- Cart 0
- English
Unlocking a Novel Immunotherapy Target! Deciphering the TRIM21-PD-1 Axis Mechanism—Molecular Therapy Validates Core Experimental Findings
April 14, 2026
Clicks:78
In the field of tumor immunotherapy, while PD-1/PD-L1 inhibitors and CAR-T therapy have achieved breakthrough progress, drug resistance and low response rates remain critical bottlenecks constraining clinical efficacy. A landmark study recently published in Molecular Therapy has, for the first time, revealed a novel regulatory mechanism whereby TRIM21 stabilizes PD-1 through K63-linked ubiquitination, providing an innovative target for enhancing immunotherapeutic efficacy. Notably, Brefeldin A from Absin (Catalog No.: abs810012), serving as a core experimental reagent, played an indispensable role in intracellular cytokine detection and provided reliable support for validating the research conclusions.
Article Title: Targeting the TRIM21-PD-1 axis potentiates immune checkpoint blockade and CAR-T cell therapy
Journal: Mol Ther. (IF=12)
DOI: https://doi.org/10.1016/j.ymthe.2025.01.047
Absin Product Used: Brefeldin A (Cat. No.: abs810012)

I. Research Strategy: Focusing on the "Hidden Functions" of TRIM21 in Immune Cells
Previous studies have predominantly focused on the role of TRIM21 in tumor cells, while its function in immune cells remains unclear. Given the high expression of TRIM21 in lymphoid organs and the central role of PD-1 as a core molecule in T cell functional inhibition, the research team proposed the central scientific question: Does TRIM21 influence the anti-tumor activity of CD8⁺ T cells by regulating PD-1?
To address this question, the study designed a progressive validation pathway:
- In vivo Experiments: TRIM21 knockout (KO) mice were generated and inoculated with LLC lung carcinoma cells and MC38 colorectal carcinoma cells to observe tumor growth and CD8⁺ T cell functional changes;
- In vitro Mechanisms: Investigation of the regulatory mechanisms of TRIM21 on PD-1 expression (transcriptional vs. post-translational levels), validation of their interaction and ubiquitination modification types;
- Therapeutic Potential: Validation in animal models of the synergistic effects of TRIM21 deficiency on immune checkpoint inhibitors (anti-CTLA-4) and CAR-T therapy.
II. Core Research Findings: The TRIM21-PD-1 Axis as a Novel Breakthrough for Immunotherapy
1. TRIM21 Deficiency Significantly Enhances CD8⁺ T Cell Anti-Tumor Capability
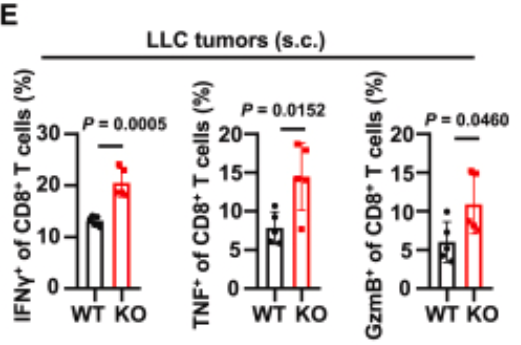
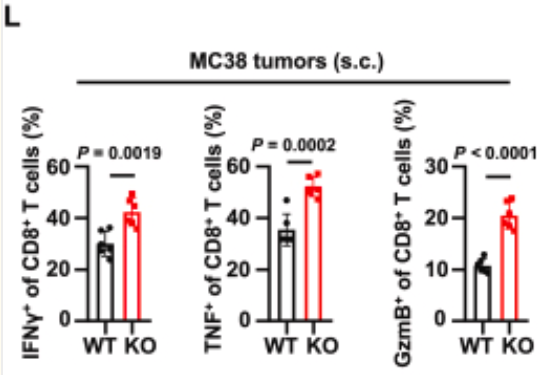
In vivo experiments demonstrated that tumor growth was markedly suppressed in TRIM21 KO mice following tumor cell inoculation (Original Fig. 1A, 1B, 1H, 1I). Flow cytometry analysis confirmed significantly elevated proportions of CD8⁺ T cells in the tumor microenvironment of KO mice, with substantially enhanced capacity to secrete effector cytokines including IFN-γ, TNF, and GzmB (Original Fig. 1E, 1L). Further CD8⁺ T cell depletion experiments indicated that this anti-tumor effect was entirely dependent on CD8⁺ T cells (Original Fig. 1N-S), establishing the inhibitory role of TRIM21 on CD8⁺ T cell function.
