- Cart 0
- English
Macrophage siRNA Delivery "Bottleneck" Cracked? Absin Empowers Nanoparticle System to Break Through Degradation Barrier, Illuminating New Pathways for Bone Defect Repair
April 15, 2026
Clicks:81
In the field of tissue regeneration, the polarization state of macrophages directly determines the repair outcome—pro-inflammatory M1 phenotype exacerbates tissue damage, while pro-regenerative M2 phenotype creates an ideal microenvironment for repair. How to efficiently and precisely induce macrophage polarization from M1 to M2 has remained a core challenge in clinical translation. A landmark study recently published in Advanced Materials innovatively constructed an siAkt2-loaded nanoparticle delivery system, successfully achieving macrophage metabolic reprogramming and polarization regulation, providing a novel strategy for the repair of inflammation-related bone defects such as periodontitis. As a key reagent supplier, Absin's high-quality products provided solid support for the successful execution of this research.
Title: siAkt2-Loaded Nanoparticles Reprogramming Macrophages to M2 Phenotype for Effective Bone Defect Repair
Journal: Advanced Materials (IF 26.8) | DOI: https://doi.org/10.1002/adma.202410507
Absin Product Used: iNOS Antibody

I. Research Strategy: Precision Targeting + Efficient Delivery to Overcome Macrophage Regulation Challenges
Core Challenge
siRNA-mediated gene silencing is a powerful tool for regulating cellular functions; however, macrophages contain abundant nucleases that rapidly degrade siRNA, and traditional carriers have limited loading capacity, making it difficult to reach effective silencing thresholds. Meanwhile, Akt2 kinase, as a key regulator of glycolysis, its overactivation drives macrophage M1 polarization, exacerbating inflammation and bone resorption.
Solution
The research team designed a triple delivery strategy of "multi-repeat sequence loading + cholesterol modification compression + PLLA encapsulation":
- 1. Rolling circle transcription (RCT) technology was employed to construct RNA microspheres containing numerous repetitive siAkt2 sequences, achieving high-density siRNA loading;
- 2. Cholesterol-modified DNA (DNA-Chol) was used to compress the RNA into nano-sized RNA complexes (siAkt2 RNPs), enhancing intracellular internalization efficiency;
- 3. Biocompatible PLLA encapsulation formed siAkt2 RNP@PLLA NPs, enhancing physiological stability and lysosomal escape capability, ultimately achieving sustained intracellular release of siAkt2, Akt2 gene silencing, and induction of macrophage M2 polarization (Figure 1, corresponding to original Scheme 1).
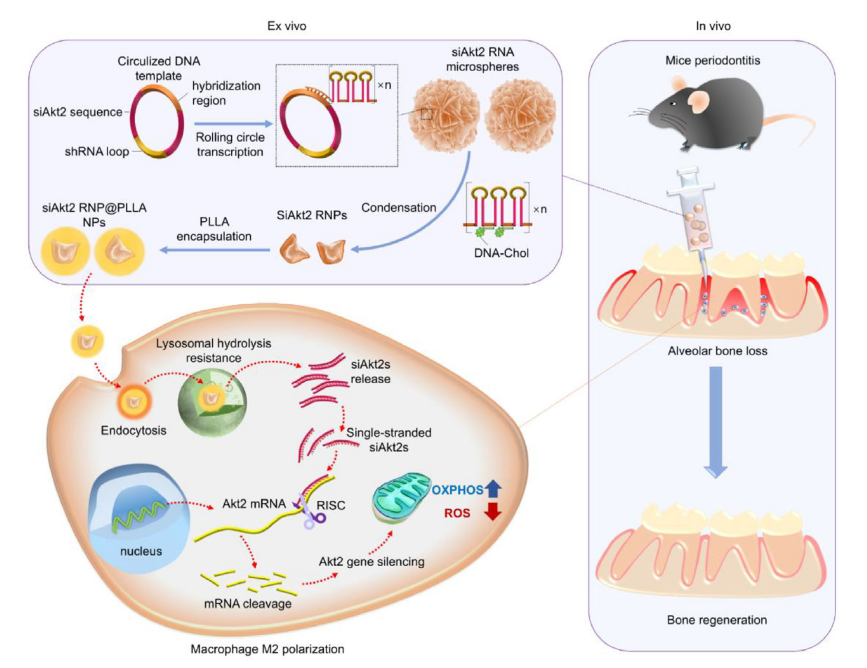
Figure 1 (Original Scheme 1): Construction process of the nanoparticle delivery system, intracellular action pathway, and bone regeneration mechanism
II. Core Research Findings: Comprehensive Validation from In Vitro Mechanisms to In Vivo Repair
1. Excellent Nanocarrier Performance with Superior Stability and Delivery Efficiency
Structure and Stability:
siAkt2 RNP@PLLA NPs exhibited a clear core-shell structure with an average particle size of 293.16±33.18 nm, maintaining 75.85% integrity in physiological environments for up to 120 hours, and demonstrated tolerance to lysosomal acidic conditions (Figure 2, corresponding to original Figure 1c-e);

Figure 2 (Original Figure 1): a) Morphology of siAkt2 microspheres and RNPs; c) TEM image of NP core-shell structure; d) Stability in physiological fluids; e) Morphological changes under acidic conditions
Intracellular Delivery:
The PLLA shell significantly promoted macrophage endocytosis, achieving efficient lysosomal escape within 24 hours with sustained siAkt2 release, demonstrating transfection efficiency far exceeding traditional liposome carriers (Figure 3, corresponding to original Figure 2a-b).
