- Cart 0
- English
How do Mycoplasma detection kits build a defense line in the quality control of cell culture?
April 20, 2026
Clicks:82
In cell culture laboratories, mycoplasma contamination is known as the "silent killer"—it does not cause culture medium turbidity or rapid cell death, yet it can silently alter cellular metabolism, proliferation kinetics, and gene expression profiles, leading to distorted or completely erroneous experimental results. It is estimated that approximately 15%-35% of cell cultures worldwide are contaminated with mycoplasma. Facing this invisible threat, mycoplasma detection kits based on different detection principles (PCR method, quantitative real-time PCR method, and isothermal amplification visualization method) provide researchers with multi-level solutions ranging from routine screening to high-sensitivity quantitative detection. Which scenarios are these kits suitable for? How to select the most appropriate detection protocol based on experimental requirements?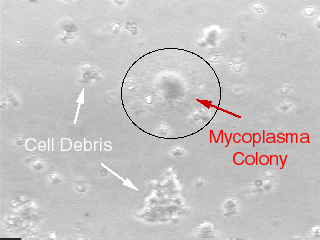 Mycoplasma-contaminated cells
Mycoplasma-contaminated cells
Why Has Mycoplasma Contamination Become the "Invisible Killer" in Cell Culture?
Mycoplasma, a class of wall-less prokaryotic microorganisms measuring only 0.2-0.8 μm, can pass through conventional 0.22 μm sterilization filters. Their unique biological characteristics make them the most difficult contaminants to control in cell culture:
High Concealment: Mycoplasma contamination does not cause immediate cell death or medium turbidity. Contaminated cells appear normal under microscopy but exhibit subtle alterations in growth rate, chromosomal abnormalities, and metabolite level changes.
Profound Impact: Mycoplasma can alter host cell DNA, RNA, and protein synthesis, interfere with cell signal transduction, and affect immune response and drug sensitivity. In biopharmaceutical applications, mycoplasma contamination may result in entire batch discard.
Rapid Transmission: Through aerosols, operators, reagents, or equipment cross-contamination, mycoplasma can spread rapidly between laboratories.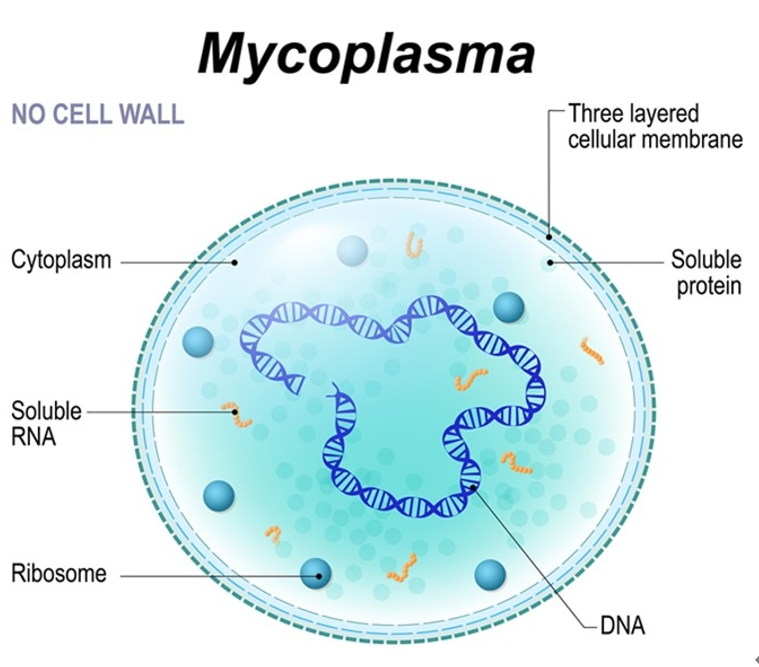 Mycoplasma structure
Mycoplasma structure
What Are the Differences Between Detection Kits Based on Different Principles?