Figure 1.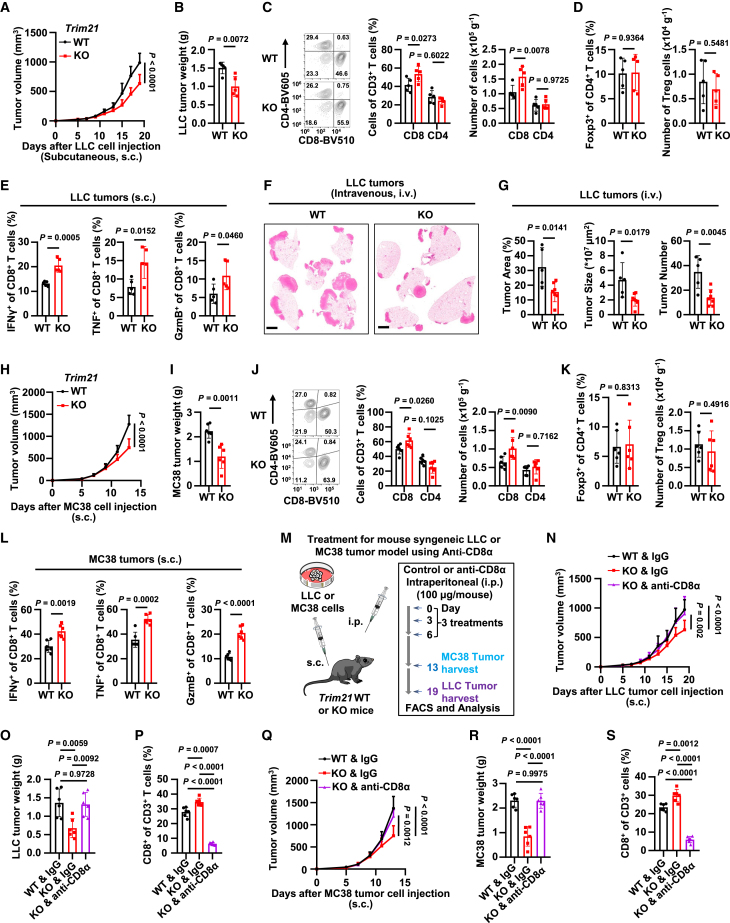
Trim21 KO mice display improved CD8+ T cell anti-tumor immune response
(A and B) Growth curves (A) and terminal tumor weights (B) of subcutaneous LLC tumors in WT or Trim21 KO C57BL/6N mice. n = 5 mice per group. (C) Flow cytometry analysis showing percentages of infiltrating CD8+ and CD4+ T cells among CD3+ T cells in LLC tumors. (D) Percentage of Foxp3+ Treg cells among CD4+ T cells. (E) Percentages of IFN-γ+, TNF+, and GzmB+ cytokine-producing CD8+ T cells in LLC tumors. n = 5 mice per group. (F and G) Representative H&E staining images of lung tissue (F), tumor area, tumor size, or tumor number (G) from WT (n = 5) and Trim21 KO (n = 7) mice following tail vein injection of LLC cells. Scale bars, 2 mm. (H and I) Growth curves (H) and terminal tumor weights (I) of MC38 tumors in WT or Trim21 KO mice. n = 6 mice per group. (J) Analysis of infiltrating CD8+ and CD4+ T cells in MC38 tumors. (K) Analysis of Foxp3+ Treg cells in MC38 tumors. (L) Analysis of cytokine production by CD8+ T cells in MC38 tumors. n = 6 mice per group. (M) Schematic diagram of treatment strategy. (N and O) Growth curves (N) and terminal tumor weights (O) of LLC tumors in WT or Trim21 KO mice treated with control IgG or anti-CD8α. n = 6 mice per group. (P) Flow cytometry analysis of infiltrating CD8+ T cells in LLC tumors across treatment groups. n = 6 mice per group. (Q and R) Growth curves (Q) and terminal weights (R) of MC38 tumors. n = 6 mice per group. (S) Flow cytometry analysis of infiltrating CD8+ T cells in MC38 tumors across treatment groups. n = 6 mice per group.
