- Cart 0
- English
Breakthrough Discovery! PHOSPHO1 Emerges as a Novel Therapeutic Target for AMD Treatment, with Absin Antibodies Powering Ferroptosis Mechanism Research
April 16, 2026
Clicks:76
Age-related macular degeneration (AMD) is one of the leading causes of vision loss in the elderly, and ferroptosis of retinal pigment epithelial (RPE) cells has been identified as a core mechanism in AMD pathogenesis. A recent landmark study published in Advanced Science revealed the dual mechanism by which PHOSPHO1 suppresses ferroptosis in RPE cells through regulating phosphatidylethanolamine (PE) metabolism and autophagy, providing a novel therapeutic target for AMD treatment. Absin premium antibody products provided comprehensive support for mechanistic validation in this study, safeguarding scientific breakthroughs!
Title: PHOSPHO1 Suppresses Ferroptosis in Retinal Pigment Epithelial Cells by Reducing the Levels of Phosphatidylethanolamine Molecular Species
Journal: Advanced Science (IF 14.1) | DOI: https://doi.org/10.1002/advs.202505359
Absin Products Used: NCOA4 antibody

I. Research Strategy: Precise Identification of Key Ferroptosis Regulators
Core Scientific Question
PE-mediated lipid peroxidation serves as the primary driving force of RPE cell ferroptosis, while autophagy activation exacerbates ferroptosis progression. Previous studies demonstrated that transcription factor reprogramming-derived induced RPE (iRPE) cells possess enhanced therapeutic potential for AMD; however, the molecular mechanisms underlying their ferroptosis resistance remained elusive.
Research Design Logic
- Cellular model comparison: Utilizing iRPE cells as the core model, compared with dedifferentiated iPSC-RPE (De-iPSC-RPE) and iPSC-RPE cells to validate their ferroptosis-resistant characteristics;
- Metabolic mechanism investigation: Focusing on PE molecular species (particularly PE-AA/AdA) levels in the endoplasmic reticulum (ER) to establish their association with ferroptosis;
- Key factor screening: Differential expression protein analysis via mass spectrometry within PE metabolic pathways, identifying PHOSPHO1 as the critical regulator;
