- Cart 0
- English
Why does the selection of cell cryopreservation solution need to vary from cell to cell?
April 21, 2026
Clicks:70
What Are the Essential Differences Between Serum-Containing and Serum-Free Cryopreservation Media?
Based on the presence or absence of Fetal Bovine Serum (FBS), cell cryopreservation media are classified into two major categories, with significant differences in protective mechanisms, application scopes, and safety profiles.
Traditional Serum-Containing Cryopreservation Medium:
Formulated with Fetal Bovine Serum (FBS) and Dimethyl Sulfoxide (DMSO) as main components, typically containing 10% DMSO and 90% serum (or medium + serum). Albumin, growth factors, and nutrients in serum stabilize cell membranes during freezing and reduce ice crystal damage. This medium is suitable for cryopreservation of various mammalian primary cells, passage cells, and hybridoma cells, serving as a classic universal formulation.
Serum-Free Cryopreservation Medium:
Adopts chemically defined formulations with absolutely no animal-derived proteins or serum, usually combined with 10% or 5% DMSO, specific osmotic pressure regulators, buffer systems, and nutritional factors. Its advantages include:
- Defined Composition: Eliminates experimental repeatability issues caused by serum batch variations
- High Safety: Eliminates contamination risks of animal-derived pathogens (e.g., mad cow disease, mycoplasma)
- Excellent Downstream Compatibility: Suitable for serum-free cultured cells and cells for subsequent protein expression and antibody production
For sensitive cells such as stem cells (ES cells, MSCs) and immune cells (T cells, NK cells), serum-free formulations significantly reduce the risk of cell differentiation and phenotypic alterations.
How Does DMSO Concentration Affect Cryopreservation Strategies?
DMSO (Dimethyl Sulfoxide) is the most commonly used cryoprotectant, functioning by penetrating cells to lower the freezing point and reduce ice crystal formation. However, DMSO exhibits certain cytotoxicity, and tolerance varies drastically among cell types. Cryopreservation media are categorized by DMSO concentration as follows:
High-DMSO Type (10%):
The classic golden standard concentration, suitable for most conventional cell lines (e.g., HeLa, 293T, CHO). High-concentration DMSO provides stronger cryoprotection, but some cells (e.g., primary neurons, certain stem cells) may be sensitive to it, requiring DMSO clearance after thawing to restore viability.
Low-DMSO Type (5%):
Reduces DMSO toxicity while maintaining cryopreservation efficacy by optimizing other protective components (e.g., trehalose, propylene glycol). Suitable for DMSO-sensitive cell types and scenarios requiring immediate experimental use after rapid thawing.
DMSO-Free Type:
Utilizes innovative amphiphilic polymer amino acid derivatives as cryoprotectants, completely DMSO-free. Specially designed for extremely sensitive cells such as primary hepatocytes, nerve cells, and certain stem cell lines. Its advantage is no concerns about residual DMSO affecting cell functions post-thaw, enabling faster cell recovery and growth.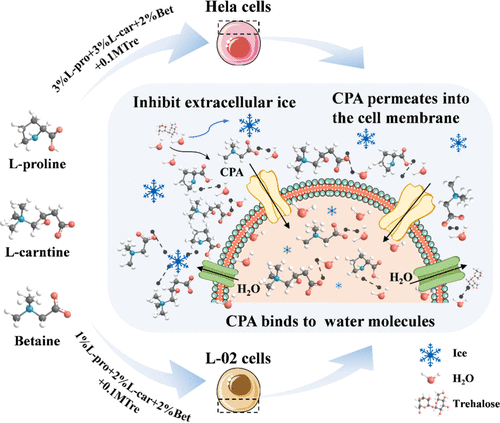
Protective Mechanism of DMSO-Free Cryopreservation Medium
What Exclusive Formulations Are Required for Special Cell Types?
With the development of cell therapy, regenerative medicine, and immunotherapy, dedicated cryopreservation media for distinct cell types have emerged:
Dedicated for Human Pluripotent Stem Cells (hES/hiPS):
Embryonic stem cells and induced pluripotent stem cells are highly sensitive to cryopreservation conditions, prone to differentiation or apoptosis during freeze-thaw cycles. Dedicated stem cell cryopreservation media maintain cell pluripotency at -80°C or in liquid nitrogen by adding Rho kinase inhibitors (e.g., Y-27632) and specific growth factors, achieving a post-thaw cloning efficiency of over 90%.
