- Cart 0
- English
Unlocking a new mechanism underlying the malignant progression of lung adenocarcinoma! absin helps reveal a novel regulatory pathway of MYC+ M2 macrophages
April 15, 2026
Clicks:82
As a brand deeply committed to life science research tools, absin has always focused on cutting-edge scientific research, empowering scientists to explore the core mechanisms of disease pathogenesis and progression. Recently, a landmark study published in Advanced Science revealed a novel regulatory pathway driving the malignant progression of micropapillary-patterned lung adenocarcinoma (MP-LUAD), with absin tissue dissociation solution providing crucial support for the successful advancement of this research.
Title: Excessive MYC Orchestrates Macrophages induced Chromatin Remodeling to Sustain Micropapillary-Patterned Malignancy in Lung Adenocarcinoma
Journal: Advanced Science (IF 14.1) | DOI: https://doi.org/10.1002/advs.202403851
Absin Product Used: Tissue Dissociation Solution (abs9482)

I. Research Background: MP-LUAD—A Highly Invasive "Refractory Subtype"
Lung adenocarcinoma (LUAD) is the most common subtype of lung cancer, among which the micropapillary (MP) subtype has become a clinical therapeutic challenge due to its high invasiveness, high metastatic rate, and poor prognosis. Previous studies have established that MP-LUAD exhibits characteristics such as anchorage-independent growth and anoikis resistance, but the core regulatory mechanisms remained unclear.
The team discovered aberrant activation of the MYC pathway in MP-LUAD patients; however, MYC overexpression alone was insufficient to induce the MP phenotype, suggesting that immune cells in the tumor microenvironment (TME) may play a synergistic role—this critical insight became the starting point for the present study.
II. Research Strategy: Progressive Investigation to Unlock the "MYC + M2 Macrophage" Synergistic Regulatory Network
- Clinical Sample Validation: Through tissue microarray analysis of 66 MP-LUAD patients (MAPes cohort), the correlation between MYC pathway activation and MP phenotype was confirmed, along with the unique molecular characteristics of MP-LUAD (such as abnormal expression of EMT markers and shear stress resistance markers).
- In Vitro and In Vivo Functional Validation: MYC overexpression combined with M2-like macrophages was validated in LUAD cell lines, mouse xenograft models, and zebrafish xenotransplantation models to induce MP phenotypes (such as polarity changes and increased circulating tumor cells).
- Mechanistic Elucidation: Through RNA-seq, ChIP-seq, and CUT&Tag-seq technologies, the molecular pathway was revealed: M2-like macrophages secrete TGFβ, inducing FOSL2 expression in tumor cells, which subsequently remodels chromatin accessibility and promotes MYC binding to MP-associated gene promoters.
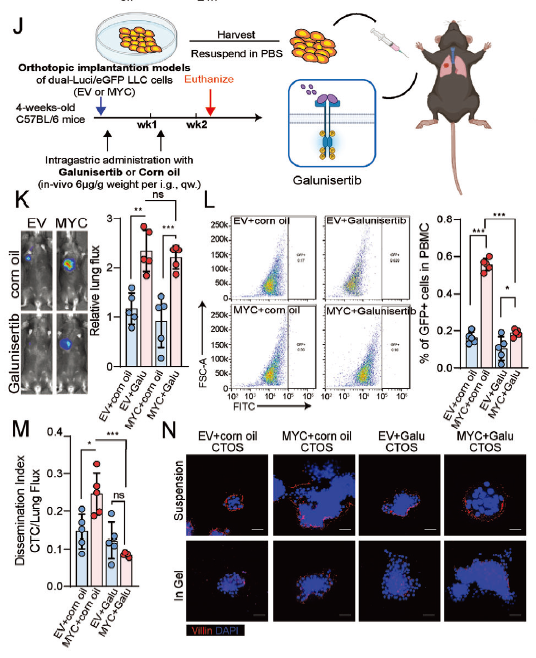
- Therapeutic Target Validation: Blockade of this pathway using the TGFβ receptor inhibitor Galunisertib effectively reversed the malignant phenotype of the MP subtype, providing a new direction for clinical treatment.
