- Cart 0
- English
Uncovering the Pathogenesis of Obstetric Antiphospholipid Syndrome from a Single-Cell Perspective, with Absin Multicolor Kits Empowering Groundbreaking Research!
April 16, 2026
Clicks:75
Obstetric antiphospholipid syndrome (OAPS), as an autoimmune disorder, frequently leads to recurrent pregnancy loss, fetal demise, and other adverse pregnancy outcomes, posing severe threats to maternal and fetal health. Recently, a landmark study published in Advanced Science employed single-cell sequencing technology to comprehensively analyze the correlation between decidual and peripheral blood immune microenvironments in OAPS patients, revealing the central role of monocyte-derived macrophage (MDM) infiltration in disease pathogenesis. The Absin multiplex immunofluorescence kit (abs50013) provided critical technical support for the key experiments throughout this study, offering reliable validation for mechanistic investigations.
Article Title:Deciphering the Role and Mechanism of Decidual Monocyte-Derived Macrophage Infiltration in Obstetric Antiphospholipid Syndrome at Single-Cell Resolution
Journal:Advanced Science (IF 14.1)
DOI:https://doi.org/10.1002/advs.202503480
Absin Products Used:Five-Color Multiplex Immunofluorescence Kit (Mouse/Rabbit Universal Secondary Antibody) (abs50013)

I. Research Strategy: Focusing on Immune Cell Cross-Compartment Interactions to Decode Pathological Pregnancy
To address the scientific question of significant decidual macrophage infiltration with unclear cellular origins in OAPS patients, the research team designed a progressive investigation scheme:
- 1. Construction of single-cell atlases: Integration of single-cell sequencing data from decidual tissue and peripheral blood mononuclear cells (PBMCs) from OAPS patients and healthy controls (HCs), systematically comparing immune cell composition and functional differences between the two compartments;
- 2. Tracing cellular origins: Correlation analysis of immune cell proportion changes to validate the transformation pathway from peripheral monocytes to decidual MDMs;
- 3. Deciphering regulatory mechanisms: Investigation of MDM infiltration molecular mechanisms from three dimensions—chemokine recruitment, signaling pathway activation, and intercellular crosstalk;
- 4. Therapeutic target validation: Testing the therapeutic efficacy of TLR4 inhibitors and CCR2 antagonists in OAPS mouse models to provide evidence for clinical translation.
II. Core Research Findings: MDM Infiltration as the Key Driver of OAPS Pathogenesis
1. Dual Immune Microenvironment Disruption: Peripheral and Decidual Compartments Cooperate in Pathogenesis
- Peripheral blood: OAPS patients exhibited decreased monocyte proportions, reduced CD4⁺/CD8⁺ T cell ratios, and T cells displayed high cytotoxicity characteristics (Figure 2);
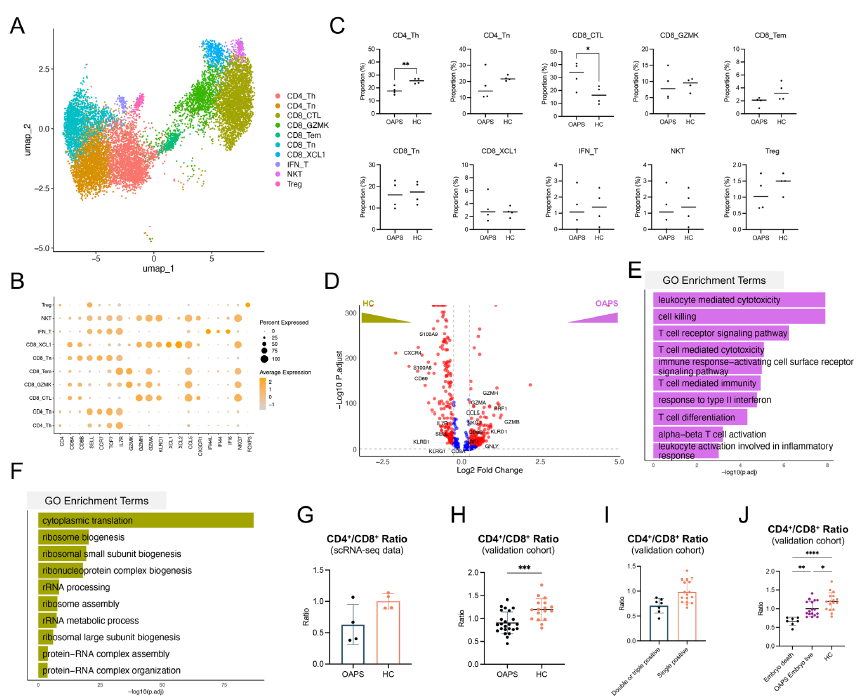
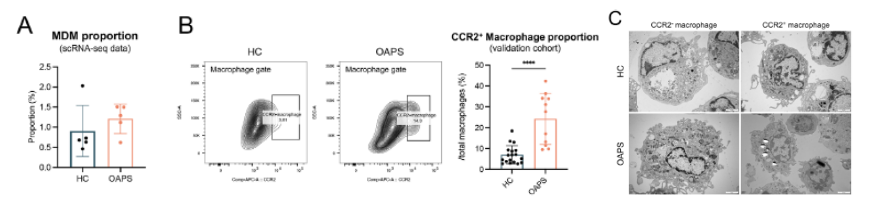
- Decidual tissue: Elevated macrophage proportions, reduced NK cell proportions, and massive infiltration of MDMs (CCR2⁺) with strong pro-inflammatory characteristics (Figure 3A-C).