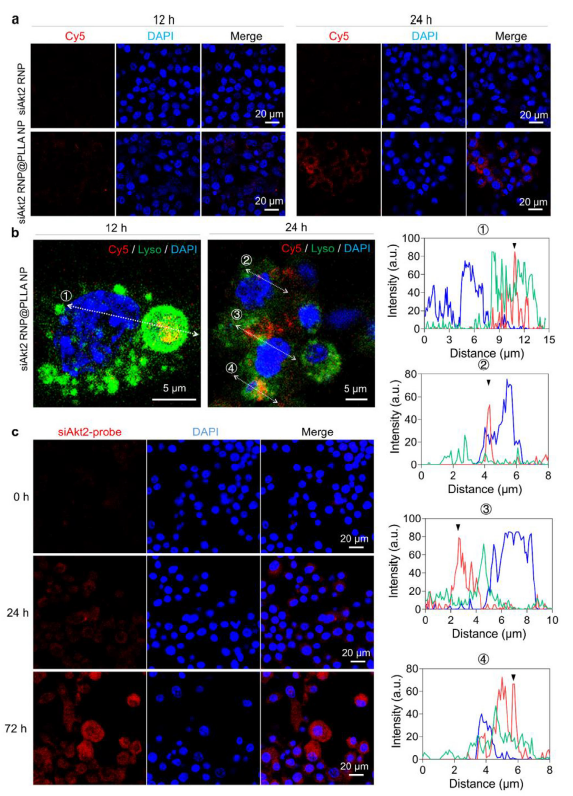
Figure 3 (Original Figure 2): a) Endocytosis efficiency at different time points; b) Fluorescence imaging at 12 h (lysosomal colocalization) and 24 h (lysosomal escape)
2. Precise Macrophage Functional Regulation Achieving Metabolic Reprogramming and M2 Polarization
Gene Silencing Efficiency:
qPCR and Western blot confirmed that this system could suppress Akt2 gene expression to 40-50%, with silencing effects persisting for more than 7 days (Figure 4, corresponding to original Figure 3a-b);
Phenotype Switching:
Significantly increased the positive rate of M2 marker CD206 (Figure 4c, corresponding to original Figure 3c), downregulated pro-inflammatory factors including IL-1β and TNF-α, and upregulated anti-inflammatory factors including IL-4 and IL-10 (Figure 4d-e, corresponding to original Figure 3d-e);
Metabolic Reprogramming:
Suppressed anaerobic glycolysis, enhanced oxidative phosphorylation (OXPHOS), reduced ROS generation, and restored mitochondrial function (Figure 5, corresponding to original Figure 4).
