As a classical anticoagulant, heparin sodium has long transcended its purely "anticoagulant" function in life science research. This highly sulfated glycosaminoglycan, derived from porcine intestinal mucosa, has become an important tool in cardiovascular research, cell culture, exosome studies, and drug development due to its unique molecular structure and diverse biological activities. This article will delve into the multidimensional research value of this classical molecule, drawing upon existing technical literature.
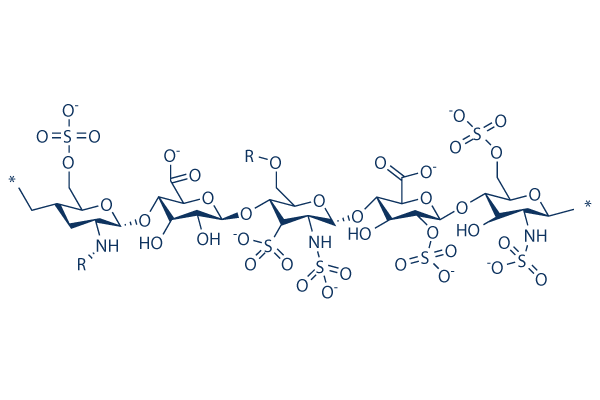
Heparin Sodium Molecular Structure Schematic
What Is Heparin Sodium? — Source and Chemical Nature
Heparin sodium is the sodium salt form of heparin, belonging to a complex mixture of linear polysaccharides. Its molecular chains are composed of sugar units of variable lengths and sequences, exhibiting significant heterogeneity. This highly sulfated glycosaminoglycan carries dense negative charges, displaying polyelectrolyte behavior in solution analogous to that of DNA.
This substance is primarily extracted from porcine intestinal mucosa and subjected to multi-step purification processes, ultimately presenting as a white or off-white powder. Its chemical structure contains multiple sulfonate and carboxyl groups, and the spatial arrangement of these functional groups determines the specificity of its interactions with proteins.
What Is Unique About Its Anticoagulant Mechanism?
The core function of heparin sodium lies in its reversible binding to antithrombin III (AT III), an interaction that significantly accelerates the rate at which AT III inhibits serine proteases in the coagulation cascade. Specifically, upon binding of heparin to AT III, a conformational change is induced in AT III, exposing its reactive site loop and thereby greatly enhancing its inactivation capacity toward thrombin (factor IIa) and factor Xa.
Studies have shown that formation of this ternary complex (heparin-AT III-thrombin) can increase the bimolecular rate constant for thrombin inhibition by approximately 2,000-fold. Notably, the binding between heparin and AT III is reversible, meaning its anticoagulant activity can be regulated according to physiological requirements.
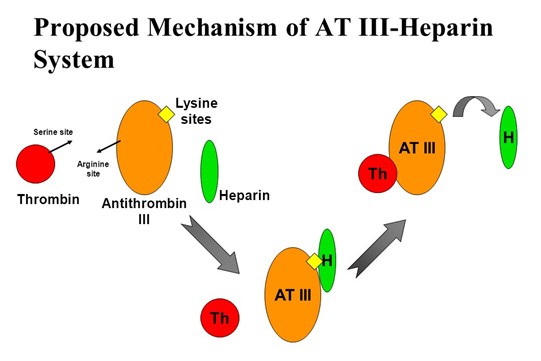
AT III-Heparin System Mechanism of Action
How Should Purity Standards Be Interpreted?
In terms of quality control, heparin sodium is evaluated by two distinct purity assessment systems:
● Biological Potency Standard
Expressed in USP units/mg, typically requiring a potency of no less than 175 USP units/mg. This evaluation method is based on in vitro anticoagulant activity assays, reflecting the actual biological function of the molecule.
● Chemical Purity Standard
Expressed as mass percentage, such as 98% purity, reflecting the proportion of the target molecule in the sample.
