommendation
| Cat. No. | Product Name | Size |
|---|---|---|
| abs9161 | Broad-Spectrum Protease Inhibitor Cocktail (EDTA-free, 100× DMSO solution) | 1mL/1mL×5 |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
