- Cart 0
- English
Breaking Through the Bottleneck of Cervical Cancer Immunotherapy: A Novel Strategy Targeting Glutamine Metabolism Transporter SLC25A22 to Enhance CD8+ T-Cell Function
April 16, 2026
Clicks:75
Recently, a significant study was published in Advanced Science, revealing the intrinsic connection between glutamine metabolism dysregulation and the immunosuppressive microenvironment in cervical squamous cell carcinoma (CSCC). The study demonstrated that targeting the glutamine transporter SLC25A22 significantly enhances CD8+ T-cell function and improves anti-PD-1 therapy efficacy.
Article Title:Targeting Glutamine Metabolism Transporter SLC25A22 Enhances CD8+ T-Cell Function and Anti-PD-1 Therapy Efficacy in Cervical Squamous Cell Carcinoma: Integrated Metabolomics, Transcriptomics and T-Cell-Incorporated Tumor Organoid Studies
Journal:Advanced Science (IF 14.1)
DOI:https://doi.org/10.1002/advs.202502225
Absin Reagent Used:Ionomycin


I. Clinical Challenge: The Critical Problem of Low Response Rates to Immunotherapy in Cervical Cancer
Cervical squamous cell carcinoma accounts for 70% of cervical cancer cases, with limited efficacy of traditional treatment modalities. Although immune checkpoint inhibitors have brought new hope for treatment, the objective response rate of anti-PD-1 therapy in CSCC is only 16.4%, representing a clinical dilemma that urgently requires resolution.
II. Multi-Omics Integration Analysis Reveals Novel Metabolism-Immunity Mechanisms
1. Metabolomics Identifies Glutamine Metabolism Abnormalities
The research team conducted metabolomic analysis of 44 CSCC and 18 normal tissue samples, identifying 2,083 metabolites. Glutamine and related metabolites were significantly enriched in CSCC tissues.
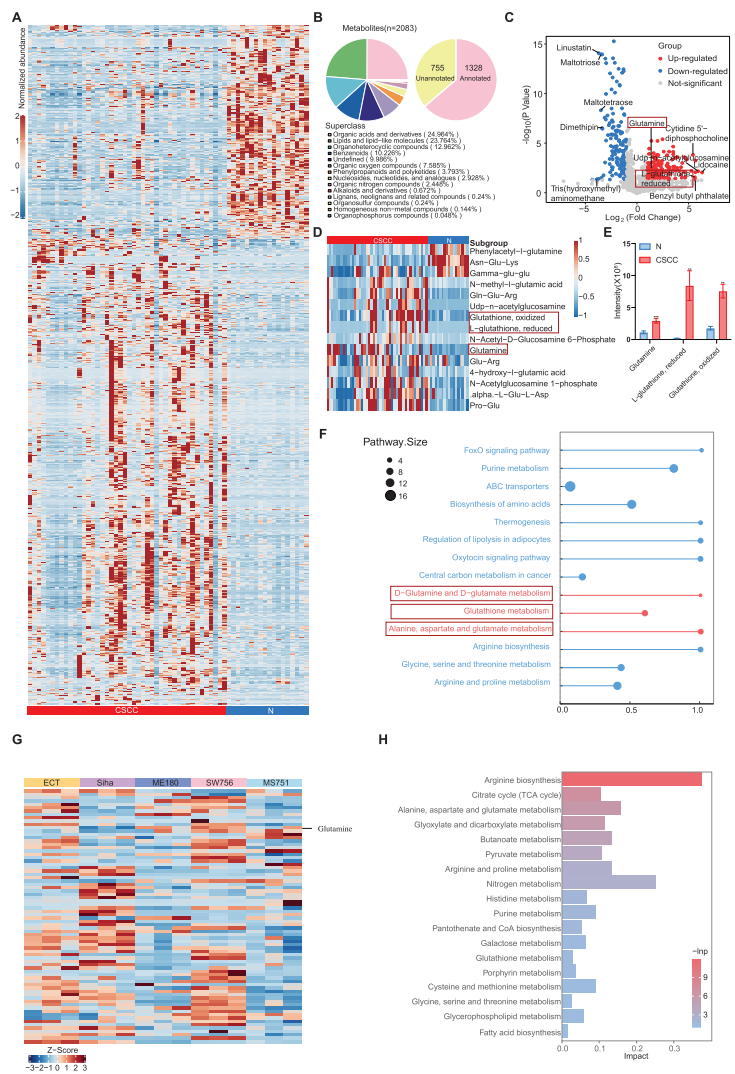
2. Transcriptomics Confirms Immunosuppressive Microenvironment
Transcriptomic analysis revealed distinct immunosuppressive characteristics in CSCC, including activation of T-cell function negative regulatory pathways and significant enrichment of PD-1/PD-L1 signaling pathways.