2. TRIM21 Stabilizes PD-1 Protein Through K63-Linked Ubiquitination
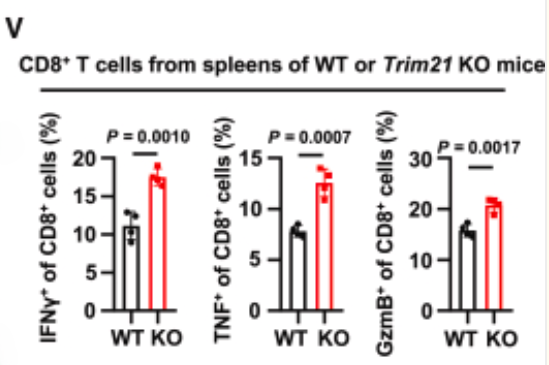
Mechanistic studies revealed that TRIM21 does not affect PD-1 mRNA levels but rather regulates its stability through post-translational modifications. TRIM21 binds to the cytoplasmic domain of PD-1 and catalyzes K63-linked ubiquitination at the K233 residue, thereby antagonizing K48-linked ubiquitination-mediated degradation (Original Fig. 3). Following TRIM21 knockout, the PD-1 protein half-life was shortened, expression of the T cell activation marker CD69 was upregulated, and proliferative capacity was significantly enhanced (Original Fig. 2U, 2S).
Figure 2. 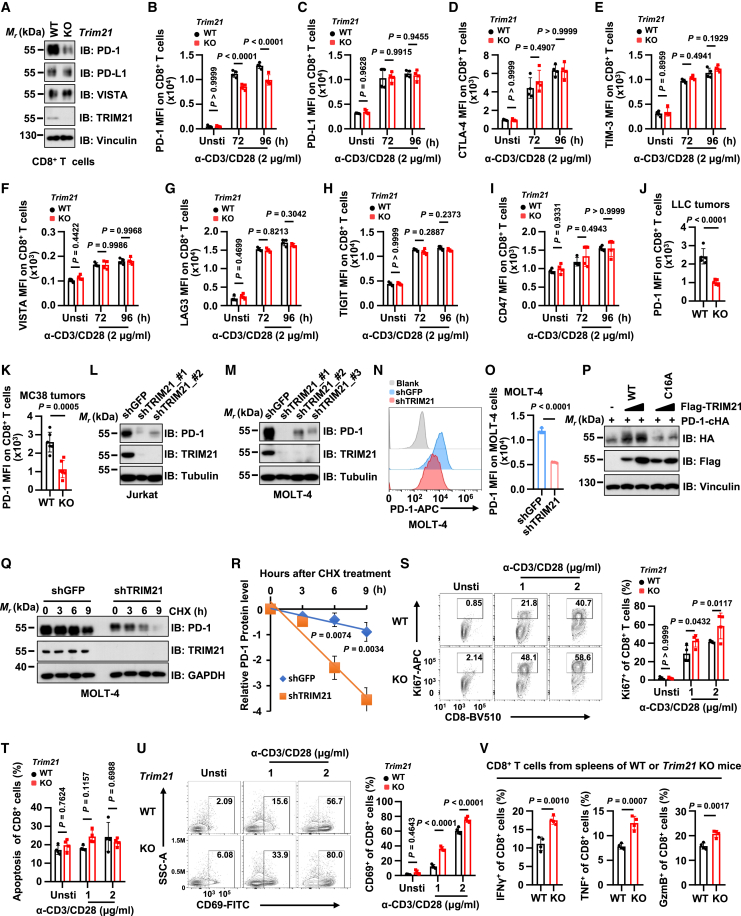
TRIM21 deficiency destabilizes PD-1 and promotes the activation of CD8+ T cells in vitro
(A) Immunoblot (IB) analysis of whole cell lysates from CD8+ T cells of WT or Trim21 KO mice. CD8+ T cells were stimulated with α-CD3/CD28 (2 μg/mL) for 48 h. (B–I) Quantification of mean fluorescence intensity (MFI) for PD-1 (B), PD-L1 (C), CTLA-4 (D), TIM-3 (E), VISTA (F), LAG3 (G), TIGIT (H), and CD47 (I) in splenic CD8+ T cells from WT or Trim21 KO mice. n = 4 mice per group. (J and K) MFI of PD-1 in infiltrating CD8+ T cells from LLC tumors (n = 5) or MC38 tumors (n = 6). (L–O) IB analysis of whole cell lysates from Jurkat or MOLT-4 cells stably expressing shTRIM21 or shGFP (L, M). Flow cytometry analysis (N) and relative MFI quantification (O) of surface PD-1 in MOLT-4 cells. n = 3 biologically independent samples. (P) IB analysis of HEK293T cells transfected with PD-1-cHA and Flag-tagged TRIM21 WT or its