III. Core Research Findings: Four Major Breakthroughs Refreshing Understanding of MP-LUAD
1. Key Driver Pathway:
The malignant progression of MP-LUAD depends on the synergistic interaction of "MYC overexpression + M2-like macrophage infiltration"; neither factor alone can induce the complete MP phenotype.
2. Molecular Regulatory Chain:
TGFβ secreted by M2-like macrophages activates FOSL2 (a pioneer transcription factor) in tumor cells; FOSL2 opens the chromatin structure of MP-associated genes, providing "targets" for MYC binding, ultimately activating malignant programs including EMT and anoikis resistance (corresponding to original Figures 6, 7).
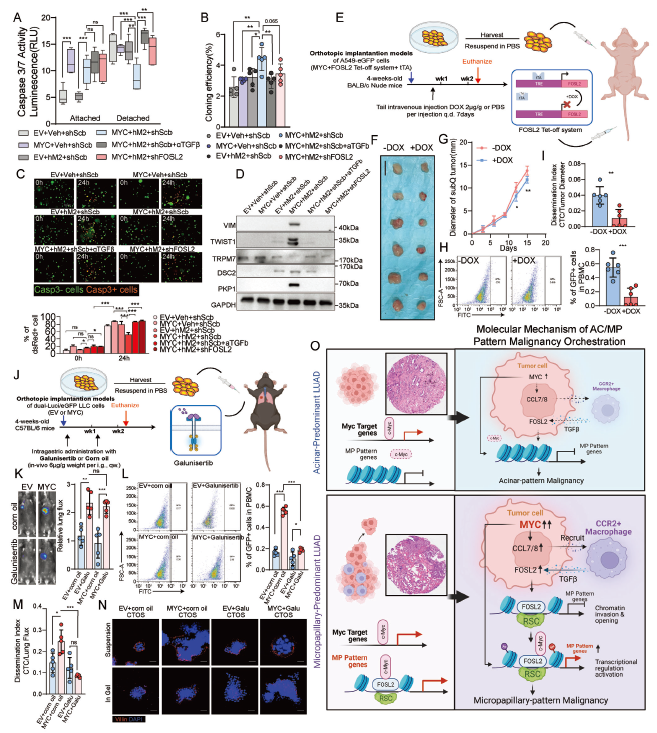
3. Cellular Origin:
The M2-like macrophages involved in this regulation primarily originate from bone marrow hematopoietic stem cells, recruited by MYC-overexpressing tumor cells through the CCL7/CCR2 pathway (corresponding to original Figure 3).
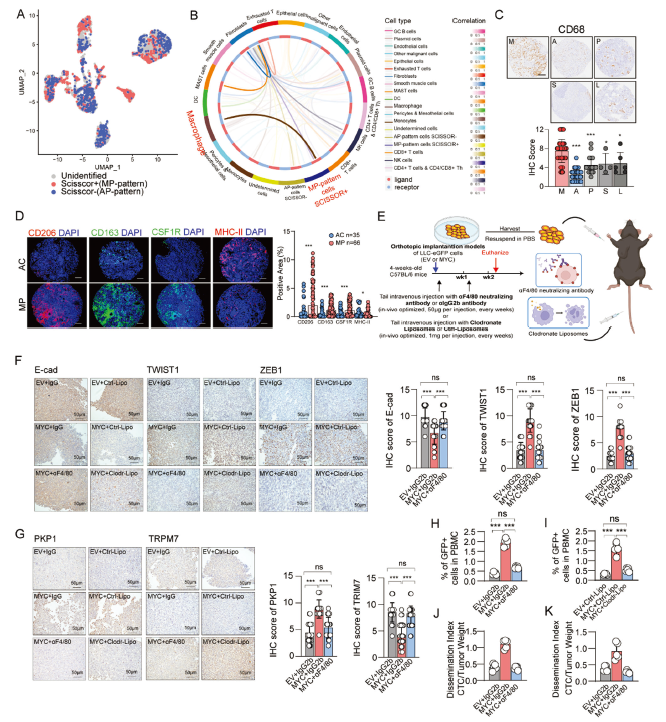
4. Novel Therapeutic Target:
Blockade of the TGFβ-FOSL2 axis (e.g., using Galunisertib) significantly reduces the metastatic capacity of MP-LUAD, providing a new strategy for clinical precision therapy (corresponding to original Figure 7J-N).