According to detection principles and technology platforms, mycoplasma detection kits are mainly classified into three categories, with significant differences in sensitivity, detection time, and equipment requirements:
| Detection Method | Detection Target | Sensitivity | Detection Time | Main Equipment | Applicable Scenarios |
|---|---|---|---|---|---|
| Conventional PCR | 16S rRNA conserved region | ~20 copies | 1-2 hours | Thermal cycler, electrophoresis equipment | Routine screening, laboratory self-inspection |
| Quantitative Real-Time PCR | 16S rRNA (TaqMan probe) | ~10 CFU/mL | 1-1.5 hours | Real-time PCR thermal cycler | High-sensitivity detection, quantitative analysis |
| Isothermal Amplification Visualization | Species-specific DNA sequence | High sensitivity | 30 minutes | Water bath/heating block (65°C) | Rapid screening, point-of-care testing |
How Does Conventional PCR Identify Mycoplasma Contamination?
PCR detection based on the 16S rRNA gene is the most classical method for mycoplasma detection. The 16S rRNA gene exhibits high conservation among mycoplasma species while containing species-specific variable regions, making it an ideal molecular detection target.
Detection Principle:
The specific primers provided in the kit are designed to target conserved regions of the mycoplasma 16S rRNA sequence. When mycoplasma DNA is present in the sample, the primers amplify a ~500 bp specific DNA fragment through PCR. The reaction system typically contains 2× PCR Mix (including dNTPs, Taq polymerase, etc.) and specific primer mix.
Operational Procedure:
1. Sample Preparation: Collect cell culture supernatant, heat at 95°C for 5 minutes to release DNA (no complex DNA extraction required)
2. PCR Amplification: 98°C denaturation for 2 minutes, followed by 30 cycles (98°C for 20 seconds → 56°C for 25 seconds → 72°C for 10 seconds), final extension at 72°C for 5 minutes
3. Result Interpretation: 1% agarose gel electrophoresis analysis, positive samples show ~500 bp band
Method Advantages:
• Capable of detecting at least 11 common mycoplasma species (e.g., M. orale, M. arginini, M. bovis, M. pneumoniae, etc.)
• Sensitivity reaches 20 copies
• Simple operation, cost-effective
• Species identification possible through sequencing
Limitations:
Requires opening tubes for electrophoresis analysis, posing aerosol contamination risk; cannot quantify.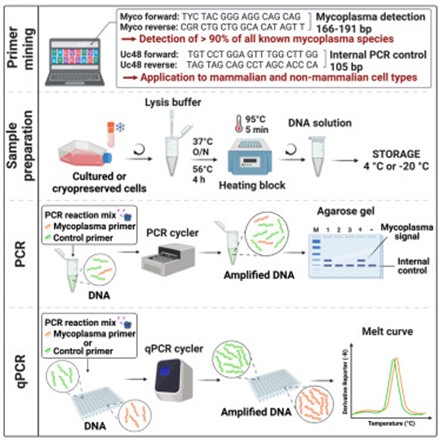 PCR detection workflow
PCR detection workflow
How Does Quantitative Real-Time PCR Achieve High-Sensitivity Quantitative Detection?
When higher sensitivity or quantitative analysis is required, TaqMan probe-based quantitative real-time PCR (qPCR) kits are the optimal choice. This method complies with European Pharmacopoeia <2.6.7> standards and is suitable for biopharmaceutical release testing.
Technical Principle:
Adopting multiplex PCR strategy using two differently fluorescent-labeled probes:
• FAM fluorophore: Labels mycoplasma-specific probe (Reporter)
• CY5 fluorophore: Labels internal control gene probe (Internal Control)
The TaqMan probe is an oligonucleotide probe with a fluorophore at the 5' end and a quencher at the 3' end. During PCR amplification, the 5'→3' exonuclease activity of Taq polymerase degrades the probe, separating the fluorophore from the quencher and generating fluorescent signal. Fluorescence signal accumulation is completely synchronized with PCR product formation, enabling quantitative analysis through Ct values (Cycle threshold).
Detection Limit and Specificity:
• Detection limit reaches 10 CFU/mL
• 100% detection rate for 10 mycoplasma standard strains mentioned in pharmacopoeia
• No cross-reactivity with genomic DNA from common biopharmaceutical sources (e.g., Human, Vero, CHO, HEK293, E. coli, etc.)
Anti-Contamination Design:
The kit typically contains dUTP/UNG (uracil-DNA glycosylase) system, which can degrade potential PCR product contamination to prevent false positives.
