- Cart 0
- English
Insufficient Antigen Presentation + Immune Evasion? Cascade-Responsive Peptide Self-Assembly Technology Breaks the Deadlock! Absin Empowers New Breakthroughs in Combined Tumor Immunotherapy
April 15, 2026
Clicks:152
In the field of tumor immunotherapy, defective antigen presentation has remained a critical bottleneck limiting the efficacy of T cell-based immunotherapies. Tumor cells frequently exhibit insufficient MHC-I molecule-mediated antigen presentation, resulting in low antigenicity and susceptibility to antigen escape, thereby preventing cytotoxic T lymphocytes from precisely recognizing and attacking malignant cells. Recently, an innovative study published in Advanced Materials reported a cascade-responsive peptide self-assembly-guided tumor-targeting co-delivery system for T cell antigens and camptothecin (CPT), successfully reshaping tumor antigenicity and providing a novel strategy for cancer immunotherapy—with Absin's high-quality products serving as crucial support for this breakthrough research.
Journal: Advanced Materials (IF 26.8) | DOI: https://doi.org/10.1002/adma.202516767
Absin Products Used: Rabbit anti-CD274 Polyclonal Antibody (abs115675), Antifade Mounting Medium (with DAPI) (abs9235)


I. Core Research Strategy: Precision Targeting + Dual Enhancement to Overcome Antigen Presentation Defects
Addressing the central challenge of defective tumor antigen presentation, the research team designed a three-dimensional "cascade-responsive + targeted delivery + synergistic therapy" strategy:
- Cascade-Responsive Peptide Carrier Design: Two self-assembling peptide carriers, PSA (Nap-GFFpYE-ss-Ag) and CPSA (CPT-GFFpYE-ss-Ag), were constructed, both containing an alkaline phosphatase (ALP)-responsive phosphorylated tetrapeptide (-GFFpY-) and a glutathione (GSH)-cleavable disulfide bond (-ss-), enabling precise regulation through "extracellular ALP-mediated dephosphorylation triggering assembly → intracellular GSH-triggered cargo release."
- Dual Active Component Delivery: PSA was loaded with the OVA-derived antigenic epitope OVA₂₅₇₋₂₆₄, while CPSA incorporated the topoisomerase I inhibitor CPT in addition to the antigen, achieving both exogenous antigen targeted delivery and CPT-mediated enhancement of MHC-I molecule expression for dual improvement of tumor antigen presentation efficiency.
- Combination Therapy Enhancement: Combined with adoptive OT-I CD8⁺ T cell infusion, and leveraging CPSA-induced PD-L1 expression, the strategy was further integrated with anti-PD-L1 antibody (aPD-L1) to generate synergistic effects through "enhanced antigen presentation + T cell targeted activation + immune checkpoint blockade" (Figure 1).
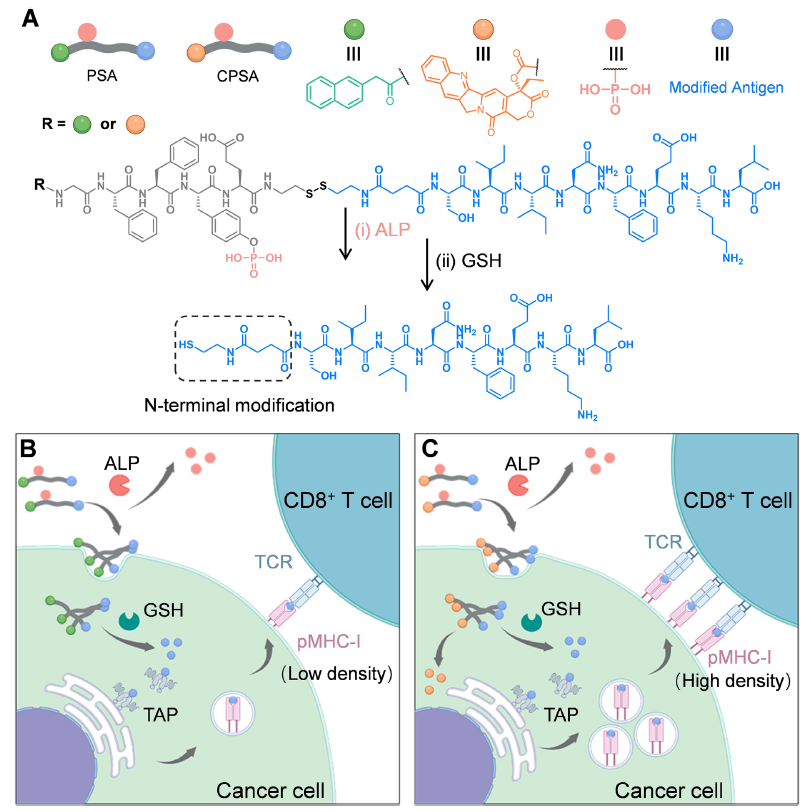
Note: Original Figure 1, illustrating the chemical structures of PSA/CPSA, cascade-responsive delivery process, and antigen presentation mechanism; panels B and C were created with BioRender
II. Key Research Findings: Validation of Potent Antitumor Activity from Cellular to In Vivo Levels
1. Excellent Carrier Performance Enabling Precise Delivery and Antigen Release
- In vitro experiments confirmed that PSA/CPSA underwent ALP-mediated dephosphorylation, triggering structural reorganization to form high-density nanofibers (Figure 2A-D), with 100% dephosphorylation achieved within 60 minutes (Figure 2E); intracellular GSH triggered sustained antigen release, and the N-terminally extended OVA₂₅₇₋₂₆₄ exhibited significantly enhanced binding stability to H-2Kᵇ compared to the native antigen (Figure 2G-H).