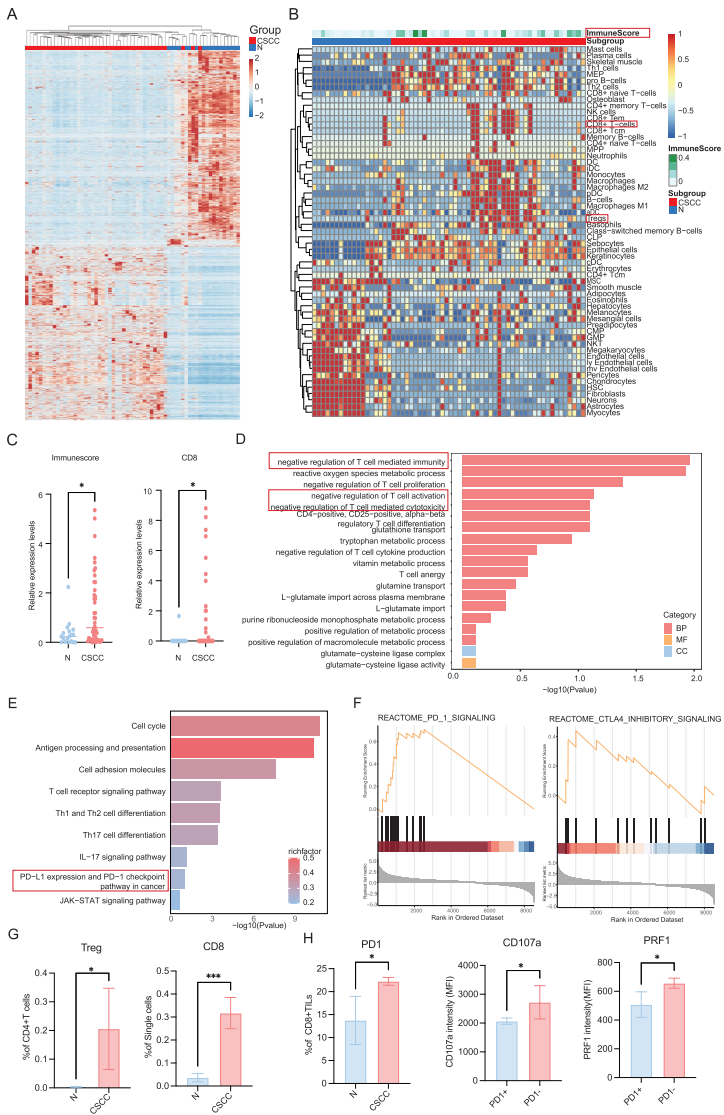
III. Key Discovery: SLC25A22 as the Bridge Connecting Metabolism and Immunity
1. Core Role of SLC25A22
Integrated analysis revealed that the glutamine transporter SLC25A22 showed significant positive correlation with multiple immune checkpoint molecules (PD-L1, CTLA-4, etc.), establishing it as the key molecule connecting metabolism and immunity.