catalytic mutant C16A. (Q and R) IB analysis of whole cell lysates from MOLT-4 cells stably expressing shTRIM21 or shGFP (Q). Quantification of PD-1 band intensity at different time points following treatment with 200 μg/mL cycloheximide (CHX) (R). (S) Ki67 expression in splenic WT or Trim21 KO CD8+ T cells. n = 4 per group. (T) Apoptosis analysis of splenic CD8+ T cells. n = 4 mice per group. (U) CD69 expression in splenic WT or Trim21 KO CD8+ T cells. n = 4 per group. (V) Analysis of IFN-γ+, TNF+, and GzmB+ in splenic CD8+ T cells. n = 4 mice per group.
Figure 3.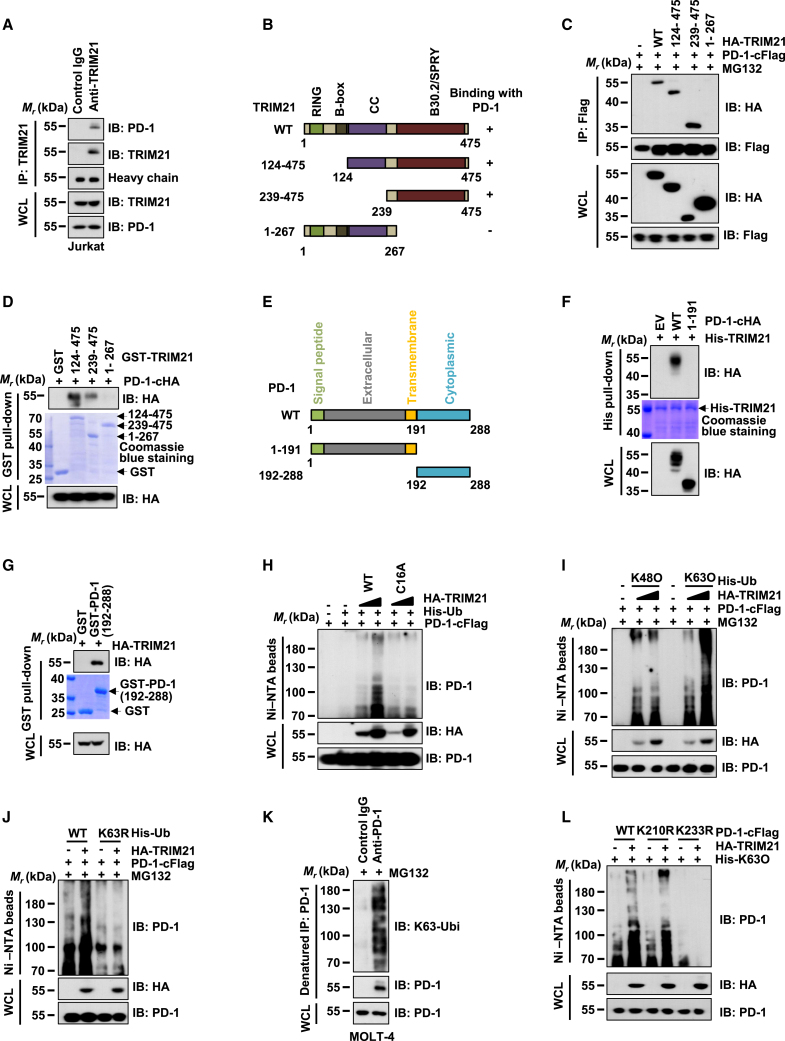
TRIM21 interacts with PD-1 and promotes its K63-linked ubiquitination
(A) IB analysis of whole cell lysates (WCLs) and anti-TRIM21 immunoprecipitates (IPs) from Jurkat cells. IgG was used as a negative control. (B and E) Schematic diagrams of TRIM21 (B) and PD-1 (E) protein sequences. (C) IB analysis of WCLs and anti-Flag IPs from HEK293T cells transfected with PD-1-cFlag and HA-TRIM21 WT or truncation mutants. (D) IB analysis of GST pull-down precipitates from incubation of GST and GST-tagged TRIM21 mutants with WCLs from HEK293T cells transfected with PD-1-cHA. (F) IB analysis of His pull-down products from incubation of His-TRIM21 with WCLs from HEK293T cells transfected with PD-1-cHA or truncation mutants. (G) IB analysis of GST pull-down precipitates from incubation of GST and GST-tagged PD-1 (amino acids 192–288) with WCLs from HEK293T cells transfected with HA-TRIM21. (H) His-tag pull-down assay in guanidine hydrochloride denaturation buffer showing that TRIM21 WT but not C16A mutant promotes