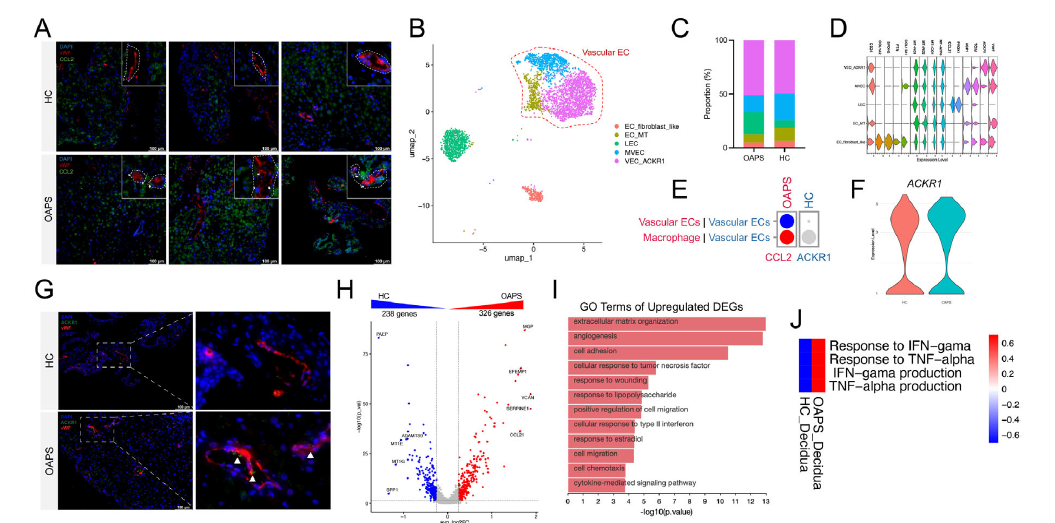
2. Molecular Mechanisms of MDM Infiltration: Triple-Pathway Regulatory Network
- Chemokine recruitment: The aPL-IgG/β₂GPI complex stimulates decidual macrophages to secrete CCL2 via the TLR4-NF-κB pathway (Figure 6);

- Vascular endothelial enrichment: Decidual vascular endothelial cells highly express ACKR1, which aggregates CCL2 on the cell surface, enhancing chemotaxis toward CCR2⁺ monocytes (Figure 8A-G);
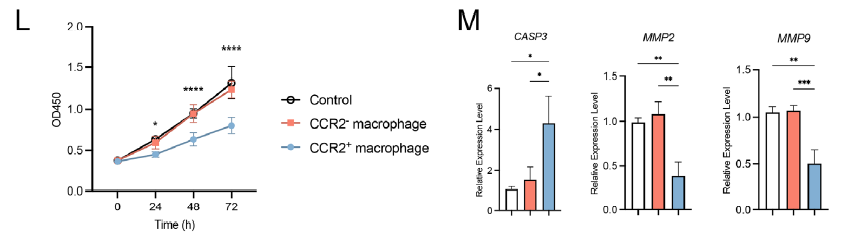
- Pathological effect amplification: MDMs secrete pro-inflammatory cytokines including IL1B and TNF, inhibiting trophoblast proliferation and invasion while promoting apoptosis, thereby disrupting maternal-fetal interface homeostasis (Figure 3L-M).
3. Therapeutic Target Validation: CCR2 and TLR4 as Potential Breakthrough Points
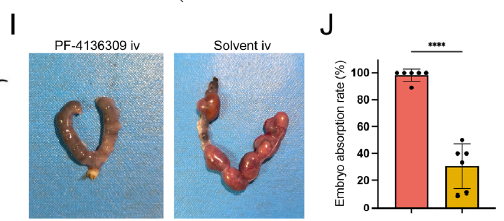
In OAPS mouse models, both CCR2 antagonist (PF-4136309) and TLR4 inhibitor (TAK-242) significantly reduced embryo resorption rates and decreased decidual MDM infiltration (Figure 5I-J, Figure 7G-H), providing novel therapeutic directions for refractory OAPS.
