- Cart 0
- English
Top Journal STTT Highlight | Cu–DHN Nanozyme Prodrug Triggers Triple-Pathway Cell Death, Absin ELISA Kits Fully Support the New Paradigm of In Situ Cancer Vaccines
April 13, 2026
Clicks:74
Recently, a groundbreaking study was published in the international top-tier journal Signal Transduction and Targeted Therapy — the conductive coordination nanozyme prodrug Cu–DHN, which achieves precise activation through a GSH/H₂O₂ dual-responsive logic gate, simultaneously triggering pyroptosis, cuproptosis, and ferroptosis, transforming the primary tumor into an "in situ vaccine factory" to potently inhibit both primary and metastatic tumors, opening an entirely new avenue for precision tumor immunotherapy. Absin, as a core reagent supplier, safeguarded the critical experiments with highly sensitive and stable ELISA kits, facilitating the successful publication of these achievements in a top-tier journal!
Article Title: Conductive coordination nanozyme prodrugs precisely trigger pyroptosis, cuproptosis and ferroptosis for in situ cancer vaccination
Journal: Signal Transduction and Targeted Therapy (IF=52.7)
DOI:https://doi.org/10.1038/s41392-026-02607-6
Absin Product Used: Mouse TNF-α ELISA Kit (Cat. No.: abs520010)
![]()
I. Research Rationale: Addressing Three Critical Bottlenecks in Pyroptosis Clinical Translation
Pyroptosis represents an ideal approach for inducing in situ tumor vaccines, capable of rapidly releasing tumor-associated antigens (TAAs) and damage-associated molecular patterns (DAMPs) to activate systemic antitumor immunity. However, clinical translation faces three major bottlenecks:
- Inert prodrugs cannot be spatially activated with tumor specificity, leading to systemic toxic side effects;
- Traditional nanozymes exhibit low catalytic efficiency with insufficient reactive oxygen species (ROS) generation, failing to support potent therapeutic efficacy;
- Most tumors exhibit epigenetic silencing of gasdermin D (GSDMD), making pyroptosis initiation difficult and limiting therapeutic efficacy.
To address these challenges, the team innovatively constructed the conductive coordination nanozyme prodrug Cu–DHN:
- ▸ Utilizing a π-conjugated polyphenol backbone to create an "electron highway," achieving efficient electron shuttling throughout the entire nanoparticle volume to maximize peroxidase-like activity;
- ▸ Equipped with a GSH "AND" H₂O₂ dual-responsive logic gate, remaining inert during systemic circulation while achieving precise "ON" activation within the tumor microenvironment;
- ▸ Prodrug on-site conversion to juglone, reversing GSDMD epigenetic silencing + activating the NLRP3 inflammasome, initiating pyroptosis through dual mechanisms, while simultaneously叠加 cuproptosis and ferroptosis to achieve immunogenic cell death (ICD), activating systemic antitumor immunity.