PD-1 ubiquitination. (I and J) His-tag pull-down assays showing that TRIM21 promotes K63-linked ubiquitination rather than K48-linked ubiquitination. (K) Detection of endogenous K63-linked ubiquitination of PD-1 in MOLT-4 cells. (L) K233 rather than K210 is the primary modification site for TRIM21-mediated ubiquitination or K63-linked ubiquitination.
3. Targeting TRIM21 Significantly Enhances Immunotherapy Efficacy
- Sensitization to Immune Checkpoint Inhibitors: TRIM21 KO mice receiving anti-CTLA-4 therapy demonstrated tumor control effects far exceeding those of wild-type mice, with further enhanced CD8⁺ T cell infiltration and cytokine secretion (Original Fig. 4);
- Optimization of CAR-T Therapy: TRIM21 KO anti-CD19 CAR-T cells exhibited lower PD-1 expression and stronger tumor-killing activity in LLC-hCD19 and MC38-hCD19 tumor models (Original Fig. 5).
These results indicate that targeting TRIM21—through reducing PD-1 expression and enhancing CD8⁺ T cell function—provides a new direction for combination immunotherapy strategies.
Figure 4.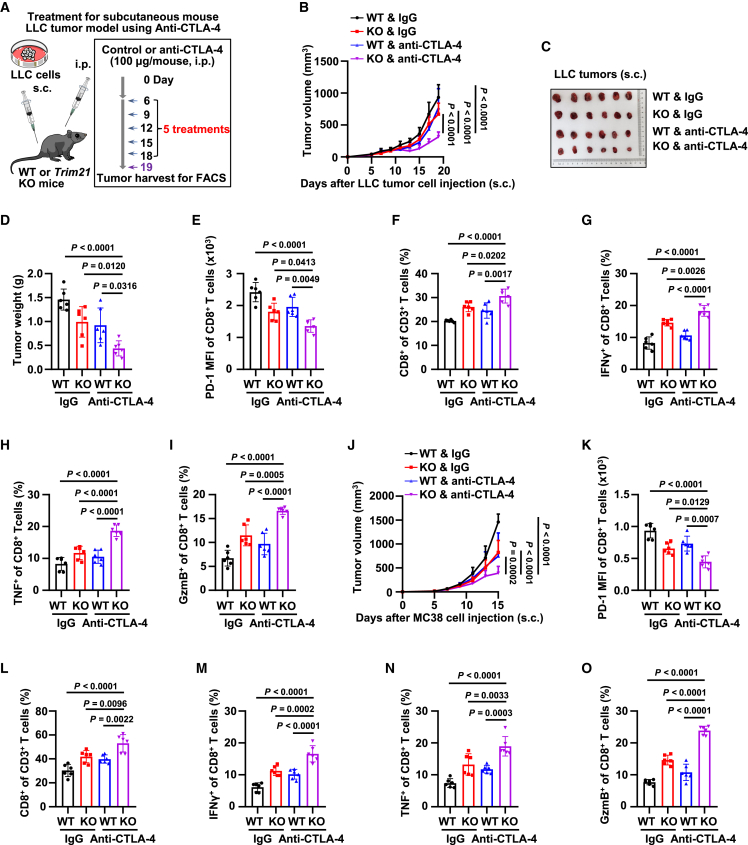
TRIM21 KO sensitizes tumors to anti-CTLA-4 immunotherapy
(A) Schematic diagram of anti-CTLA-4 treatment strategy in mouse LLC tumor model. (B–D) Growth curves (B), terminal tumor images (C), and terminal tumor weights (D) of subcutaneous LLC tumors in WT or Trim21 KO mice. n = 6 mice per group. (E) MFI of PD-1 in infiltrating CD8+ T cells from LLC tumors across treatment groups. n = 6 mice per group. (F) Percentage of infiltrating CD8+ among CD3+ T cells in LLC tumors across treatment groups. n = 6 mice per group. (G–I) Percentages