These two standards each have their own emphases: biological potency is more applicable to experimental scenarios focused on anticoagulant function, whereas chemical purity holds greater reference value in structural biology and physicochemical research. Researchers should select the appropriate quality standard based on specific experimental requirements.
What Other Experiments Can It Be Used for Beyond Anticoagulation?
In recent years, the application scope of heparin sodium has expanded significantly, encompassing multiple cutting-edge research fields:
Exosome Research
Recent studies have found that heparin sodium can significantly inhibit the interaction between exosomes and cells. This characteristic makes it an important tool in exosome isolation and purification processes, where it can be used to block nonspecific binding of exosomes to target cells, thereby improving isolation purity.
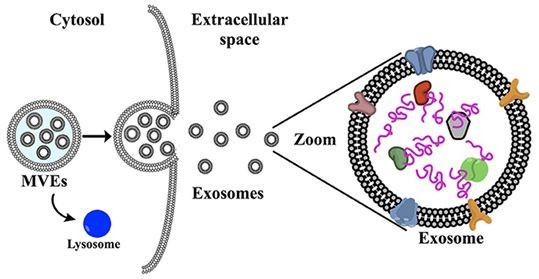
Exosome Research Applications
Cell Signaling Transduction
Heparin can interact with fibroblast growth factor (FGF-2) and its receptor (FGFR-1), stabilizing the FGF-FGFR binding and promoting FGFR dimerization. This makes it play an important role in studies of FGF signaling pathways, cell proliferation, and differentiation experiments.
Cell Culture and Apoptosis Research
As a common additive in cell culture media, heparin sodium can be used to maintain the growth state of certain sensitive cells. Meanwhile, it also has specific applications in cell apoptosis, cytoskeleton, and extracellular matrix (ECM) research.
Affinity Chromatography Purification
Heparin-coupled affinity resins are widely used for the isolation and purification of DNA-binding proteins, coagulation factors, growth factors, etc., utilizing its specific binding capacity with these proteins.
Heparin Affinity Chromatography Applications
Drug Discovery
As a tool for lead compound discovery and small molecule drug screening, heparin sodium continues to demonstrate value in the development of anticoagulant drugs and antitumor drug delivery systems (such as heparin-drug conjugates).
How to Ensure Stability and Correct Operation?
Proper storage and handling are crucial for maintaining heparin sodium activity:
● Solubility
Heparin sodium is readily soluble in water (solubility up to 50-100 mg/mL) but insoluble in organic solvents such as methanol, ethanol, or acetone. Sterile water or physiological saline is recommended when preparing stock solutions.
● Storage Conditions
The powder form exhibits extremely high stability, with literature reports indicating storage stability up to 7 years. Prepared solutions should be stored at 2-8°C protected from light and remain stable for 2 years. Special attention should be paid to the fact that freezing is not recommended, as freeze-thaw cycles may cause molecular aggregation or activity loss.
● Sterilization Method
Solutions should not be subjected to 121°C autoclaving (5-10 minutes), as high temperatures can cause cross-linking reactions in the polysaccharide chains, resulting in activity loss. Filtration sterilization using a 0.22 μm membrane filter is recommended. At the same time, strict prevention of microbial contamination is essential, as certain microorganisms can secrete heparinase, leading to heparin degradation.
● Laboratory Safety
Wear a lab coat and disposable gloves during operation, avoiding dust inhalation and direct skin contact.
Conclusion
From a classical anticoagulant to a tool molecule in modern exosome research, heparin sodium has demonstrated its versatility as a research reagent. Understanding its structure-activity relationship, mastering correct operational protocols, and selecting appropriate purity standards according to research objectives are key to ensuring experimental success. As our understanding of the biological functions of sulfated glycosaminoglycans continues to deepen, the application prospects of this classical molecule in life science research will become even broader.

 Follow us on Facebook: Absin Bio
Follow us on Facebook: Absin Bio