II. Core Research Findings: Triple-Death Synergy for Potent In Situ Vaccine-Mediated Tumor Suppression
1. Structural Design: L-Cys Precise Modulation for Maximized Catalytic Performance
- ● Using L-cysteine as a modulator to precisely control Cu–DHN particle size, with the optimal molar ratio of Cu:DHN:Cys=0.6:1:0.2, yielding ~50 nm particles with 27-fold increased specific surface area and fully exposed catalytic sites;
- ● Formation of mixed Cu⁺/Cu²⁺ valence states and mixed Cu–O, Cu–S coordination shells, dramatically enhancing electron transfer efficiency, achieving efficient •OH generation + DHN-to-juglone conversion (conversion rate >55%). Corresponding figures: Fig. 2b, 2c, 2h, 2j, 3a, 3b
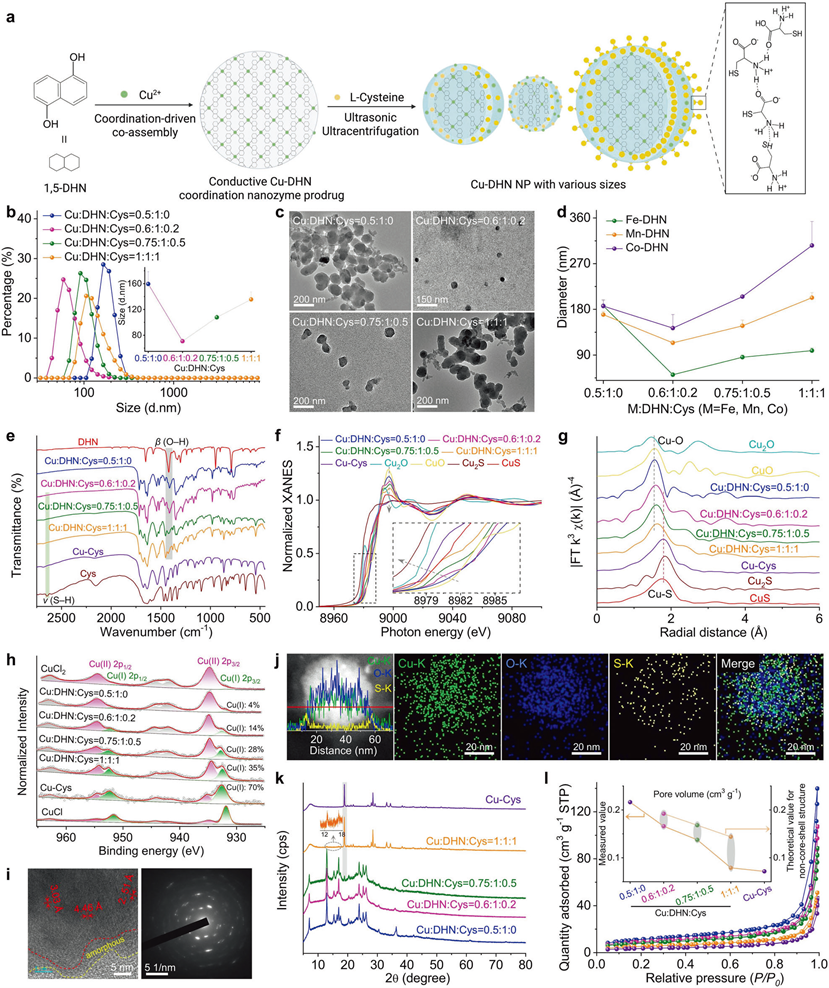
Engineering the architecture and coordination environment
a Synthesis scheme of Cu–DHN, illustrating the role of L-Cys as a coordination terminator and size regulator. The zoomed-in region illustrates the intermolecular hydrogen bonding between L-Cys molecules. b Hydrodynamic size distribution profiles (n = 3), and c TEM images of Cu–DHN with varying Cu:DHN:Cys feed molar ratios, revealing a non-monotonic size trend. d Variation profiles of hydrodynamic size for different M-DHN (M = Fe, Mn, Co) nanoparticles (n = 3) demonstrating the generalizability of Cys-mediated size control. e Fourier transform infrared (FT-IR) spectra confirming the involvement of phenolic –OH and thiol –SH groups; f normalized Cu K-edge XANES spectra, g Fourier transforms of k3-weighted Cu K-edge EXAFS spectra, and h high-resolution Cu 2p XPS spectra of Cu–DHN with varying Cu:DHN:Cys molar ratios, collectively evidencing the evolution of a mixed (O, S)-coordination shell and a rising Cu⁺/Cu2⁺ ratio with increasing Cys content. i HR-TEM image and corresponding SAED pattern, and j elemental distribution mapping and corresponding line-scan profile of Cu–DHN at a Cu:DHN:Cys molar ratio of 0.6:1:0.2. k XRD patterns, and l Brunauer–Emmett–Teller (BET) N2 adsorption–desorption isotherms demonstrating the pore structure evolution of Cu–DHN with varying Cu:DHN:Cys molar ratios, confirming the progressive surface passivation that defines the core–shell pore structure. Data are represented as mean ± SD
2. Mechanistic Breakthrough: Single-Agent Triggering of Triple-Pathway Cell Death, Overcoming GSDMD Low-Expression Constraints
Pyroptosis: Juglone inhibits DNA methyltransferases, reversing GSDMD epigenetic silencing; K⁺ efflux activates the NLRP3 inflammasome, caspase-1 cleaves GSDMD, forming pyroptotic pores;
Cuproptosis: Nanocarrier efficiently delivers Cu⁺, downregulating FDX1 and LIAS to trigger cuproptosis;
Ferroptosis: GSH depletion, GPX4 inhibition, inducing lipid peroxidation;
Triple-pathway synergy enables potent pyroptosis even in GSDMD low-expression tumors, releasing CRT, HMGB1, and ATP to strongly promote dendritic cell (DC) maturation and CD8⁺ T cell infiltration.