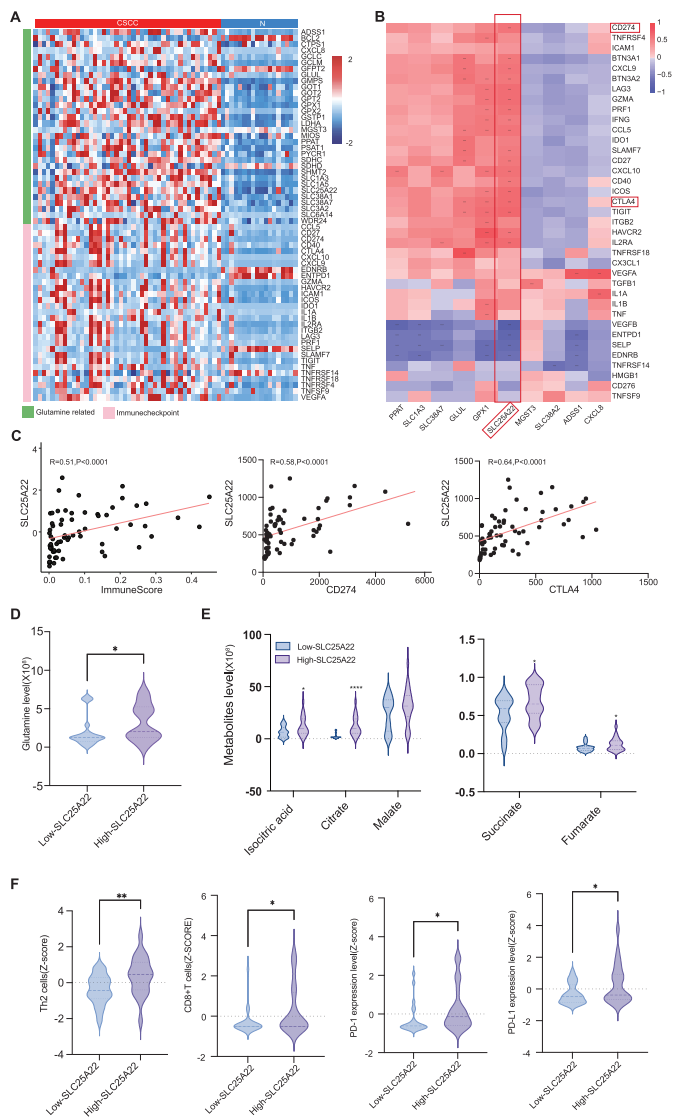
2. Clinical Validation Confirms Its Clinical Significance
Validation in 20 CSCC clinical samples demonstrated that high SLC25A22 expression was significantly correlated with elevated PD-1 expression and reduced CD8+ T-cell cytotoxicity, particularly pronounced in PD-L1-high-expression patients.
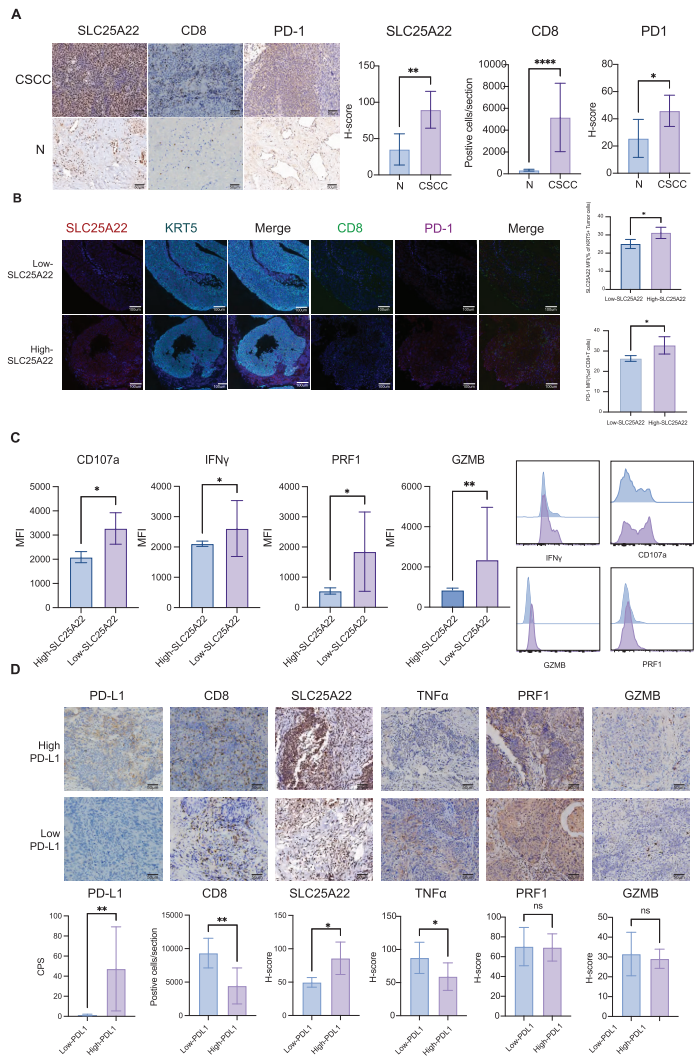
IV. Innovative Model: Establishment of 3D T-Cell-Incorporated Organoid Platform