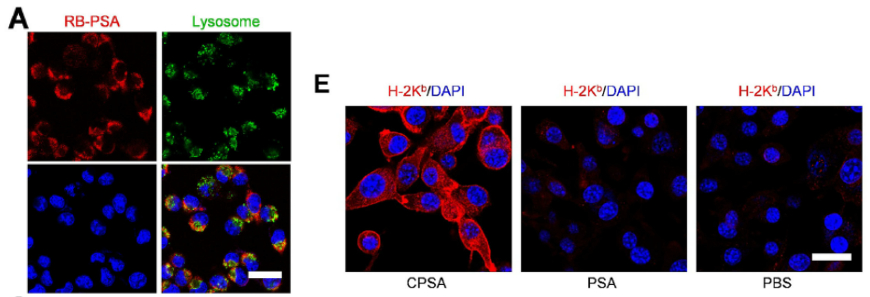
- Cellular uptake experiments demonstrated that PSA entered MC38 cells through dynein-dependent endocytosis, successfully escaped lysosomal degradation, and formed intracellular self-assemblies (Figure 3A-C); antigen presentation efficiency increased progressively over time, reaching 82.3% H-2Kᵇ-antigen complex positivity at 12 hours (Figure 3F).
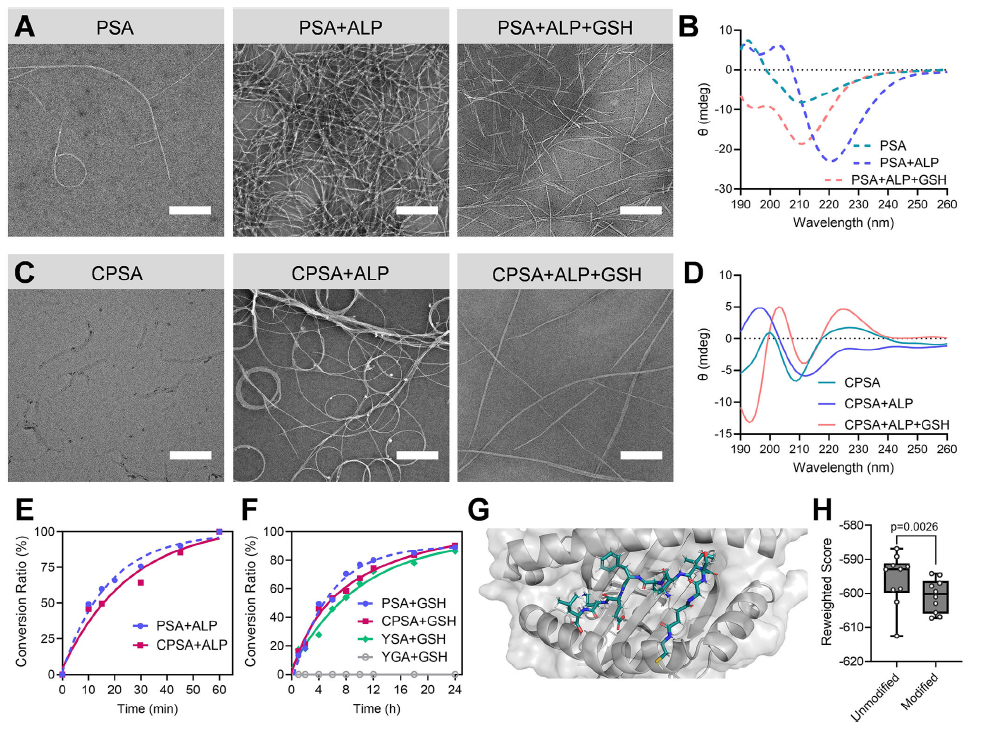
Original Figure 2: A-D show TEM images and CD spectra, E shows dephosphorylation kinetics, F shows antigen release curves, G-H show antigen-H-2Kᵇ binding models
2. Cellular Level: Enhanced T Cell Targeted Cytotoxicity and Cytokine Secretion
- PSA/CPSA-treated MC38 cells efficiently recruited OT-I CD8⁺ T cells, with the cell contact index increased 4.14-fold compared to untreated controls (Figure 3K-L); LDH release assays confirmed significantly enhanced tumor cell killing rates (Figure 3M).