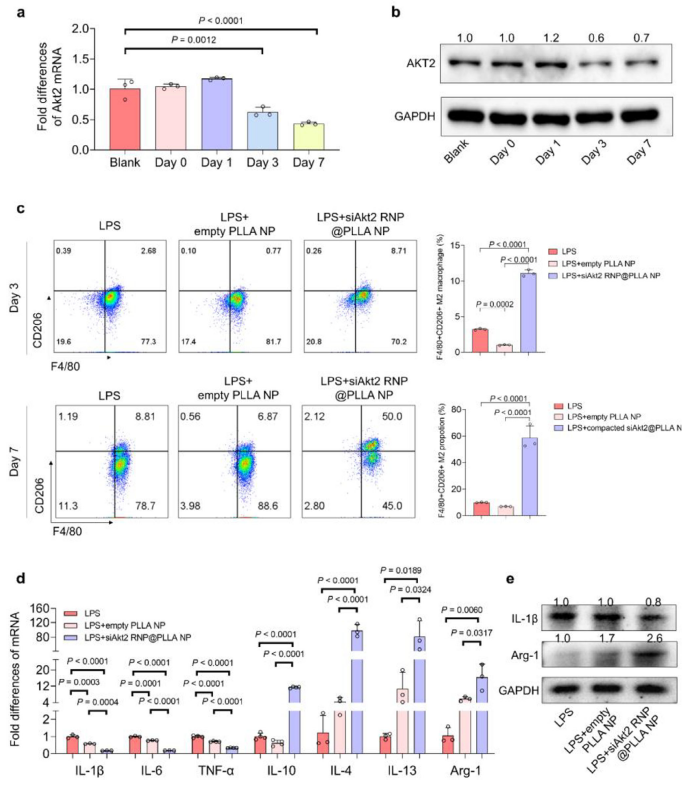
Figure 4 (Original Figure 3): a) Akt2 gene silencing efficiency; b) AKT2 protein expression changes; c) F4/80+CD206+ M2 cell proportion; d-e) Inflammatory factor expression regulation

Figure 5 (Original Figure 4): a) Extracellular acidification rate (glycolysis) and oxygen consumption rate (OXPHOS); f) ROS generation inhibition; h) Mitochondrial membrane potential restoration; i) Schematic diagram of metabolic reprogramming mechanism
3. Significant In Vivo Bone Repair Effects with Tremendous Clinical Translation Potential
In a mouse periodontitis bone defect model, following local injection of siAkt2 RNP@PLLA NPs:
Bone Regeneration Capability:
After 4 weeks, bone volume fraction (BV/TV) was significantly increased, with bone defect height restored to more than 2/3 of normal levels (Figure 6, corresponding to original Figure 5d-e);

Figure 6 (Original Figure 5): d) Micro-CT 3D reconstruction and bone volume analysis; e) HE staining showing bone tissue regeneration and periodontal ligament formation
Immune Microenvironment Improvement:
Significantly reduced CD68+iNOS+ M1 macrophages, increased CD68+CD163+ M2 macrophages, creating a pro-repair microenvironment (Figure 7, corresponding to original Figure 6b).
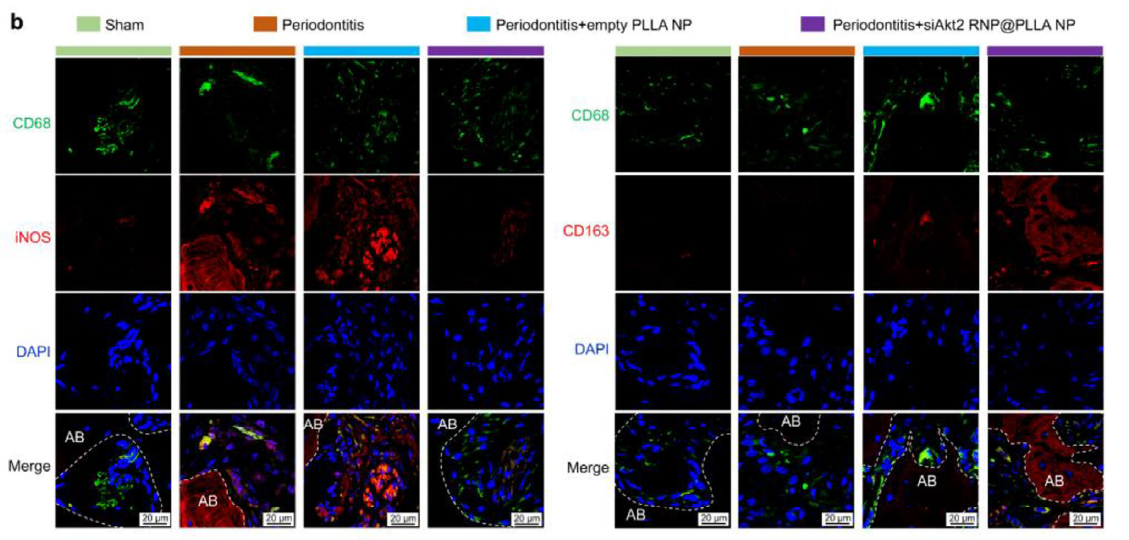
Figure 7 (Original Figure 6b): Immunofluorescence staining showing changes in M1 (CD68+iNOS+) and M2 (CD68+CD163+) macrophage proportions
III. Absin Product Support: Key Reagents Empowering Core Research Components
In the core mechanism validation and phenotypic analysis of this study, Absin's iNOS antibody played an indispensable role:
Product Information
| Product Name | Application Scenario |
|---|---|
| iNOS Antibody | Immunofluorescence Staining (IF) |
Core Functions