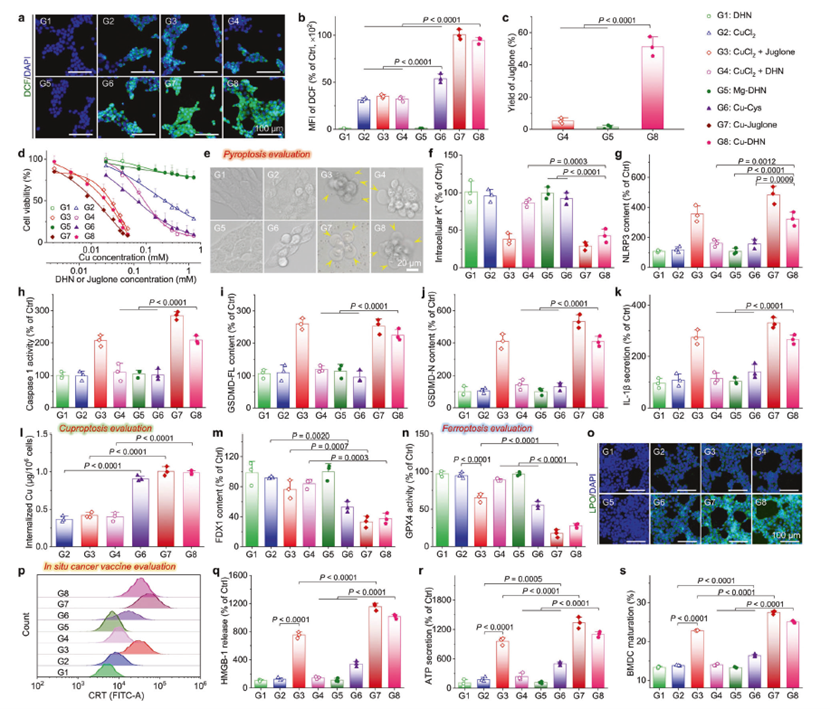
In vitro effects of Cu–DHN (Cu:DHN:Cys = 0.6:1:0.2) on 4T1 cells
a CLSM images and b mean fluorescence intensity (MFI) of 4T1 cells stained by DCFH-DA (ROS probe) after co-incubation with Cu–DHN for 6 h (n = 3). c Juglone yield in 4T1 cells after co-incubation with Cu–DHN for 12 h (n = 3). d Cell viability curves of 4T1 cells after co-incubation with Cu–DHN for 24 h (n = 6). e Bright-field microscopic images of 4T1 cells after co-incubation with Cu–DHN for 18 h (yellow arrows highlight membrane-bound vesicular protrusions indicative of pyroptotic morphology). f Intracellular K+ content in 4T1 cells after co-incubation with Cu–DHN for 6 h (n = 3). g NLRP3 content, h Caspase-1 activity, i GSDMD-FL content, j GSDMD-N content in 4T1 cells after co-incubation with Cu–DHN for 18 h (n = 3). k IL-1β secretion from 4T1 cells after co-incubation with Cu–DHN for 18 h (n = 3). l Internalized amounts of Cu by 4T1 cells after co-incubation with Cu–DHN for 6 h (n = 3). m FDX1 content in 4T1 cells after co-incubation with Cu–DHN for 18 h (n = 3). n GPX4 activity in 4T1 cells after co-incubation with Cu–DHN for 18 h (n = 3). o CLSM images of 4T1 cells stained by Liperfluo (LPO probe) after co-incubation with Cu–DHN for 12 h (n = 3). p Flow cytometry analysis of 4T1 cells labeled by AF488-CRT antibody after co-incubation with Cu–DHN for 18 h. Release of q HMGB-1, and r ATP from 4T1 cells after co-incubation with Cu–DHN for 18 h (n = 3). s BMDC maturation after co-incubation with Cu–DHN treated 4T1 cells for 24 h (n = 3). Cu concentration: 0.020 mM, DHN or juglone concentration: 0.033 mM. Data are represented as mean ± SD
3. In Vivo Efficacy: Primary Tumor Suppression, Metastasis Control, and Pristine Safety
- ✓ Orthotopic 4T1 Breast Cancer Model: 75% tumor growth inhibition rate with significantly extended survival;
- ✓ Bilateral Tumor Model: Triggering abscopal effects to suppress contralateral tumor growth;
- ✓ Lung Metastasis Model: >90% reduction in lung metastatic nodules with substantially improved survival rates;
- ✓ Safety Profile: No activation in normal tissues, normal hepatic/renal function and platelet counts, significantly outperforming direct juglone treatment.
