- Cart 0
- English
Unraveling the Mechanism of Lactate-Driven Invasive Klebsiella pneumoniae Liver Abscess Syndrome — Absin ELISA Kits Empower Critical Inflammatory Cytokine Detection
April 16, 2026
Clicks:76
Globally, the incidence of invasive Klebsiella pneumoniae liver abscess syndrome (IKPLAS) continues to rise, with its high mortality rate and severe complications remaining challenging clinical problems. A landmark study published in Nature Communications has, for the first time, revealed the core mechanism by which lactate regulates capsular polysaccharide (CPS) biosynthesis and enhances bacterial virulence in Klebsiella pneumoniae through the PTS-CRP axis, providing novel therapeutic targets for IKPLAS diagnosis and treatment. As an experimental tool provider, Absin's ELISA kits played a crucial role in inflammatory cytokine detection, assisting the research team in precisely deciphering host immune response mechanisms.
Article Title:Lactate promotes invasive Klebsiella pneumoniae liver abscess syndrome by increasing capsular polysaccharide biosynthesis via the PTS-CRP axis
Journal:Nature Communications (IF 15.7)
DOI:https://doi.org/10.1038/s41467-025-61379-9Absin Products Used:Mouse IL-6 ELISA Kit (abs520004)、Mouse TNF-α ELISA Kit (abs520010)、Mouse IL-1β ELISA Kit (abs520001)


I. Research Strategy: Focusing on Host-Pathogen Interactions to Decode IKPLAS Pathogenesis
Previous studies have primarily focused on the microbiological characteristics of Klebsiella pneumoniae, but could not fully explain the pathogenesis of IKPLAS. This study innovatively integrates host factors with bacterial virulence regulation, adopting a three-step strategy of "clinical sample analysis → in vitro mechanistic validation → in vivo animal experiments":
- Clinical Correlation Analysis: Comparison of clinical data and bacterial isolates from 68 KPLA (simple Klebsiella pneumoniae liver abscess) and IKPLAS patients to screen for independent risk factors;
- In Vitro Functional Validation: Investigation of the regulatory effects and molecular mechanisms of the key host factor (lactate) on Klebsiella pneumoniae virulence factors (CPS, siderophores, etc.);
- In Vivo Animal Modeling: Validation of the pathophysiological significance of lactate-enhanced bacterial virulence through mouse liver abscess models, clarifying the central role of CPS.
II. Core Research Findings: Lactate as the "Key Driver" of IKPLAS Progression
1. Clinical Breakthrough: Elevated Blood Lactate as an Independent Predictor of IKPLAS
The study found that IKPLAS patients had significantly higher blood lactate levels than simple KPLA patients, and also higher than those with Klebsiella pneumoniae-induced respiratory tract infections or urinary tract infections (Supplementary Figure 1). Multivariate regression analysis confirmed that elevated blood lactate (OR=4.547, P=0.008) was an independent risk factor for IKPLAS occurrence, while there were no significant differences in molecular characteristics (sequence types, serotypes, virulence genes, etc.) of Klebsiella pneumoniae between the two groups (Figure 1), suggesting that host factors play a dominant role in disease progression.
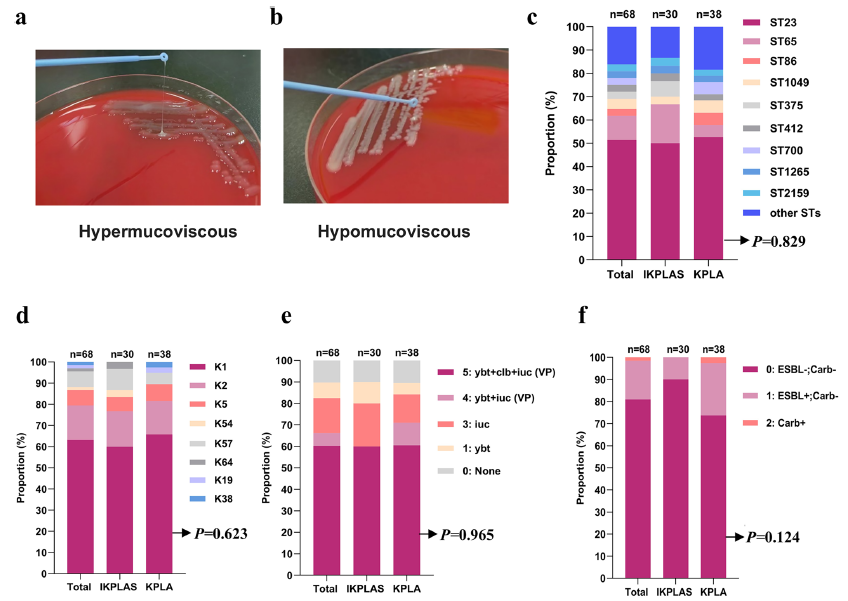
Comparison of molecular characteristics of Klebsiella pneumoniae between IKPLAS and KPLA groups (Original Figure 1)
2. Mechanistic Innovation: Lactate Promotes CPS Synthesis Through the PTS-CRP Axis
In vitro experiments showed that 40 mM lactate (simulating liver abscess microenvironment concentration) could induce peak CPS biosynthesis in Klebsiella pneumoniae, while simultaneously enhancing bacterial anti-phagocytic capacity (Figure 3f). At the molecular level, lactate downregulates gfrA-D gene expression of the mannose-specific phosphotransferase system (man-PTS), reducing intracellular cAMP levels, thereby attenuating cAMP binding to CRP and ultimately relieving the inhibition of CPS synthesis (Figures 5-7).
3. In Vivo Validation: Lactate Enhances Bacterial Virulence and Aggravates Infection Damage
In mouse liver abscess models, infection with lactate-treated Klebsiella pneumoniae resulted in significantly increased mortality (Figure 4b), elevated bacterial loads in liver and other tissues, more severe hepatic inflammatory damage (Figure 4c), and significantly increased serum levels of IL-6 and other inflammatory cytokines (Figure 4e), confirming that lactate enhances bacterial virulence through promoting CPS synthesis.