1. Construction of CSCC Organoid Models
CSCC organoids were successfully established, preserving the histological and genomic characteristics of parental tumor tissues, providing an ideal platform for in vitro studies.
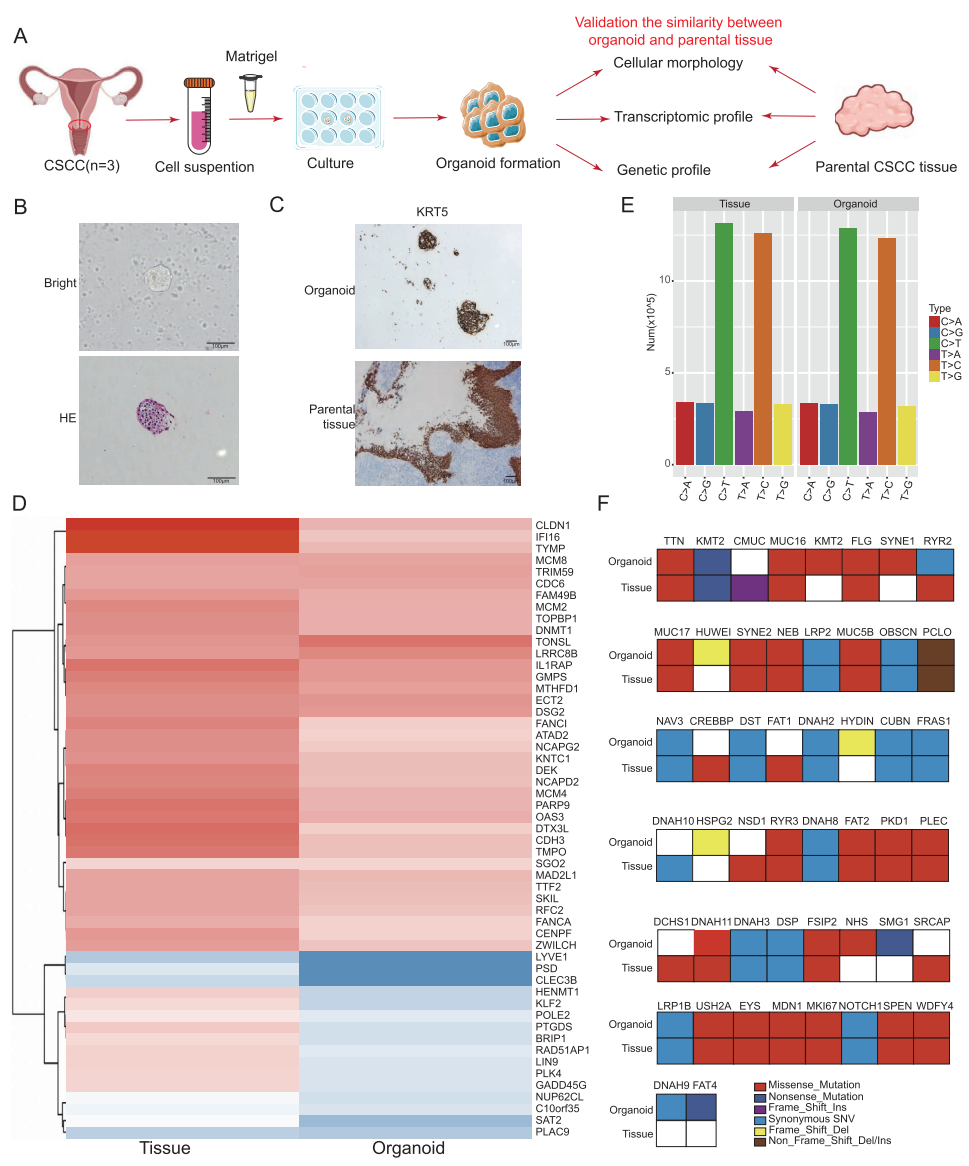
2. 3D T-Cell-Incorporated Organoid Model
CD8+ T-cells were innovatively co-cultured with tumor organoids to authentically simulate cell-cell interactions within the tumor immune microenvironment.
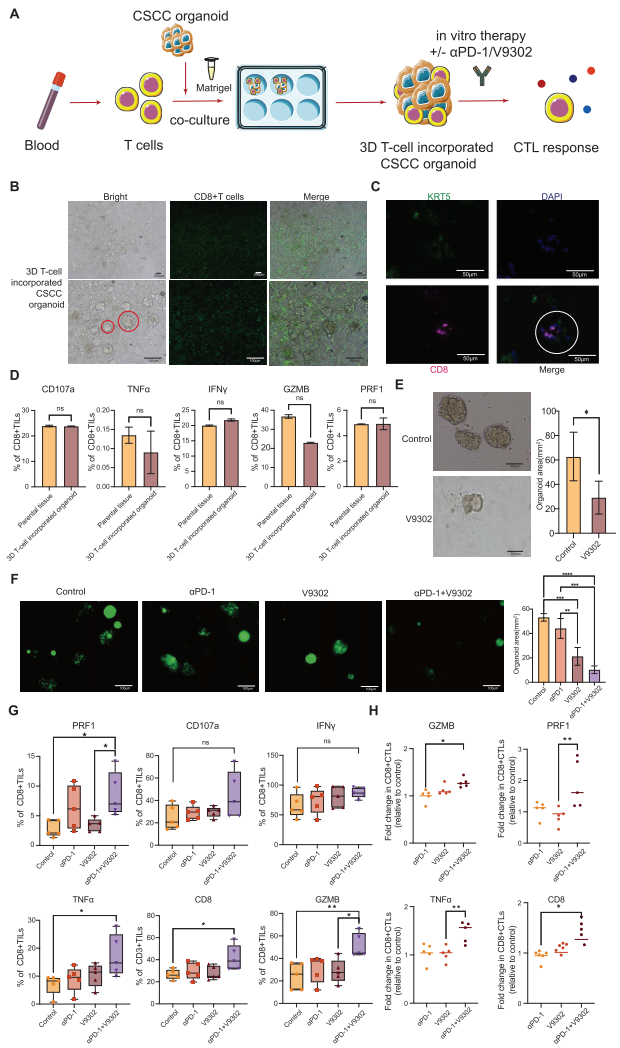
V. Therapeutic Breakthrough: Targeting SLC25A22 Enhances Immunotherapy Efficacy
1. Validation of V-9302 Efficacy
Using the glutamine transporter inhibitor V-9302 to target SLC25A22 significantly enhanced the expression of CD8+ T-cell cytotoxic molecules (such as PRF1, GZMB) in the 3D organoid model.
2. Synergistic Anti-PD-1 Therapy
The combination of V-9302 and anti-PD-1 antibody demonstrated significant synergistic effects, substantially improving therapeutic outcomes.