IV. absin Product Support: The "Core Tool" for Critical Experiments
In this study, absin's tissue dissociation solution (Cat. No.: abs9482) played a key role in tumor organoid (CTOS) preparation, directly supporting functional validation experiments related to the MP phenotype.
Product Information and Application Scenarios
| Product Name | Cat. No. | Application Scenario |
|---|---|---|
| Tissue Dissociation Solution | abs9482 | Isolation of single cells from fresh tumor tissue for preparation of tumor organoids (CTOS) |
Core Role of the Product in the Research
The study required validation of MP phenotype polarity characteristics through patient-derived tumor organoids (CTOS)—MP-derived CTOS cultured in Matrigel maintain a unique "apical-out" polarity, which is a key characteristic of anchorage-independent growth in MP-LUAD (corresponding to original Figures 1G, 4D).
With its efficient tissue digestion capability, absin tissue dissociation solution enabled rapid isolation of high-viability single cells from fresh MP-LUAD tissue, successfully establishing a stable, passagable CTOS model. Subsequent immunofluorescence staining (Villin labeling of apical membranes) validated polarity changes, providing direct functional evidence for "MYC + M2 macrophage synergistic regulation of MP phenotype" (corresponding to original Figures 4D-G).
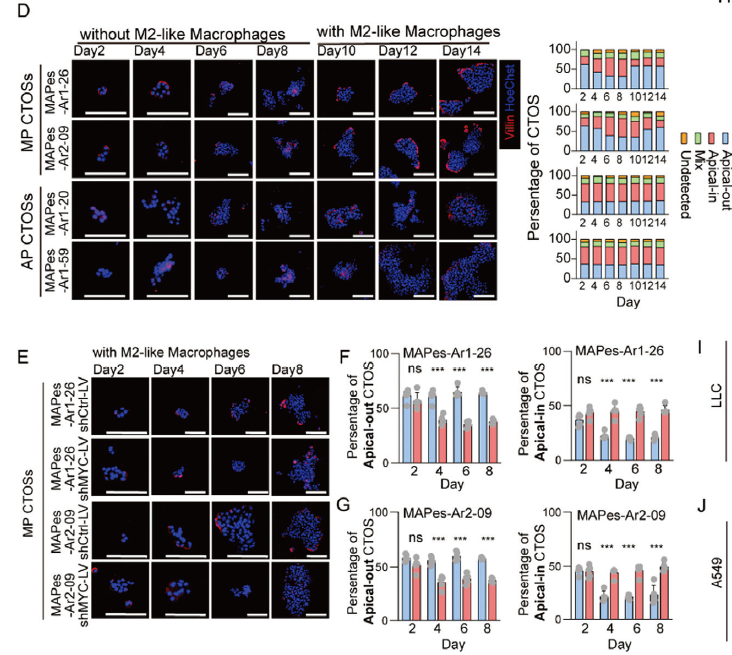
In summary, the reliable performance of absin tissue dissociation solution ensured the successful establishment of the tumor organoid model, building a critical bridge for in vitro and in vivo functional validation.
V. Research Significance: From Mechanism to Translation, Opening New Paths for MP-LUAD Treatment
This study is the first to reveal the cross-cellular regulatory network of "tumor cells - immune cells - chromatin remodeling" in MP-LUAD, not only deepening the understanding of lung cancer subtype heterogeneity but also clarifying the value of the TGFβ-FOSL2 axis as a potential therapeutic target.
As a provider of research tools, absin has always supported the connection between basic research and clinical translation with high-quality products—from tissue sample processing to cellular functional validation, absin products are providing reliable support for more tumor mechanism studies and target discovery.
This article is based on the original publication in Advanced Science (DOI: 10.1002/advs.202403851); all original figures, data, and intellectual property rights belong to the original journal and research team. Should any infringement occur, please contact us promptly for removal, and we will actively cooperate in addressing such matters.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