Result Interpretation:
• Positive: FAM channel Ct value <40 with obvious amplification curve
• Negative: Ct value ≥40 or no obvious amplification
• PCR Inhibition: Abnormal internal control CY5 signal (requires re-extraction of sample)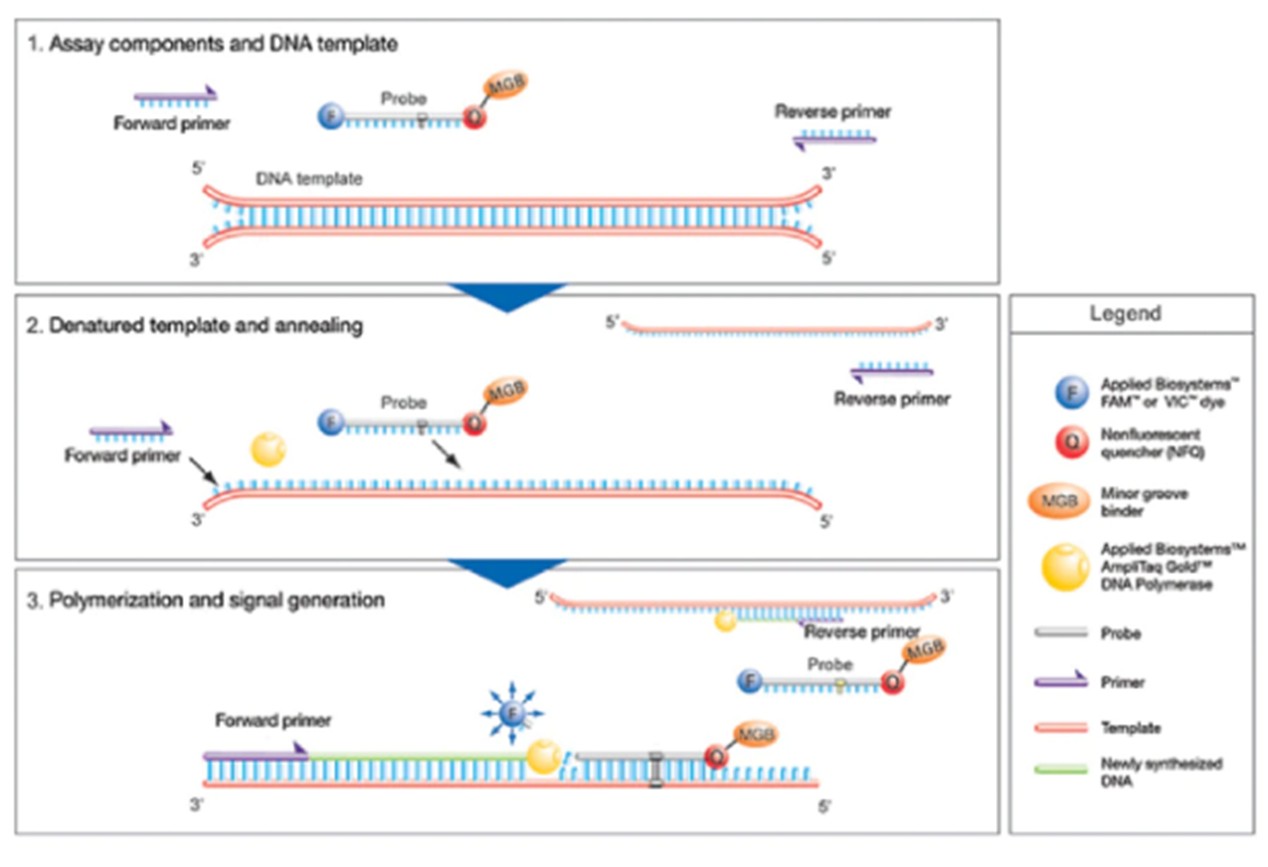 TaqMan probe principle
TaqMan probe principle
How Does Isothermal Amplification Visualization Enable Rapid Screening?
For scenarios requiring rapid results or lacking PCR equipment, visualization detection kits based on isothermal amplification technology provide a convenient "sample-in, result-out" solution.
Technical Principle:
Utilizing specially designed primers and strand-displacing DNA polymerase (such as Bst DNA polymerase) to perform nucleic acid amplification at constant temperature (65°C). This method does not require thermal cycling of PCR instruments and only needs a water bath or heating block.