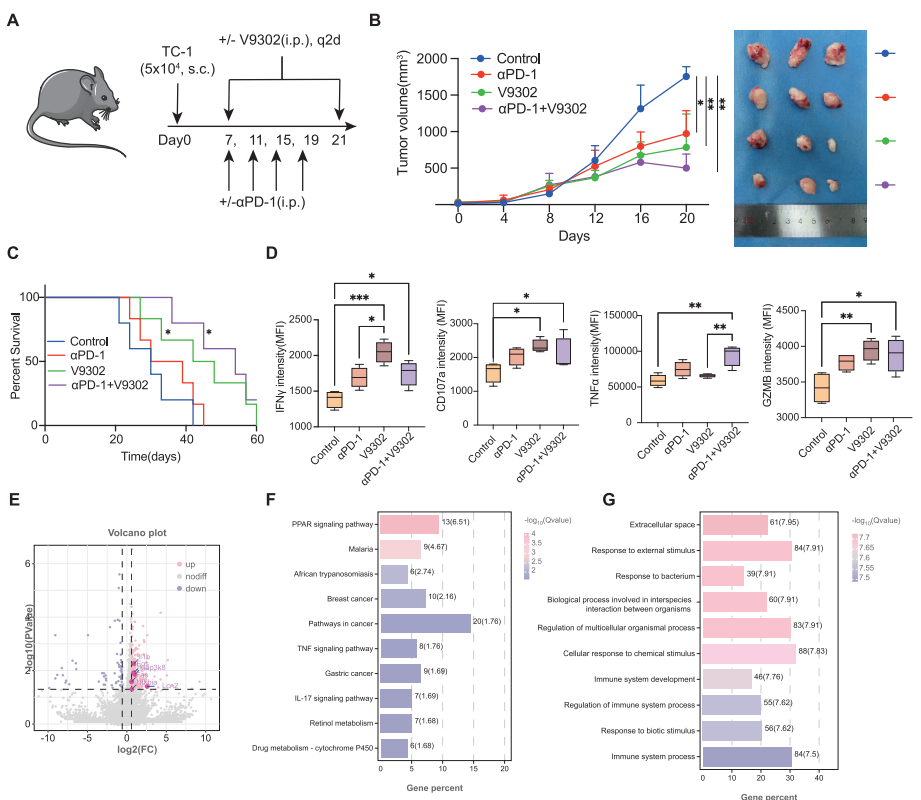
VI. Mechanistic Elucidation: Metabolic Reprogramming Improves the Immune Microenvironment
1. Signaling Pathway Regulation
Combination therapy significantly enriched TNF signaling pathway and IL-17 signaling pathway, activating immune system processes.
2. Immune Cell Functional Remodeling
Targeting SLC25A22 downregulates immune checkpoint (PD-1, CTLA-4) expression and promotes CD8+ T-cell effector molecule (IFN-γ, TNF-α, PRF1) secretion.
VII. Research Significance and Clinical Translation Value
- Biomarker Development:SLC25A22 can serve as a predictive biomarker for immunotherapy efficacy in PD-L1-high-expression CSCC patients.
- Novel Combination Therapy Strategy:Provides new treatment options for immunotherapy-resistant patients, particularly in the PD-L1-high-expression population.
- New Direction in Metabolic Immunotherapy:Opens a new avenue for enhancing immunotherapy efficacy through targeting tumor metabolism.
Conclusion
This study systematically elucidated for the first time the central role of SLC25A22 in immune-metabolic regulation of cervical cancer, providing a novel target for overcoming immunotherapy resistance. Absin's high-quality research reagents provided reliable technical support for the realization of this important discovery.
[Disclaimer]This article is based on the original publication in Advanced Science (DOI: 10.1002/advs.202502225), interpreted and organized by AI; all original figures, data, and intellectual property rights belong to the original journal and research team. If any infringement occurs, please contact us promptly for removal, and we will actively cooperate in handling such matters.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