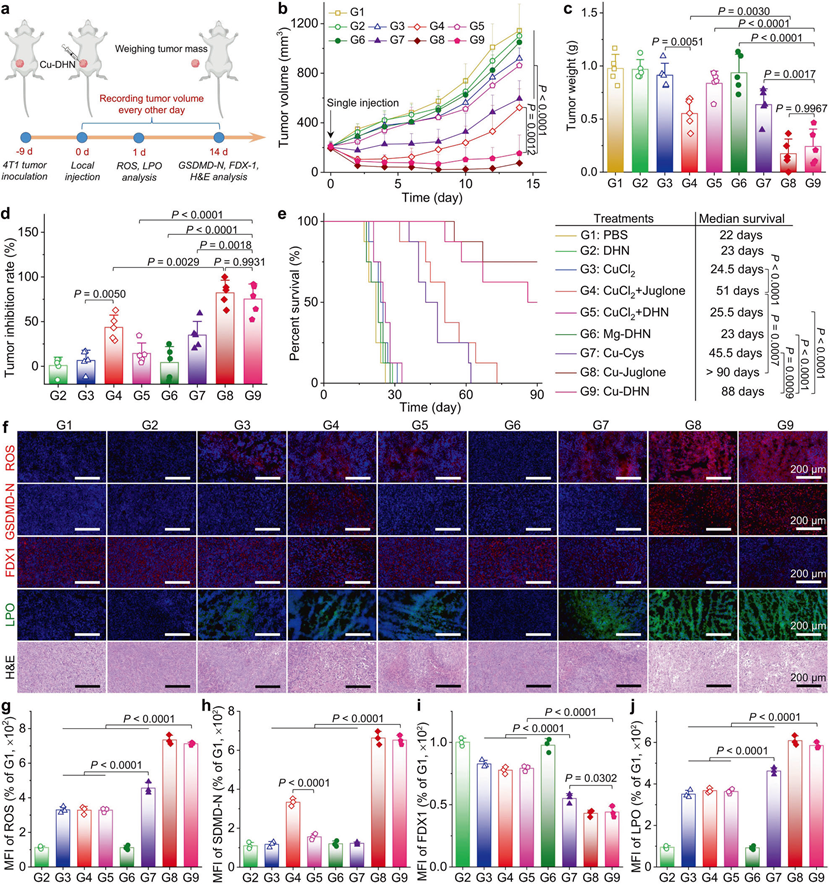
In vivo effects of Cu–DHN (Cu:DHN:Cys = 0.6:1:0.2) on 4T1 tumor
a Schematic illustration of inhibiting tumor growth via inducing pyroptosis, cuproptosis, and ferroptosis by a single intratumoral injection of Cu–DHN. b Longitudinal monitoring of tumor growth following single-dose Cu–DHN intratumoral administration (3.78 μmol kg−1 Cu, 6.24 μmol kg−1 DHN or juglone) (n = 5). c Weights of dissected tumors, and d tumor inhibition rate at 14 days post-treatment (n = 5). e Survival rate analysis of 4T1 tumor-bearing BALB/c mice treated with Cu–DHN (n = 8). f CLSM images and MFI of tumor slices labeled with g DHE (1 day post-treatment), h GSDMD-N antibody (14 days post-treatment), i FDX1 antibody (14 days post-treatment), j Liperfluo (1 day post-treatment), and H&E (14 days post-treatment) (n = 3). Data are represented as mean ± SD
III. Absin's Robust Support: Critical Reagents Empowering Top-Tier Publication Data
In this study, the Absin Mouse Tumor Necrosis Factor-α (TNF-α) ELISA Kit (abs520010-96T) served as the core tool for immune activation validation, playing an irreplaceable role in three critical experiments: systemic immune response, abscopal effects, and anti-metastasis efficacy:
Product Information
Core Functions
1. Precise Quantification of Serum/Tumor Tissue TNF-α
