In immunohistochemistry and immunofluorescence experiments, when detecting low-abundance target proteins or performing multiplex labeling, conventional fluorescent secondary antibody labeling methods often face limitations including insufficient signal intensity, high background noise, or antibody cross-reactivity. Tyramide Signal Amplification (TSA) technology achieves signal enhancement of several-fold to several ten-fold through catalytic deposition reactions at antigen-antibody binding sites. The signal amplification solution (TSA fluorophore reaction solution), as the core component of this technology, contains concentrated fluorescent dyes and a specialized buffer system. Under the catalysis of horseradish peroxidase (HRP), the non-activated fluorophore substrate is converted into highly reactive free radicals that covalently bind to protein tyrosine residues, forming high-density fluorescent deposits. This significantly improves detection sensitivity and enables multiplex protein co-localization analysis at the single-cell level.
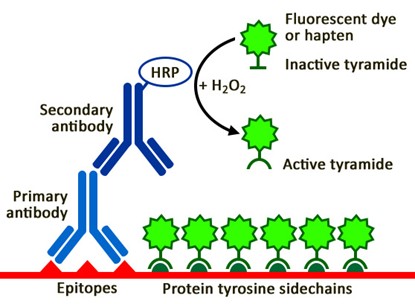
TSA Signal Amplification Principle Schematic
What Is the Chemical Principle of Tyramide Signal Amplification?
TSA technology is based on the HRP-catalyzed tyramide free radical reaction mechanism:
1
Activation Process
The fluorescent dyes in the signal amplification solution use tyramide molecules as the scaffold, linked to fluorophores to form non-activated substrates. In the presence of H₂O₂, HRP (typically localized to the target site through polymeric HRP-conjugated secondary antibodies or streptavidin-HRP conjugates) catalyzes the oxidation of tyramide substrates, generating highly reactive tyramide free radicals.
2
Deposition Process
Activated tyramide free radicals possess extremely high reactivity and rapidly form covalent bonds with adjacent protein tyrosine residues, achieving site-specific deposition of fluorescent dyes. Since each HRP molecule can catalyze the deposition of multiple tyramide molecules, and the deposited fluorescent molecules themselves exhibit HRP-like activity (in certain systems), a cascade amplification effect is formed. Ultimately, hundreds to thousands of fluorescent molecules are deposited at each antigenic site, achieving exponential amplification of signal intensity.

TSA Workflow Schematic
How to Optimize the Dilution Ratio of Signal Amplification Solution?
The dilution ratio of the signal amplification solution (volume ratio of fluorescent dye to buffer) directly affects the signal-to-noise ratio and experimental success, requiring precise optimization based on primary antibody incubation conditions:
Dilution Ratio Range
The standard dilution ratio is 1:100 (fluorescent dye : TSA buffer), but the optimal range is typically between 1:50 and 1:400, adjusted according to the following factors:
Room Temperature Incubation (1-3 hours)
Relatively lower primary antibody binding; higher dye concentrations are recommended (1:50 to 1:200 dilution) to ensure sufficient signal intensity
4°C Overnight Incubation (12-16 hours)
Primary antibody binding is extensive and highly specific; lower dye concentrations are recommended (1:200 to 1:400 or higher dilution) to avoid excessive labeling and elevated background
Pre-Experimental Validation Strategy
Due to significant differences in antigen abundance across tissue sections, pre-experimental validation is recommended before formal experiments:
- Prepare the reaction solution at the recommended ratio (e.g., 1:100), apply to the sample, and react at room temperature for 1 minute
- Wash with PBS and observe staining results under a microscope
- If positive signal is weak, continue adding fluorescent dye to enhance staining intensity until appropriate; if background is too high, increase the dilution ratio or shorten the reaction time
How Is It Applied in Multiplex Immunofluorescence Experiments?