- Following CPSA treatment, MC38 cells exhibited 1.81-fold increased H-2Kᵇ expression and 14.94-fold increased H-2Kᵇ-antigen complex expression (Figure 5A-D), and promoted secretion of effector molecules including IFN-γ, IL-2, and granzyme B by OT-I CD8⁺ T cells (Figure 5G-J).
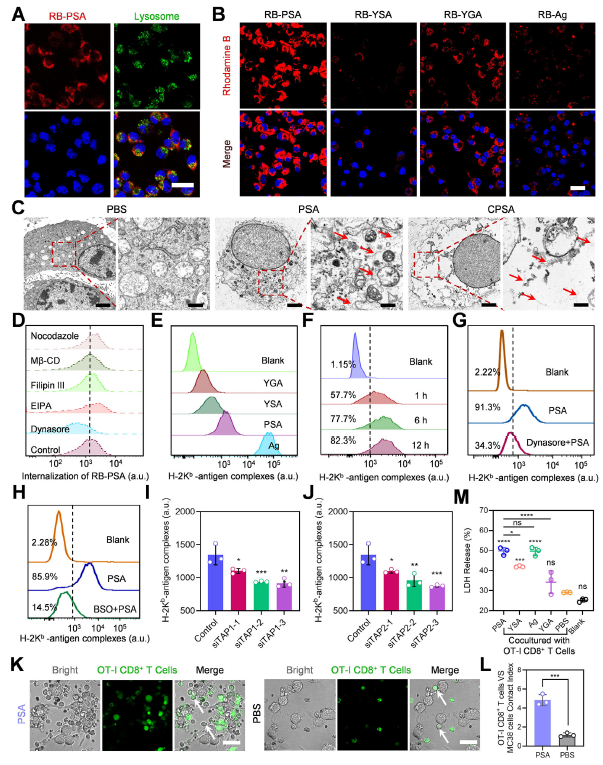
Original Figure 3: A shows RB-PSA cellular localization, B shows cellular uptake of different peptide carriers, E shows antigen presentation levels, K-M show T cell targeted cytotoxicity results
3. In Vivo Level: Significant Tumor Growth Inhibition and Enhanced Immune Infiltration
- In MC38 tumor-bearing mouse models, intravenous PSA injection achieved efficient tumor enrichment with sustained retention for 48 hours, resulting in 63.5% tumor growth inhibition (Figure 4E-F); CPSA combined with aPD-L1 therapy further improved tumor inhibition to 74.5% (Figure 6C).
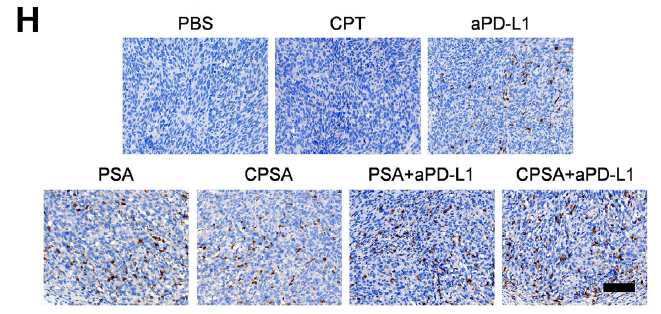
- Immune infiltration analysis revealed significantly increased infiltration of CD3⁺CD8⁺ T cells, granzyme B⁺ CD8⁺ T cells, and other effector cells in tumor tissues from PSA/CPSA-treated groups (Figure 4I-M, Figure 6F-I), confirming effective activation of antitumor immune responses.