Visualization Mechanism:
The reaction system contains pH indicator. The byproduct (hydrogen ions) produced during amplification changes the reaction solution pH, causing color change:
• Negative: Reaction solution remains yellow (original color)
• Positive: Reaction solution turns red or pink
Some kits use paraffin oil to seal the liquid surface, preventing aerosol contamination when opening tubes.
Operational Procedure:
1. Add Mycored Buffer and enzyme mix to reaction tube
2. Add 20 μL paraffin oil to seal
3. Insert pipette tip below liquid surface to add 1 μL cell culture supernatant
4. Incubate at 65°C for 30 minutes
5. Observe color change (do not open tube to observe, avoid contamination)
Method Advantages:
• Rapid: Results obtained in 30 minutes
• Simple: No PCR instrument or electrophoresis equipment needed, suitable for point-of-care testing
• Closed-tube detection: Avoids aerosol contamination
• High tolerance: Tolerates various PCR inhibitors in culture medium
Applicable Scenarios:
Daily rapid screening, teaching laboratories, point-of-care testing, high-throughput preliminary screening.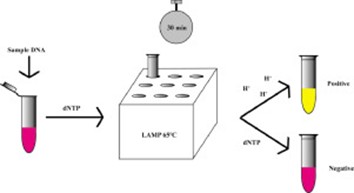 Isothermal amplification color change
Isothermal amplification color change
How to Ensure the Reliability of Detection Results?
Regardless of the detection method used, a rigorous quality control system is the prerequisite for ensuring accurate results:
Control Setup:
• Negative Control (NC): Use mycoplasma-free water or kit-provided negative control to monitor environmental contamination
• Positive Control (PC): Use kit-provided positive control DNA to verify PCR system validity
• Internal Control (IC): Internal control gene in qPCR method monitors whether samples contain PCR inhibitors
Sample Collection Key Points:
• Adherent cells: Directly aspirate culture supernatant; recommended sampling when cell confluence reaches ~90% and culture exceeds 3 days, when mycoplasma content is higher
• Suspension cells: Centrifuge at 1000 rpm for 5 minutes, then collect supernatant
• Serum or reagents: Direct sampling to exclude raw material contamination
Anti-Contamination Measures:
• Zoned operation: Separate sample preparation area, reagent preparation area, and amplification area
• Use filter-barrier pipette tips
• Do not open tubes to observe PCR products (especially for isothermal amplification method)
• Regularly use DNA decontamination reagents to treat workbenches
Detection Frequency:
• Mandatory testing for newly established cell lines or after cryopreserved cell revival
• Routine culture cells recommended for testing every 1-2 months
• Biopharmaceutical release testing must use pharmacopoeia-recognized methods (such as qPCR)
Conclusion
Mycoplasma detection is the first line of defense in cell culture quality control. Conventional PCR offers simplicity and cost advantages for routine laboratory screening; quantitative real-time PCR, with its high sensitivity and quantitative capabilities, has become the gold standard for biopharmaceutical release testing; isothermal amplification visualization, with its rapid, equipment-free characteristics, is suitable for point-of-care rapid screening. Researchers should select appropriate methods based on detection purpose (qualitative vs. quantitative), sensitivity requirements, and equipment availability. Remember, regular monitoring is more important than post-hoc remediation—establishing a strict mycoplasma surveillance system is the fundamental guarantee for ensuring the reliability of cell experimental data.
Absin Mycoplasma Detection Kit Recommendations:
| Catalog No. | Product Name | Specification |
|---|---|---|
| abs9588 | Mycoplasma Detection Kit (PCR Method) | 50 Tests |
| abs9925 | Mycoplasma Detection Kit (qPCR Method) | 50 Tests |
| abs90062 | One-Step Rapid Mycoplasma Detection Kit (Isothermal Amplification Method) | 50 Tests |
| abs90283 | MYCO-L™ Luciferase Mycoplasma Detection Kit | 10T/25T/50T |
| abs90284 | MYCO-T™ Nested PCR Mycoplasma Detection Kit | 25T/50T |
| abs90199 | Bioluminescent Mycoplasma Detection Kit | 20T/200T |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
