- Cart 0
- English
Breaking Through Diabetic Wound Healing Barriers: What Makes This Smart Hydrogel Achieve Near-Complete Wound Closure in Just 14 Days?
April 13, 2026
Clicks:75
Diabetic wounds, characterized by hyperglycemia, persistent oxidative stress, and dysregulated inflammation, often fall into a "chronic non-healing" predicament, representing a significant global clinical challenge. Recently, a research team from the PLA General Hospital and Beijing Institute of Technology published a groundbreaking study in Bioactive Materials, developing a glucose/ROS dual-responsive, redox-gated adaptive hydrogel HPSL@SG. Through synergistic inhibition of the cGAS/STING pathway combined with oxidative stress alleviation, this hydrogel achieves efficient diabetic wound repair, with a healing rate of 89.7% at day 10 and near-complete wound closure by day 14.
As the core reagent provider for this study, Absin's β-Tubulin antibody and GAPDH antibody provided comprehensive support throughout the experiments, ensuring precision and reliability in protein detection.
Article Title: Glucose/ROS-responsive and redox-gated adaptive hydrogel dressing for accelerating diabetic wound repair via synergistic cGAS/STING pathway inhibition and oxidative stress alleviation
Journal: Bioactive Materials (IF=20.3)
DOI:https://doi.org/10.1016/j.bioactmat.2026.03.025
Absin Products Used: β-Tubulin and GAPDH-specific antibodies

I. Core Research Strategy: Targeting the Diabetic Wound "Vicious Cycle"
Core Mechanism of Impaired Diabetic Wound Healing:
Persistent hyperglycemia → Mitochondrial oxidative stress → mtDNA leakage → Activation of macrophage cGAS/STING pathway → Massive pro-inflammatory cytokine release → Inflammatory phase arrest and healing obstruction.
The research team designed a targeted dual-functional synergistic strategy to precisely disrupt this pathological loop:
- Construction of a glucose/ROS-responsive hydrogel enabling intelligent drug release in pathological microenvironments, preventing drug wastage;
- Loading with macrophage-targeting liposomes HPSL for precise delivery of STING inhibitor H151, minimizing off-target immune dysregulation;
- Integration of chlorogenic acid (CGA) for potent ROS scavenging, creating a dual-action approach to remodel the wound healing microenvironment.
II. Core Materials and Absin Critical Reagent Support
1. Hydrogel System Construction (SG/HPSL@SG)
- Matrix: Chlorogenic acid-grafted gelatin (Gel-e-CGA) + 3-aminophenylboronic acid-modified sodium alginate (SA-PBA), crosslinked via dynamic boronate ester bonds, conferring injectability, self-healing, stretchability, and shape adaptability—four core characteristics.
- Payload: Macrophage-targeting liposomes HPSL (encapsulating H151 + phosphatidylserine PS), achieving active macrophage targeting and precise action on core inflammatory cells.
2. Absin Reagents and Applications

![]()
(F) STING/p-STING, (G) TBK1/p-TBK1, (H) IRF3/p-IRF3, (I) NF-κB, (J) TNF-α, and (K) IL-6, Lane 1: Normal group, Lane 2: Model group, Lane 3: PSL group, Lane 4: Free H151 group, Lane 5: HPSL group. n = 3. All data are shown as mean ± SEM.
III. Key Research Findings
1. Excellent Hydrogel Performance Meeting Clinical Requirements
- Dynamic boronate ester bonds confer self-healing, injectability, and high adhesion properties, enabling direct injection into wounds with adaptability to irregular wound geometries and joint movement areas, aligning with clinical application scenarios;
- High porosity (~91.4%) and excellent water retention maintain persistent moist wound microenvironments, providing optimal conditions for cell proliferation and tissue regeneration.
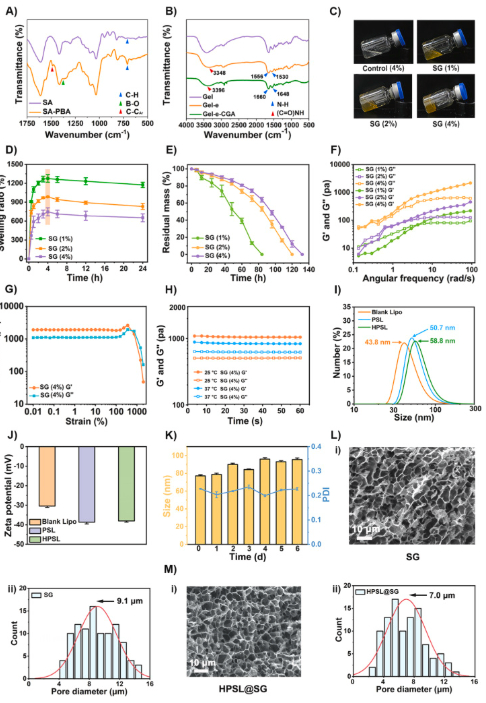
Figure 1: Preparation and characterization of HPSL@SG hydrogel
2. Dual-Responsive Intelligent Drug Release: On-Demand Delivery Without Waste (Fig. 2J-L)
- Boronate ester bonds undergo cleavage exclusively under diabetic wound-specific "high glucose + high ROS" pathological conditions, enabling rapid hydrogel degradation;
- Cumulative release of STING inhibitor H151 reaches 44.6% at 24 hours, effectively preventing burst release-associated side effects and drug wastage.