of IFN-γ+ (G), TNF+ (H), and GzmB+ (I) CD8+ T cells in LLC tumors across treatment groups. n = 6 mice per group. (J) Growth curves of MC38 tumors in WT or Trim21 KO mice. n = 6 mice per group. (K) MFI of PD-1 in infiltrating CD8+ T cells from MC38 tumors across treatment groups. n = 6 mice per group. (L) Percentage of infiltrating CD8+ among CD3+ T cells in MC38 tumors across treatment groups. n = 6 mice per group. (M–O) Percentages of IFN-γ+ (M), TNF+ (N), and GzmB+ (O) CD8+ T cells in MC38 tumors across treatment groups. n = 6 mice per group.
Figure 5.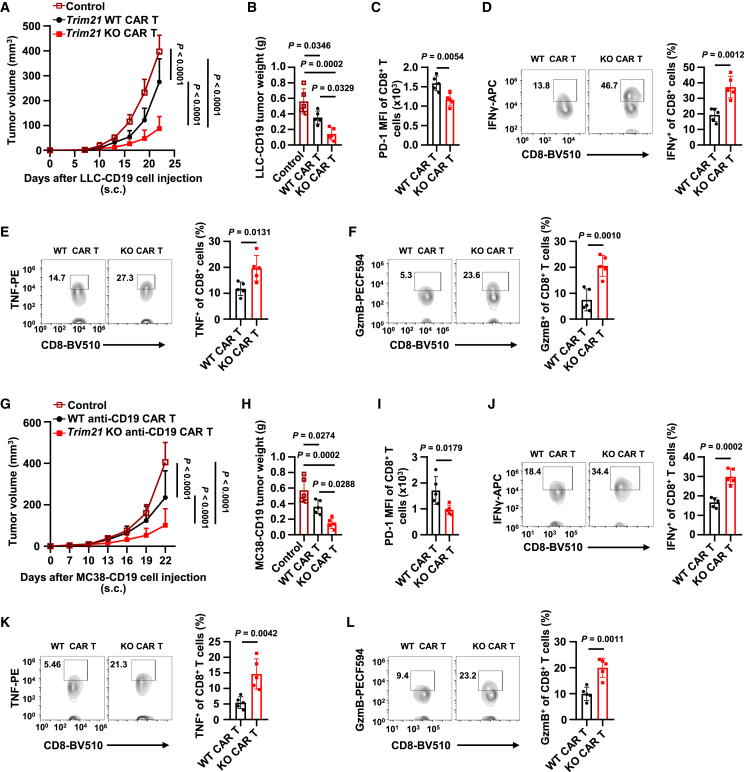
TRIM21 deficiency improves antitumor efficacy of CAR-T cells
(A and B) Growth curves (A) and terminal tumor weights (B) of subcutaneous LLC-hCD19 tumors in immunocompetent mice following adoptive transfer of WT and Trim21 KO anti-CD19 CAR-T cells and T cells (control). n = 5 mice per group. (C) MFI of PD-1 in infiltrating CD8+ CAR-T cells from LLC-hCD19 tumors across treatment groups. n = 5 mice per group. (D–F) Percentages of IFN-γ+ (D), TNF+ (E), and GzmB+ (F) CD8+ CAR-T cells in LLC-hCD19 tumors across treatment groups. n = 5 mice per group. (G and H) Growth curves (G) and terminal tumor weights (H) of subcutaneous MC38-hCD19 tumors in immunocompetent mice following adoptive transfer of WT and Trim21 KO anti-CD19 CAR-T cells. n = 5 mice per group. (I) MFI of PD-1 in infiltrating CD8+ CAR-T cells from MC38-hCD19 tumors across treatment groups. n = 5 mice per group. (J–L) Percentages of IFN-γ+ (J), TNF+ (K), and GzmB+ (L) CD8+ CAR-T cells in MC38-hCD19 tumors across treatment groups. n = 5 mice per group.