Dedicated for Immune Cells:
Surface marker expression and functions of immune cells (T cells, DC cells, NK cells, CIK cells) are easily affected by cryopreservation. Dedicated formulations are usually phenol red-free (to avoid oxidative stress) and serum-free, with optional autologous serum or albumin for enhanced protection. Some formulations support "serum substitutes" to further improve the viability and functions of immune cells post-thaw.
Dedicated for Primary Cells:
Primary cells, freshly isolated from tissues, have limited proliferation capacity and high environmental sensitivity. Dedicated cryopreservation media typically adopt low-DMSO or DMSO-free formulations combined with mild cooling procedures to maximally preserve the original phenotypes and functional activities of primary cells.
How to Choose Between Programmed Cooling and One-Step Freezing?
Cryopreservation medium formulation is closely correlated with cooling methods, and modern cryopreservation media have achieved the breakthrough of "no programmed cooling required":
Traditional Programmed Cooling Method:
Suitable for traditional serum-containing or high-DMSO cryopreservation media. Requires a programmed cooling box (containing isopropanol) or a programmable freezer, with gradual cooling following the gradient of 4°C → -20°C → -80°C → Liquid Nitrogen at a rate of 1-2°C per minute. This method minimizes cell damage by controlling ice crystal formation but is time-consuming (usually overnight or hours).
One-Step Direct Freezing Method:
Modern serum-free cryopreservation media support direct placement into a -80°C freezer for freezing by optimizing osmotic pressure and adding specific ice crystal formation inhibitors, eliminating programmed cooling steps. This greatly saves experimental time and reduces operational complexity, especially ideal for high-throughput cell bank construction. Note that transfer from -80°C to liquid nitrogen for long-term storage is recommended as soon as possible (within 24 hours), or direct short-term storage at -80°C (several months).
What Are the Key Steps for Cell Thawing?
Precise operation is equally critical for the thawing of cryopreserved cells, and a correct thawing process maximizes cell survival rate:
Rapid Thawing Principle:
Immediately place cryovials removed from liquid nitrogen or -80°C into a 37°C water bath for rapid thawing with gentle and constant shaking. Remove promptly when only a rice-sized ice pellet remains in the cryopreservation medium to avoid prolonged water exposure leading to DMSO cytotoxicity.
Dilution and Washing:
Immediately add 9-15 volumes of pre-warmed complete medium to the thawed cell suspension for dilution, mix gently, and centrifuge (typically 1000-1200 rpm, 5-10 minutes). This step dilutes and removes most DMSO, reducing its persistent cytotoxicity to cells.
Culture and Observation:
Discard the supernatant after centrifugation, resuspend cells in fresh medium, and inoculate into culture vessels. Observe cell status after 24 hours; most cells should have adhered or resumed suspension growth. Replace with fresh medium subsequently to completely remove residual cryopreservation medium components.
Cell Thawing Process
What Preparations Are Required Before Cryopreservation?
Regardless of the cryopreservation medium used, cell status before cryopreservation is the key determinant of thawing success:
Cell Status Examination:
Ensure cells are contamination-free (fungi, bacteria, mycoplasma) and in the logarithmic growth phase (confluency ~80-90%), when cell viability is highest, leading to optimal post-thaw recovery rates.
Cell Density Optimization:
The recommended cryopreservation density is 1×10⁶ - 1×10⁷ cells/mL, varying by cell type. Over-low density may cause slow post-thaw growth, while over-high density may lead to nutrient competition and cell aggregation.
Pre-Cooling and Aliquoting:
Pre-cool cryopreservation medium at 2-8°C before use, aliquot immediately into cryovials after mixing with cells to minimize room-temperature exposure. Aliquoted cell suspensions should not stand at room temperature or 2-8°C for more than 10 minutes before transfer to -80°C or liquid nitrogen to prevent premature ice crystal formation.
Conclusion
The selection of cell cryopreservation medium is a systematic decision requiring comprehensive consideration of cell type, experimental purpose, and downstream applications. Traditional serum-containing formulations remain an economical choice for universal experiments; serum-free formulations provide safer support for cell therapy and protein production; low-DMSO and DMSO-free formulations offer milder protection for sensitive cells. Regardless of the formulation chosen, mastering the basic principle of "slow freezing and rapid thawing", ensuring optimal cell status, and strict aseptic operation are the ultimate approaches to achieve high post-thaw survival rates (over 90%). With continuous advances in cryopreservation technology, more cell types will achieve efficient and safe long-term preservation in the future, providing reliable cell resource support for life science research.
Recommended Absin Cell Cryopreservation Media:
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