- Dual mechanism validation: Elucidating PHOSPHO1 function through "reducing PE synthesis" and "inhibiting autophagy/ferritinophagy" pathways;
- In vivo and in vitro validation: Combining cellular experiments with rat AMD models to confirm the therapeutic potential of PHOSPHO1.
II. Core Research Findings: Dual Anti-Ferroptosis Mechanisms of PHOSPHO1
1. iRPE Cells Exhibit Intrinsic Ferroptosis Resistance
Cells were treated with three ferroptosis inducers: erastin, ferric ammonium citrate (FAC), and RSL3. iRPE cells maintained high viability even under high concentrations of inducers, with significantly lower levels of reactive oxygen species (ROS), free iron, and MDA (lipid peroxidation marker) compared to the other two cell types (Figure 1A-E). Additionally, iRPE cells showed minimal cell death proportions (Figure 1F-G), confirming that their ferroptosis resistance is independent of epithelial phenotype and is regulated by specific molecular mechanisms.
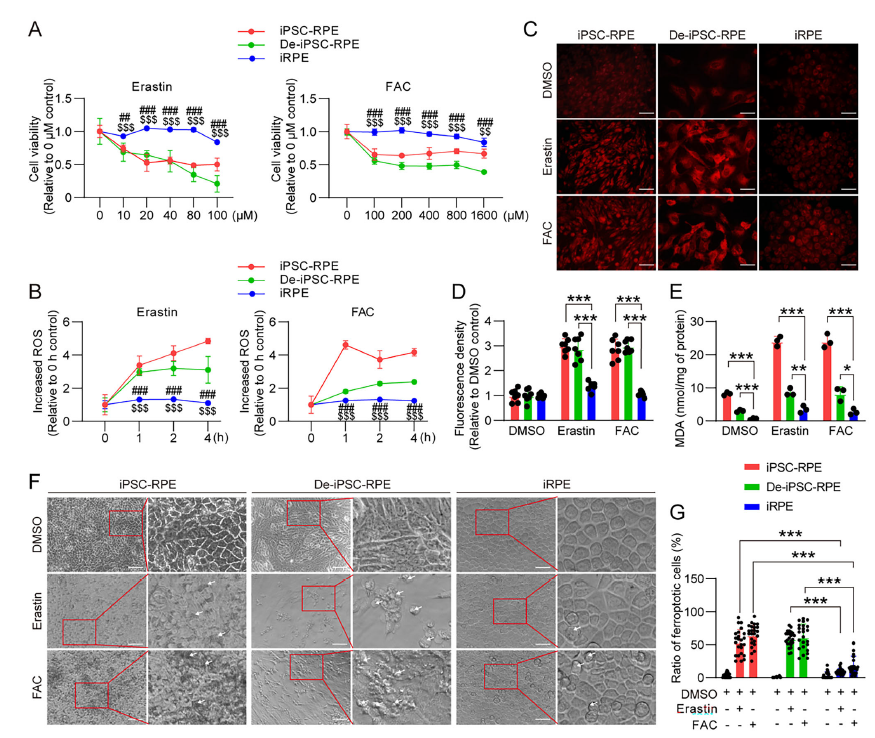
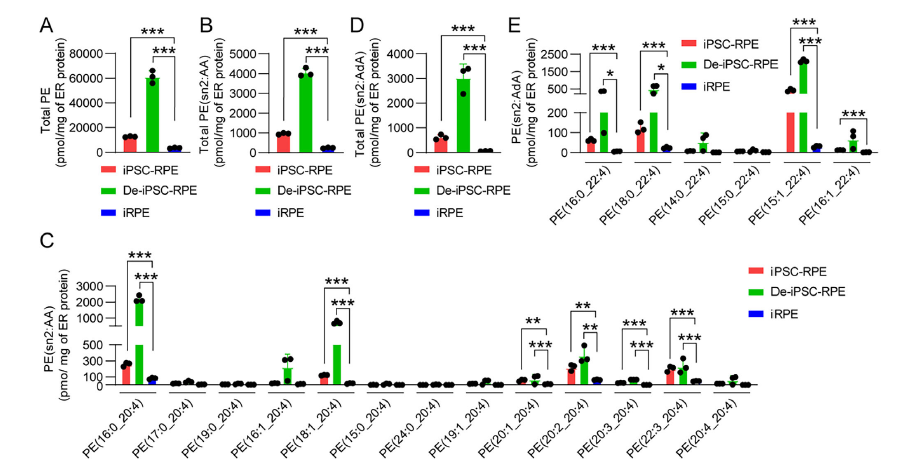
2. Reduced PE Levels Are Critical for Ferroptosis Resistance
Lipidomic analysis revealed significantly decreased total PE levels and key lipid peroxidation substrates PE-AA and PE-AdA in the ER of iRPE cells (Figure 2A-E). Intervention experiments using meclizine to inhibit PE synthesis or ethanolamine to elevate PE levels directly demonstrated that moderate PE reduction enhances RPE cell ferroptosis resistance, whereas excessive PE depletion leads to cell death.
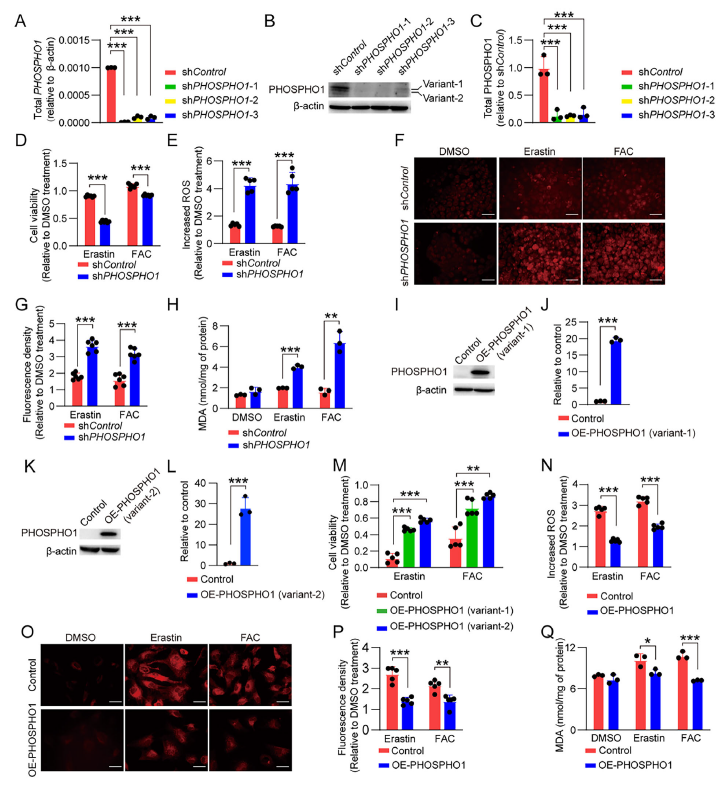
3. PHOSPHO1 Is the Core Regulatory Factor
Mass spectrometry screening identified significantly elevated PHOSPHO1 expression in iRPE cells (Figure 3D-G). Functional validation revealed:
- PHOSPHO1 knockdown: Elevated PE levels and significantly increased ferroptosis sensitivity in iRPE cells (Figure 4D-H);
- PHOSPHO1 overexpression: Reduced PE levels, decreased ROS, free iron, and MDA levels, and enhanced ferroptosis resistance in De-iPSC-RPE/iPSC-RPE cells (Figure 4M-Q).