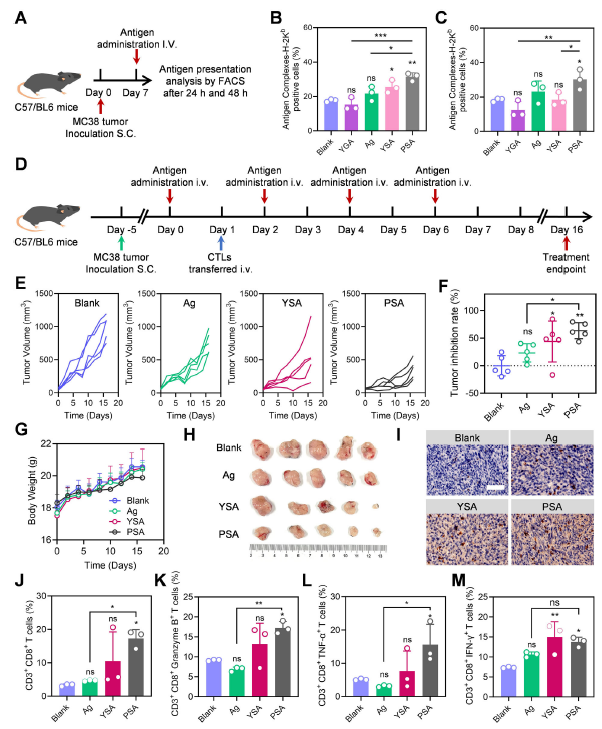
Original Figure 4: B-C show in vivo antigen presentation levels, E-F show tumor growth curves and inhibition rates, I-M show T cell infiltration analysis
III. Absin Product Support: Key Reagents Empowering the Entire Research Process
The successful execution of this study relied on the stable support of Absin's high-quality products, with the rabbit anti-CD274 polyclonal antibody (Cat. No.: abs115675) and antifade mounting medium (Cat. No.: abs9235) playing important roles:
1. Core Products and Application Scenarios
| Absin Product | Cat. No. | Research Application | Corresponding Original Figures |
|---|---|---|---|
| Rabbit anti-CD274 Polyclonal Antibody | abs115675 | Detection of PD-L1 protein expression levels on tumor cell surface; validation of CPSA-mediated PD-L1 upregulation | Figures S25-S27 (Supporting Information) |
| Antifade Mounting Medium | abs9235 | Sample preservation following immunofluorescence staining; prevention of fluorescence signal quenching; ensuring CLSM imaging quality | Figure 3A, Figure 5E, Figure 6H |
2. Core Product Functions
- Rabbit anti-CD274 Polyclonal Antibody: Specifically recognizes mouse PD-L1 (CD274) protein, providing a reliable detection tool for validating the critical mechanism of "CPSA treatment upregulates PD-L1 expression and enhances immune checkpoint blockade sensitivity," ensuring accurate quantitative analysis of PD-L1 expression.
- Antifade Mounting Medium: In cellular immunofluorescence staining (such as H-2Kᵇ molecule localization and T cell-tumor cell interaction observation), effectively maintains fluorescence signal stability, preventing imaging blur or signal loss due to photobleaching, thereby ensuring high-quality CLSM image acquisition and facilitating direct visualization of peptide carrier cellular localization, antigen presentation, and intercellular interactions.
Absin remains committed to providing high-quality reagents for life science research, from specific antibodies to experimental auxiliary reagents, comprehensively meeting diverse research needs and supporting researchers in overcoming technical bottlenecks and accelerating innovative成果转化.
IV. Research Significance and Future Perspectives
Through cascade-responsive peptide self-assembly technology, this study successfully addressed the core problem of insufficient tumor antigen presentation, providing a novel paradigm for the combined application of T cell immunotherapy with chemotherapy and immune checkpoint blockade. Its innovation lies in: achieving dual regulation of exogenous antigen precise delivery and MHC-I expression enhancement for the first time, while simultaneously overcoming the immunosuppressive microenvironment through aPD-L1 combination to significantly enhance antitumor efficacy.
In the future, this strategy is expected to be combined with clinically available antigens (such as neoantigens and public antigens) and extended to CAR-T, TCR-T, and other therapies, providing more efficient precision immunotherapy regimens for human cancer treatment. Absin will continue to deepen its commitment to the research reagent field, supporting more cutting-edge research with superior products and services, and working together with researchers to advance progress in the life sciences!
This article is based on the original publication in Advanced Materials (DOI: 10.1002/adma.202516767); all original figures, data, and intellectual property rights belong to the original journal and research team. Should any infringement occur, please contact us promptly for removal, and we will actively cooperate in addressing such matters.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