III. Absin Product Support: Multiplex Immunohistochemistry Kit Addresses Critical Validation Challenges
| Product Core Information | Detailed Content |
|---|---|
| Product Name and Catalog Number | Absin Multiplex Immunohistochemistry Kit (abs50013, based on Tyramide Signal Amplification [TSA] technology) |
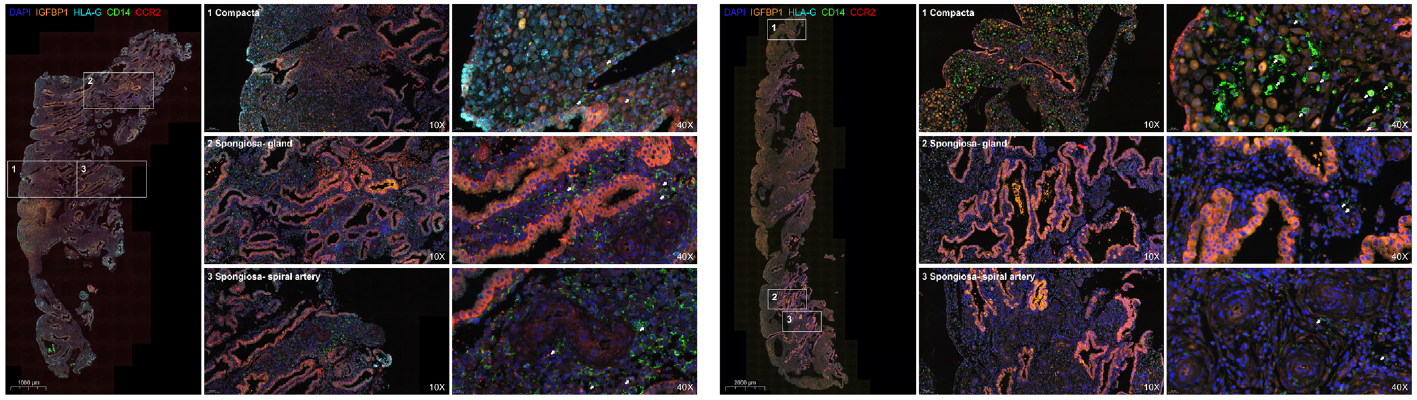
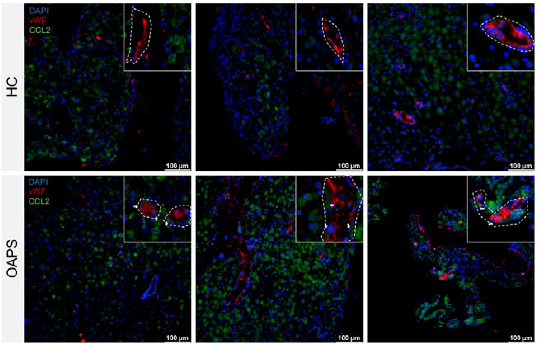
| Product Applications and Corresponding Figures | 1. Macrophage subtype localization: Simultaneous labeling of CD14 (macrophage marker), CCR2 (MDM marker), IGFBP1 (decidual zone marker), and HLA-G (trophoblast marker), demonstrating abnormal enrichment of CCR2⁺ MDMs in the decidual compact layer (Figure 3J-K); 2. Chemokine and receptor co-localization: Detection of vWF (vascular endothelial marker) co-expression with CCL2 and ACKR1, validating the aggregation function of vascular endothelial cells for CCL2 (Figure 8A, G); 3. Inflammatory cytokine expression validation: Quantitative analysis of IFN-γ and TNF-α expression levels in decidual tissue, providing evidence for ACKR1 upregulation mechanisms (Figure 8K-L). |
| Core Product Functions | 1. Multi-marker simultaneous detection: Overcoming the limitation of traditional IHC for single-target detection, enabling simultaneous visualization of cell type, functional molecules, and tissue localization indicators, clearly presenting intercellular crosstalk relationships; 2. Ensuring detection sensitivity for low-abundance molecules: TSA signal amplification technology significantly enhances detection efficiency of low-expression proteins such as CCL2 and ACKR1, ensuring experimental accuracy; 3. Providing precise quantitative basis: Through multi-channel fluorescence imaging and semi-quantitative analysis, providing reliable data support for immune cell proportion statistics and molecular expression level comparisons. |

IV. Research Implications: Precision Tools Empower Immune-Related Disease Research
The breakthrough progress of this study relies on the synergistic application of single-cell sequencing technology and high-sensitivity immunodetection tools. The Absin multiplex immunohistochemistry kit (abs50013), with its multi-target detection capability and high-sensitivity advantages, successfully assisted the research team in deciphering complex intercellular crosstalk networks, providing efficient solutions for similar research in autoimmune diseases, tumor microenvironments, and other fields.
This article is based on the original publication in Advanced Science (DOI: 10.1002/advs.202503480); all original figures, data, and intellectual property rights belong to the original journal and research team. If any infringement occurs, please contact us promptly for removal, and we will actively cooperate in handling such matters.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