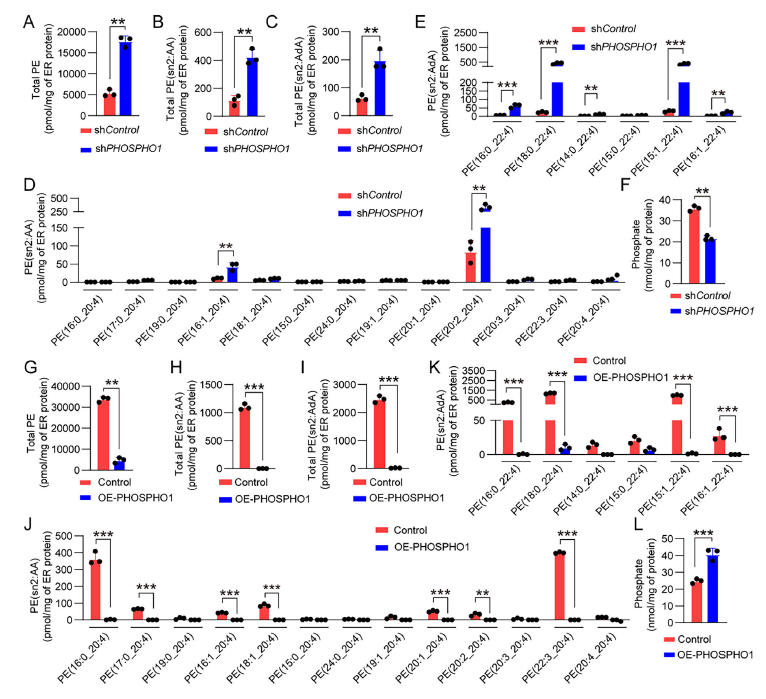
4. Dual Mechanism Elucidation: Metabolic Regulation + Autophagy Inhibition
Mechanism I: Inhibition of PE Synthesis
PHOSPHO1 hydrolyzes phosphoethanolamine, a substrate for PE synthesis, thereby reducing ER PE levels and attenuating lipid peroxidation (Figure 5A-L);
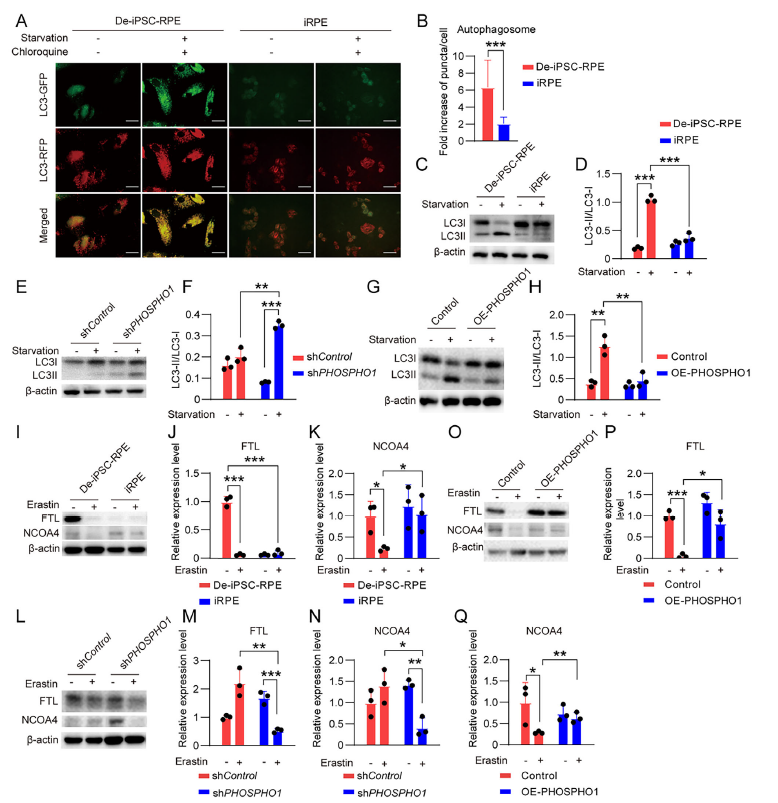
Mechanism II: Attenuation of Autophagy/Ferritinophagy
PE is a critical molecule for autophagosome formation (LC3-I→LC3-II). PHOSPHO1 inhibits autophagy by reducing PE levels (Figure 6A-H), while simultaneously decreasing ferritinophagy (mediated by NCOA4), thereby reducing intracellular free iron accumulation (Figure 6I-Q).