Detecting cytokine storm levels post Cu–DHN treatment to validate immune activation intensity and controllability;
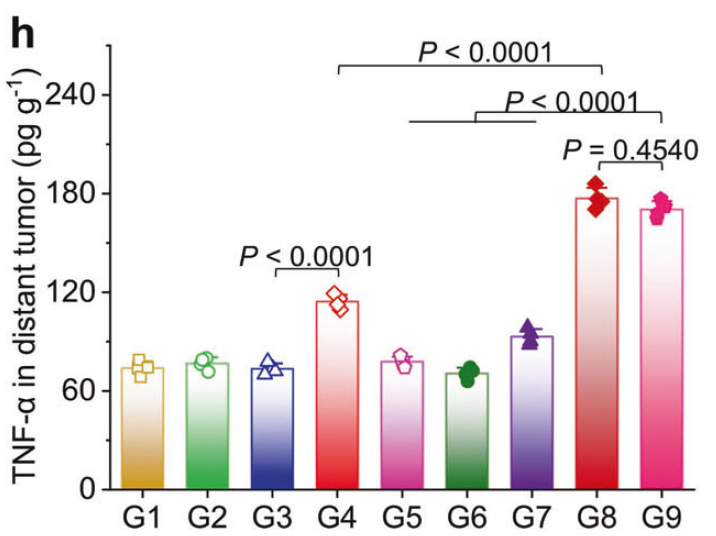
h TNF-α and i IFN-γ contents in distant tumors (n = 4).
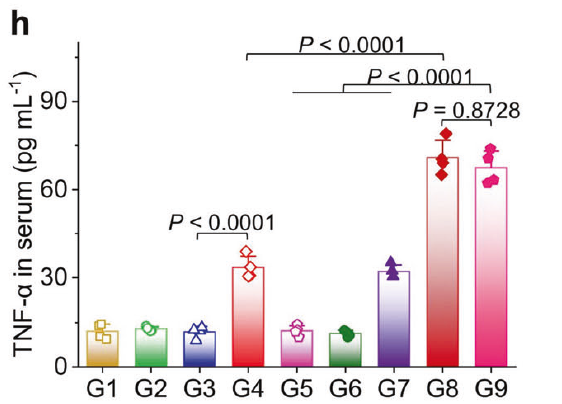
h TNF-α and (i) IFN-γ contents in serum from mice at 3 days post-treatment (n = 4).
2. High Sensitivity, Low Background
Adapted for micro-sample detection, accurately reflecting DC maturation and CD8⁺ T cell activation to support immunological mechanism conclusions;
3. Stable and Reliable
Minimal inter-batch variation and strong reproducibility, ensuring authentic and reproducible in vivo efficacy data to support the rigor of top-tier publication data.
The Cu–DHN conductive coordination nanozyme prodrug constructed in this study achieves triple breakthroughs: tumor-specific activation + triple-death synergy + in situ vaccine, addressing the three major pain points of pyroptosis-based therapy (targeting, efficiency, and safety), providing a novel design paradigm for nanozyme-catalyzed immunotherapy. Absin deeply empowers cutting-edge research with high-quality reagents and will continue to launch premium products in tumor immunology, regulated cell death, and nanomaterials to facilitate more Chinese research achievements reaching international top-tier journals!
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