TSA signal amplification technology is the core enabling technology for multiplex immunofluorescence (mIF), achieving simultaneous detection of up to 5-8 targets on the same tissue section through sequential staining strategies:
Sequential Staining Workflow
1
First Round Staining: Primary antibody A → Polymeric HRP secondary antibody → TSA fluorophore (e.g., Opal 520, green)
2
Antibody Elution: Remove bound antibodies through microwave treatment or antibody stripping solution (fluorescent deposits are preserved)
3
Second Round Staining: Primary antibody B → Polymeric HRP secondary antibody → TSA fluorophore (e.g., Opal 570, yellow)
4
Iterative Cycling: Complete staining for all targets sequentially, followed by DAPI nuclear counterstaining
Technical Advantages
- Species Independence: Each target uses TSA dyes with different spectral properties, avoiding species cross-reactivity limitations of conventional methods
- High Signal Intensity: Even low-abundance targets yield clear signals
- Low Background: Covalent binding ensures precise localization of fluorescent signals to antigenic sites
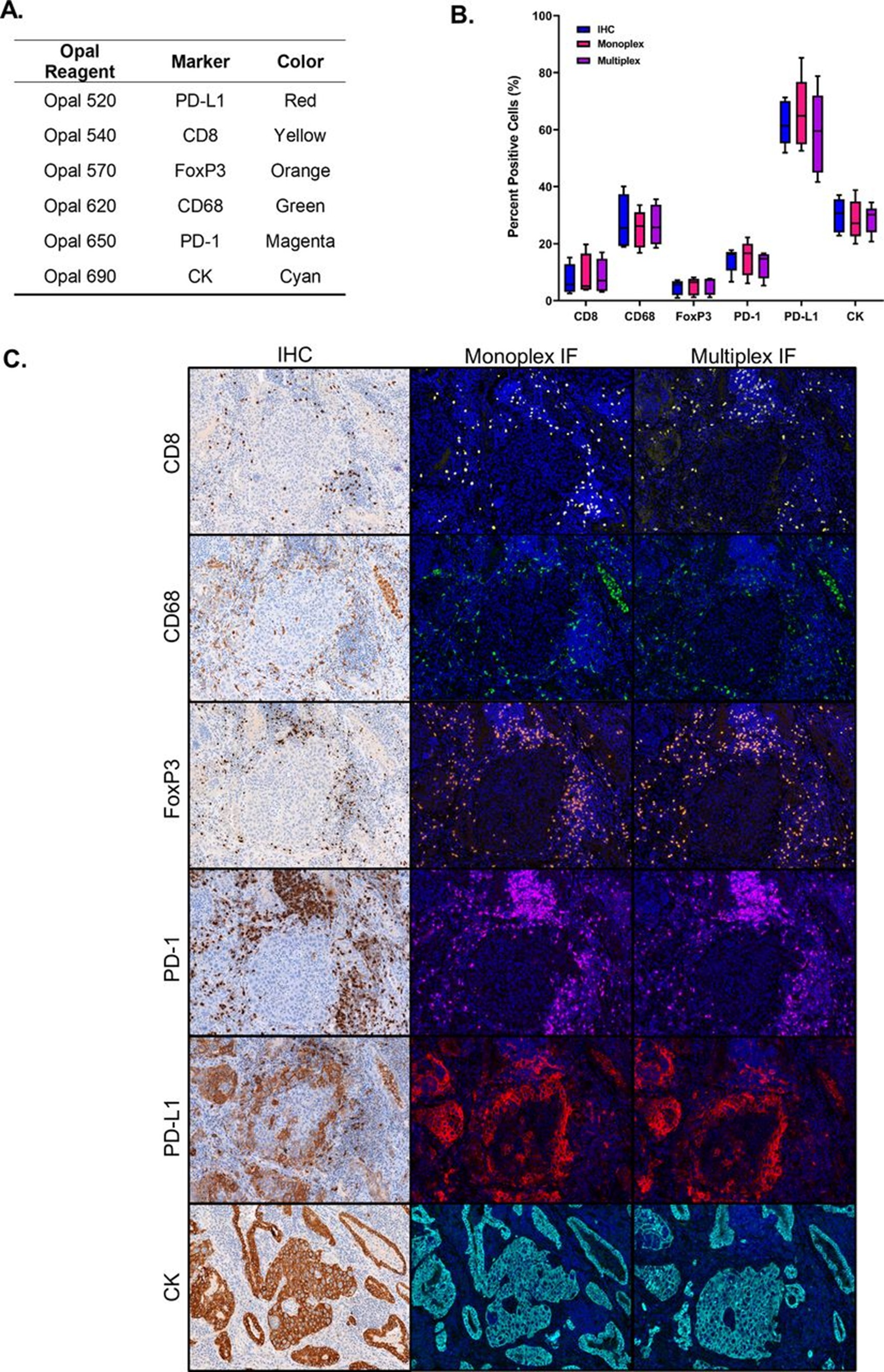
Multiplex Immunofluorescence Staining Effect Comparison
How to Establish a Standardized Operating Procedure?