III. Critical Role of abs810012: The "Gold Standard" Tool for Intracellular Cytokine Detection
In the core experiments analyzing CD8⁺ T cell function, accurate quantification of intracellular IFN-γ, TNF, and GzmB cytokine expression levels was essential for validating the conclusions. Absin's Brefeldin A (abs810012) served as the critical reagent for achieving this objective.
Product Mechanism of Action
Brefeldin A is a classical Golgi apparatus inhibitor that specifically blocks intracellular protein transport. In the experiments, researchers stimulated CD8⁺ T cells with anti-CD3/CD28 antibodies and then treated them with abs810012 for 5 hours (Original "Mouse primary T cell isolation and stimulation" section), effectively inhibiting cytokine secretion to the extracellular space and allowing their intracellular accumulation, thereby enabling precise detection of cytokine production levels by flow cytometry.
Experimental Applications and Data Support
The use of abs810012 directly supported the validation of two core conclusions:
- In vivo Experiments: Confirmed significantly enhanced secretion capacity of IFN-γ, TNF, and GzmB by tumor-infiltrating CD8⁺ T cells in TRIM21 KO mice (Original Fig. 1E, 1L);
- In vitro Experiments: Validated that activated TRIM21 KO CD8⁺ T cells exhibited significantly elevated intracellular cytokine expression levels (Original Fig. 2V).
Through the reliable Golgi apparatus inhibitory effect of abs810012, the research team was able to precisely quantify the functional status of CD8⁺ T cells, providing direct experimental evidence for the core conclusion that TRIM21 regulates T cell activity.
IV. Product Advantages and Experimental Value
Absin's Brefeldin A (abs810012) features high purity and high specificity, efficiently blocking intracellular protein transport to ensure adequate accumulation of intracellular cytokines. In this study, the product successfully supported precise detection of intracellular cytokines by flow cytometry. Its stable performance ensured the reliability and reproducibility of experimental data, establishing it as a critical tool connecting mechanistic investigation with functional validation.
V. Summary and Perspectives
This study elucidated for the first time the molecular mechanism by which TRIM21 stabilizes PD-1 through K63-linked ubiquitination, confirming that targeting TRIM21 enhances the anti-tumor activity of CD8⁺ T cells and providing novel strategies for combination therapy with immune checkpoint inhibitors and optimization of CAR-T cell therapy. As a core experimental tool, Absin abs810012 demonstrated excellent performance in intracellular cytokine detection, facilitating the efficient completion of functional validation experiments by the research team and fully exemplifying the critical supporting role of high-quality reagents in life science research.
In the future, the development of TRIM21-targeted inhibitors or TRIM21-knockout CAR-T cell therapies is expected to become a new direction for overcoming drug resistance in tumor immunotherapy. Absin will continue to deepen its commitment to the life science tools and reagents sector, providing stable and reliable product support for more innovative research, empowering researchers to unlock more mysteries of life sciences.
If you are conducting research related to T cell function, immunotherapy mechanisms, or related fields, abs810012 is the ideal choice for intracellular cytokine detection. Please feel free to contact us for product details or technical support!
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