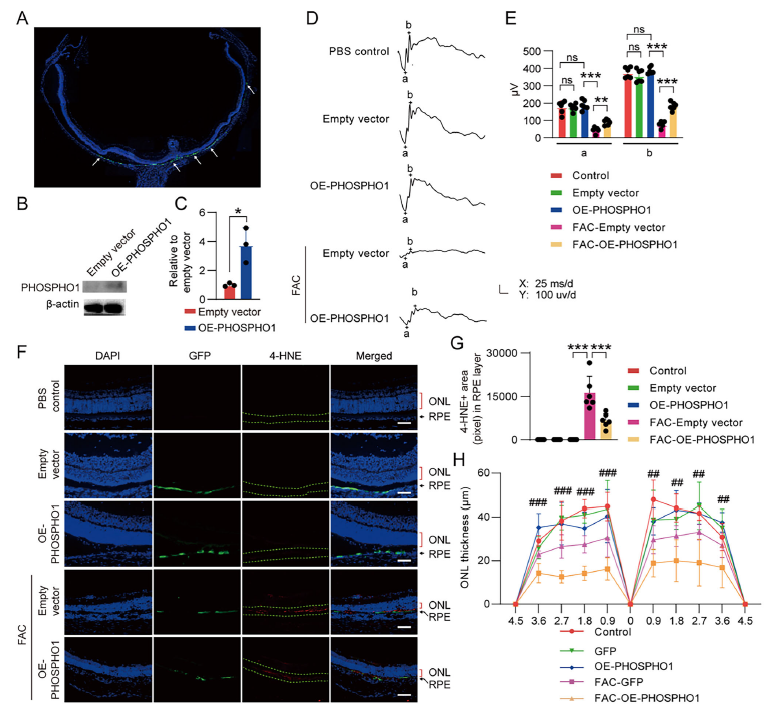
5. In Vivo Validation: PHOSPHO1 Ameliorates Pathological Phenotypes in Rat AMD Models
In FAC-induced rat AMD models, PHOSPHO1 overexpression achieved the following:
- Retinal function preservation: Maintenance of ERG a-wave and b-wave amplitudes (Figure 7D-E);
- RPE cell ferroptosis inhibition: Reduction of 4-HNE (lipid peroxidation marker) levels (Figure 7F-G);
- Retinal structural protection: Attenuation of outer nuclear layer (ONL) thinning, indirectly protecting photoreceptor cells (Figure 7F-H).
III. Absin Product Support: Core Tools for Key Mechanism Validation
During the mechanistic validation phase of this study, Absin's NCOA4 antibody (Catalog No.: abs139169) played an irreplaceable role, providing precise detection tools for analyzing ferritinophagy pathway regulation.
Product Information
| Product Name | Applications |
|---|---|
| NCOA4 Antibody | Western blot, Immunofluorescence (IF) |
Core Functions in the Study
-
Ferritinophagy pathway validation: Western blot detection revealed that following PHOSPHO1 knockdown, NCOA4 expression increased and FTL (ferritin light chain) expression decreased in iRPE cells, confirming ferritinophagy activation (Figure 6L-N); PHOSPHO1 overexpression significantly inhibited NCOA4 expression, blocking ferritinophagy (Figure 6O-Q).

-
In vivo mechanism confirmation: In rat AMD models, immunofluorescence staining demonstrated enhanced NCOA4 expression in RPE cells of the FAC-treated group, whereas PHOSPHO1 overexpression significantly reduced NCOA4 staining intensity (Figure S11), directly validating the in vivo mechanism by which PHOSPHO1 inhibits ferritinophagy to reduce free iron accumulation.
Product Advantages
- High specificity: Precise recognition of NCOA4 protein without cross-reactivity, producing clear Western blot bands (Figure 6I, L, O);
- Multi-application compatibility: Supports both Western blot and immunofluorescence experiments, meeting multi-dimensional validation requirements in vitro and in vivo;
- High stability: Reproducible experimental results providing reliable data support for mechanistic conclusions.
IV. Research Significance and Application Prospects
This study is the first to elucidate the dual mechanisms by which PHOSPHO1 functions as a ferroptosis suppressor in RPE cells, providing a novel therapeutic target for AMD treatment. Furthermore, it confirms that moderate regulation of PE metabolism can effectively inhibit ferroptosis, offering new insights for developing lipid metabolism-targeted AMD therapeutics.
As a reliable partner in life science research, Absin is committed to providing high-quality antibodies, reagents, and other experimental tools. The successful application of the NCOA4 antibody in this top-tier journal study once again validates the stability and reliability of Absin products. Moving forward, Absin will continue focusing on research hotspots, providing comprehensive tool support for studies in ferroptosis, neurodegenerative diseases, and other fields, facilitating more scientific breakthroughs!
This article is based on the original publication in Advanced Science (DOI: 10.1002/advs.202505359); all original figures, data, and intellectual property rights belong to the original journal and research team. If any infringement occurs, please contact us promptly for removal, and we will actively cooperate in handling such matters.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