Preliminary Preparation
Section or tissue microarray samples complete routine dewaxing, hydration, antigen retrieval, and primary antibody incubation steps.
Signal Amplification Stage
- Secondary Antibody Incubation: Complete incubation with polymeric HRP secondary antibody (Note: HRP-conjugated secondary antibodies or polymers must be used), wash with PBS three times for 5 minutes each to thoroughly remove unbound secondary antibody
- Reaction Solution Preparation: Mix concentrated fluorescent dye with TSA buffer at the optimized ratio (e.g., 1:100), prepare fresh, and protect from light during operation
- Room Temperature Reaction: Apply the prepared TSA reaction solution evenly to cover the tissue, react at room temperature protected from light for 1-15 minutes (optimal time 5-10 minutes)
Time Control: Insufficient reaction time leads to weak signals; excessive time increases background; optimization according to target abundance is required
- Thorough Washing: Wash with PBS three times to thoroughly remove free fluorescent dyes that have not been deposited (this step is critical for reducing background)
Subsequent Steps
- If proceeding to the next round of multiplex staining, antibody elution must be performed first (microwave treatment or specialized elution solution), preserving the fluorescent signal, then repeat the above workflow
- Finally, mount with anti-fade mounting medium containing DAPI, and observe using confocal or fluorescence microscopy

Multiplex Immunofluorescence TSA Technology Workflow
Critical Considerations for Use
Light Protection
TSA fluorescent dyes are photosensitive; the entire process from preparation to staining must be protected from light (using aluminum foil wrapping or darkroom operation) to prevent photobleaching.
HRP Activity Control
- Before TSA reaction, unbound HRP-conjugated secondary antibodies must be thoroughly washed away; otherwise, non-specific background will result
- Ensure thorough washing after primary and secondary antibody incubation; this is the key to reducing background
Reaction Time Optimization
- First-time Use: It is recommended to set up a time gradient (1 min, 5 min, 10 min, 15 min) to find the optimal balance point
- High-Abundance Targets: Shorten reaction time (1-5 min) to avoid signal oversaturation
- Low-Abundance Targets: Extend reaction time (10-15 min); increase dye concentration if necessary
Compatibility with Other Methods
- TSA technology can be combined with conventional immunofluorescence, but spectral overlap must be considered
- For antibody systems without HRP labeling, HRP enzyme must first be introduced via biotin-streptavidin-HRP or polymer-HRP secondary antibodies
Safety
Wear a lab coat and disposable gloves during operation, avoiding direct skin contact with fluorescent dyes. Dispose of waste liquids according to laboratory chemical waste disposal regulations.
Through precise optimization of the dilution ratio and reaction conditions of the signal amplification solution, researchers can achieve up to 7-9 color fluorescent labeling on a single tissue section, enabling in-depth analysis of immune cell infiltration in the tumor microenvironment, intercellular interactions, and signaling pathway activation states, thereby providing high-dimensional spatial biology information for precision medicine research.
Absin Signal Amplification Solution Recommendations
| Cat. No. |
Product Name |
Specification |
| abs90018 |
Signal Amplification Solution |
10mL/50mL |
* For specific product information, please consult the supplier for the latest documentation
【Disclaimer】This article is compiled from publicly available online information and generated by AI. If it inadvertently infringes upon any rights, please contact us promptly, and we will cooperate in handling the matter at the earliest opportunity, assuming no legal liability.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.





 Follow us on Facebook: Absin Bio
Follow us on Facebook: Absin Bio