In vivo virulence enhancement of lactate-treated Klebsiella pneumoniae (Original Figure 4)
III. Absin Product Support: Precisely Capturing Immune Response Characteristics of Inflammatory Cytokines
In this study, quantitative detection of inflammatory cytokines was the core component for deciphering host immune responses. Absin's ELISA kits, with their high specificity and sensitivity, provided reliable support for the experimental results:
1. Absin Products Used
- Mouse IL-6 ELISA Kit (Catalog No.: abs520004-96T)
- Mouse TNF-α ELISA Kit (Catalog No.: abs520010-96T)
- Mouse IL-1β ELISA Kit (Catalog No.: abs520001-96T)
2. Product Applications and Functions
(1) Macrophage Immune Response Detection
Following infection of RAW264.7 macrophages with Klebsiella pneumoniae, Absin ELISA kits were used to detect IL-1β, IL-6, and TNF-α levels in cell culture supernatants (Figures 3i-k). The results showed that IL-1β protein expression was significantly elevated in the lactate-treated group, while IL-6 and TNF-α showed no significant changes, suggesting that lactate-induced CPS synthesis can specifically activate IL-1β-mediated inflammatory responses, providing critical data for understanding bacterial immune evasion mechanisms.
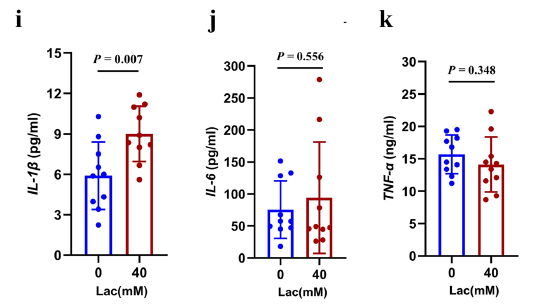
Inflammatory cytokine detection results from macrophage supernatants (Partial Original Figure 3)
(2) In Vivo Inflammatory Level Assessment in Mice
Absin ELISA kits were used to detect inflammatory cytokines in serum from infected mice (Figures 4e-f), confirming that infection with lactate-treated bacteria resulted in significantly elevated serum IL-6 and TNF-α levels, consistent with the degree of hepatic pathological damage (Figures 4c, g-h), directly validating the in vivo effects of lactate-enhanced bacterial virulence and providing in vivo evidence support for mechanistic conclusions.

Mouse serum IL-6 and TNF-α detection results (Partial Original Figure 4)
IV. Research Significance and Product Value
This study elucidated for the first time the critical role of lactate in IKPLAS pathogenesis, providing novel insights for clinical intervention through lactate level modulation. Absin's ELISA kits, as core tools for immune response detection, with their stable performance and precise quantitative results, assisted the research team in clearly delineating the pathological chain of "bacterial virulence enhancement → host inflammatory response dysregulation," providing indispensable technical support for mechanistic validation.
In infectious disease research, dynamic changes in inflammatory cytokines are important windows for deciphering host-pathogen interactions. Absin will continue to deepen its commitment to life science research tools, providing more cost-effective ELISA kits, antibodies, and other products to assist researchers in achieving more breakthrough results in infection mechanisms, immune regulation, and related fields!
This article is based on the original publication in Nature Communications (DOI: 10.1038/s41467-025-61379-9); all original figures, data, and intellectual property rights belong to the original journal and research team. If any infringement occurs, please contact us promptly for removal, and we will actively cooperate in handling such matters.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