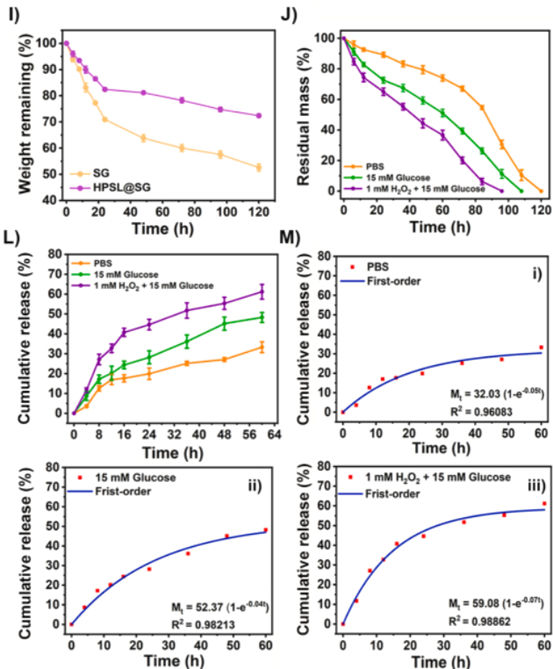
Figure 2J-L: Drug release profiles of hydrogel under high glucose/ROS conditions
3. Potent Antioxidant Activity Resolving Oxidative Stress Challenges
- Chlorogenic acid (CGA) within the hydrogel directly scavenges ROS, with experimental confirmation of 79.9% reduction in superoxide anion levels;
- Utilizing DHE probe, fluorescence intensity changes visually demonstrate oxidative stress alleviation at the wound site, providing robust support for experimental conclusions.
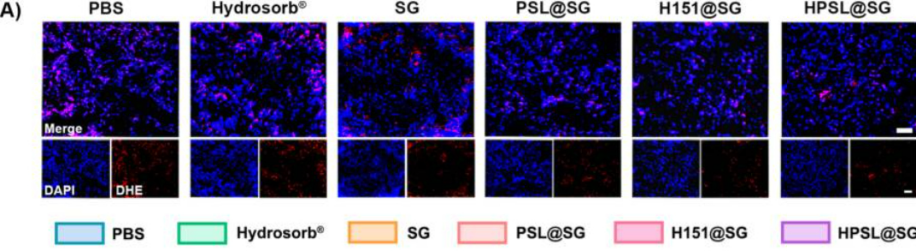
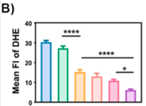
Figure 6A-B: DHE probe detection results; left: control group, right: experimental group; reduced fluorescence indicates oxidative stress alleviation
4. Precise Targeting + Anti-Inflammatory Remodeling: Restoring Healing Pathways
- Phosphatidylserine (PS) mediates HPSL macrophage targeting, achieving rapid cellular uptake within 1 hour with precise action on inflammatory core cells;
- Effective cGAS/STING pathway inhibition significantly reduces p-TBK1, p-IRF3, and NF-κB protein expression, markedly downregulating pro-inflammatory cytokines TNF-α/IL-6 while upregulating anti-inflammatory cytokine IL-10, remodeling the wound inflammatory microenvironment;
- Absin β-Tubulin and GAPDH loading control antibodies ensure Western Blot reliability, clearly demonstrating pathway inhibition and cytokine expression changes.
5. In Vivo Wound Repair Breakthrough with Significant Clinical Potential (Fig. 5)
- Animal experiments demonstrate 89.7% wound closure rate at day 10 in the experimental group, with near-complete healing by day 14, significantly outperforming commercial dressing HydroSorb®;
- Wound collagen deposition increased from 32.6% to 69.3%, while angiogenesis markers CD31 and VEGF increased 6.6-fold and 7.3-fold respectively, achieving "rapid healing + high-quality repair".
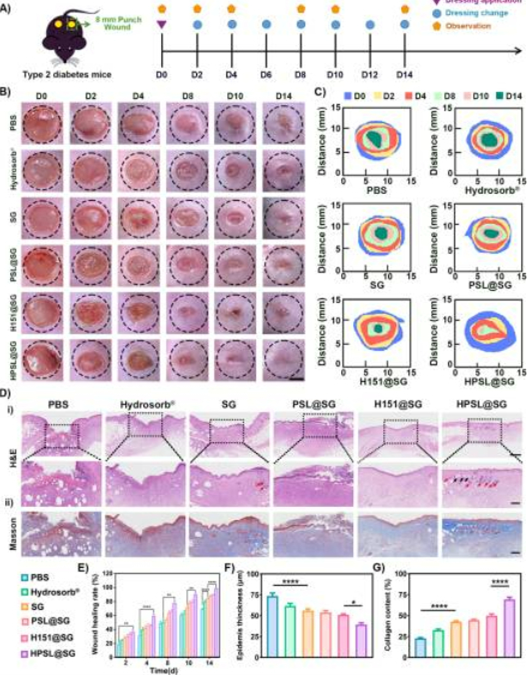
Figure 5: In vivo wound healing efficacy and histological examination
IV. Research Significance and Future Perspectives
The HPSL@SG hydrogel developed in this study innovatively achieves "intelligent responsiveness + precise targeting + dual-pathway synergy", breaking the "vicious cycle" of impaired diabetic wound healing and providing a novel therapeutic strategy for diabetic wound treatment with substantial clinical translation potential.
Future development may integrate antimicrobial components to address infected diabetic wounds, expanding application scenarios. Absin remains committed to advancing research reagent development, providing robust support for innovative research and clinical translation, empowering researchers to overcome technical bottlenecks and translate more scientific achievements into clinical applications.
[Disclaimer] This article is based on the original publication in Bioactive Materials (DOI:10.1016/j.bioactmat.2026.03.025), interpreted and compiled by AI. All original figures, data, and intellectual property rights belong to the original journal and research team. If any infringement occurs, please contact us promptly for removal, and we will actively cooperate in handling such matters.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