As a specific marker for M1 macrophages, iNOS expression levels directly reflect the proportion of pro-inflammatory macrophages. In this study, dual immunofluorescence staining using Absin's iNOS antibody together with CD68 antibody (pan-macrophage marker) precisely quantified changes in M1 macrophage numbers across different experimental groups:
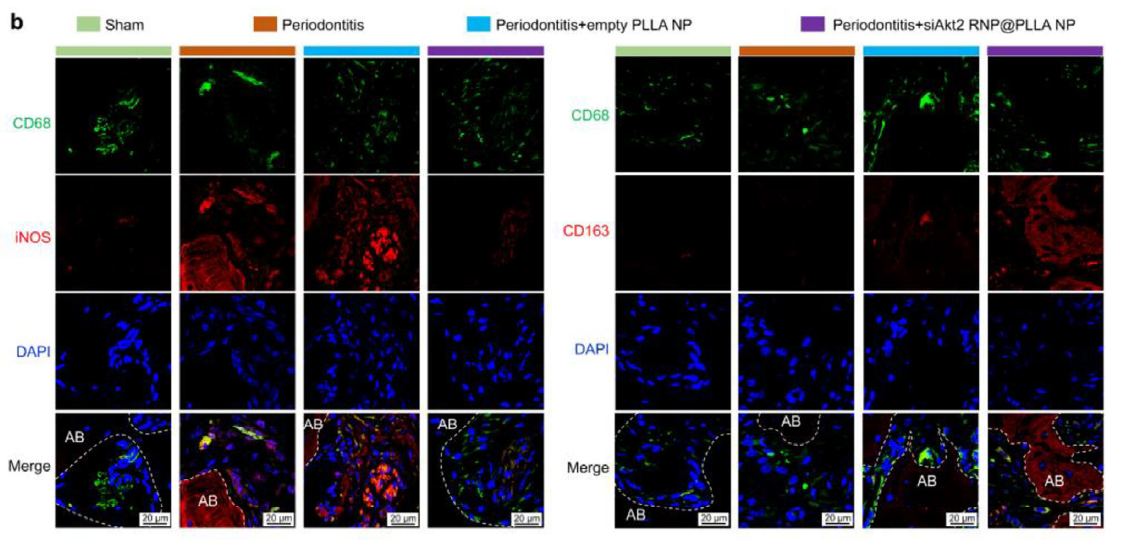
- 1. Clearly distinguished M1 (CD68+iNOS+) and M2 (CD68+CD163+) macrophage populations, visually demonstrating the regulatory effects of siAkt2 RNP@PLLA NPs on macrophage polarization;
- 2. Provided direct morphological evidence for the core conclusion that "the nanoparticle delivery system induces M2 polarization through Akt2 silencing";
- 3. The antibody's high specificity and fluorescence stability ensured reliable in vivo tissue staining results, laying the foundation for subsequent bone regeneration mechanism analysis.
IV. Summary and Future Perspectives
Through innovative nanoparticle delivery system design, this study successfully overcame the technical bottlenecks of low siRNA delivery efficiency and short duration of action in macrophages, providing an integrated solution of "gene silencing - metabolic reprogramming - immune regulation" for inflammation-related tissue repair. Absin's iNOS antibody, with its excellent specificity and applicability, became a critical tool for macrophage phenotypic analysis, enabling the research team to precisely validate the core mechanisms.
In the future, as nanoparticle delivery technology and targeted therapeutic strategies continue to integrate deeply, Absin will continue to provide high-quality reagents for immunodetection, molecular biology, and related series, empowering scientific innovation in tissue regeneration, tumor immunology, inflammation regulation, and other fields, working together with global researchers to drive breakthroughs and developments in clinical translational research!
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
