- Cart 0
- English
Breaking Through mRNA Delivery Bottlenecks: Fluorinated Lipid Nanoparticles (FLNPs) Enable Real-Time Tracking and Mechanistic Elucidation of Immune Activation — Absin mRNA Powers PNAS Top-Tier Research
April 13, 2026
Clicks:88
mRNA vaccines have emerged as a pivotal platform for infectious disease prevention and cancer immunotherapy, owing to their rapid development cycle and robust immunogenicity. However, conventional lipid nanoparticles (LNPs) face critical limitations including hepatic sequestration, suboptimal targeting efficiency, and the inability to monitor in vivo distribution in real-time, significantly compromising vaccine precision and biosafety.
Recently, a groundbreaking study published in the prestigious journal PNAS reported the development of fluorinated lipid nanoparticles (FLNPs), enabling real-time tracking of mRNA delivery via 19F NMR/MRI imaging. This innovation elucidates the spatiotemporal mechanisms of immune activation while substantially reducing hepatic accumulation. Notably, high-quality mRNA raw materials from Absin provided essential support for this breakthrough research, facilitating the translation of scientific findings!
Article Title: Fluorinated lipid nanoparticles enable real-time tracking of mRNA delivery and uncover spatiotemporal mechanisms of immune activation
Journal: PNAS (IF=9.1)
DOI: https://doi.org/10.1073/pnas.2519823123
Absin Products Used: EGFP mRNA (N1-Methylpseudo-UTP) (Cat. No.: abs60176-A), Fluc mRNA (N1-Methylpseudo-UTP) (Cat. No.: abs60178-A)

I. Critical Challenges and Innovative Strategies
1. Three Critical Limitations of Conventional LNPs
- Predominant hepatic tropism resulting in low transfection efficiency at target tissues;
- Absence of non-invasive real-time tracking modalities, precluding monitoring of in vivo fate;
- Unclear spatiotemporal correlation between mRNA delivery, expression, and immune activation, leading to empirical formulation optimization.
2. Core Innovative Rationale
Through fluorinated lipid modification of LNPs, two core capabilities were conferred:
- Enhanced Delivery: Improved serum stability, cellular uptake, and endosomal escape, ensuring complete protection of nucleic acids from degradation throughout the delivery process;
- Real-time Tracking: Via 19F NMR/MRI signal variations, precise reflection of nanoparticle disassembly and nucleic acid release, establishing quantitative correlations among signal intensity → delivery efficiency → protein expression.
Ultimately achieving the trinity of precision delivery, non-invasive tracking, and mechanistic elucidation, providing a novel platform for rational design of next-generation mRNA vaccines.
II. Key Research Findings
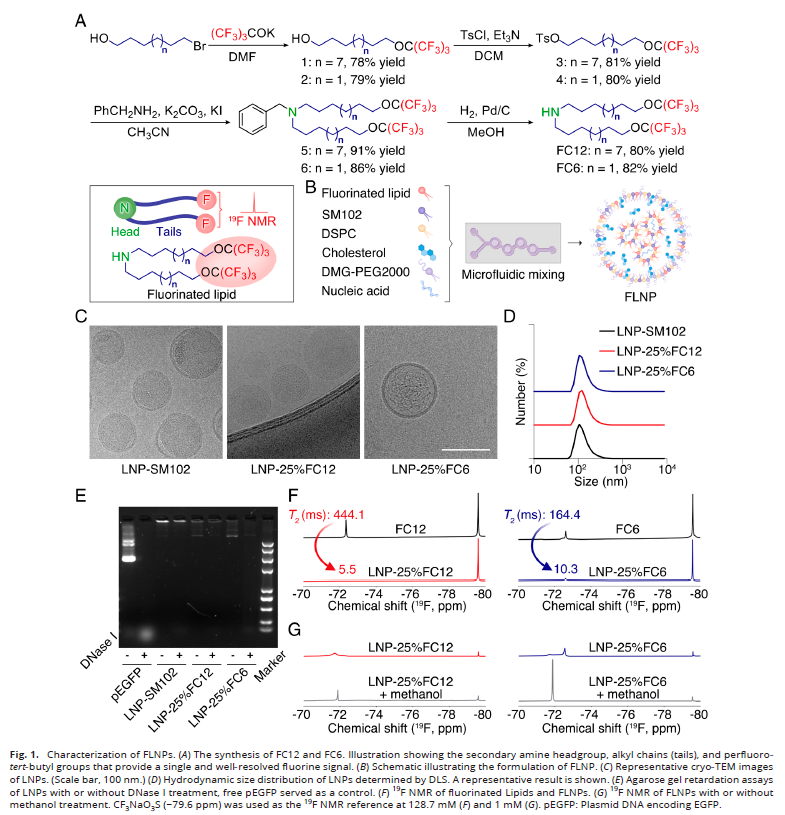
1. FLNP Design and Physicochemical Characterization (Fig. 1)
- Two fluorinated lipids, FC12 and FC6, were synthesized to replace the clinical LNP core lipid SM102;
- Cryo-electron microscopy revealed spherical multilamellar structures with particle diameters of approximately 100 nm, consistent with conventional LNP morphology;
- Encapsulation efficiency exceeded 79%, providing effective protection against nuclease degradation with excellent stability.
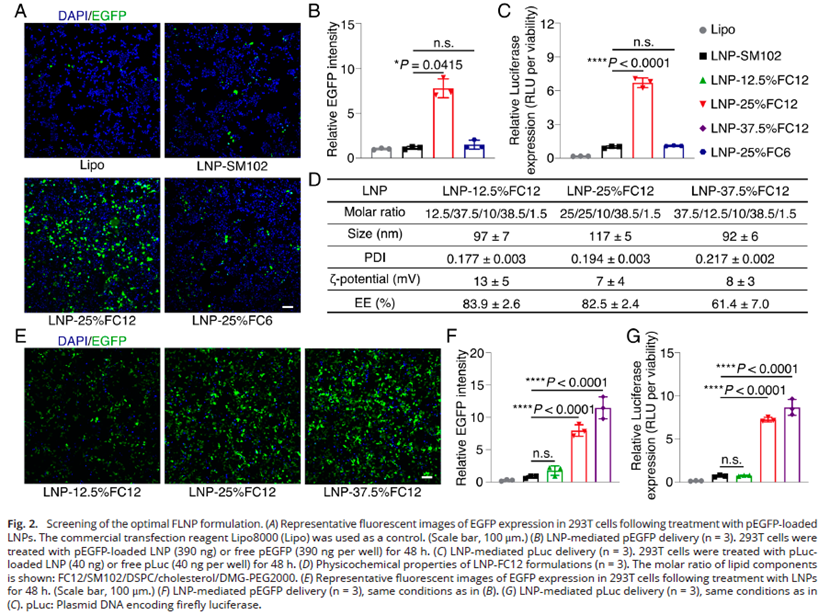
2. Significantly Enhanced In Vitro Delivery Efficiency (Fig. 2)
- The LNP-25% FC12 group demonstrated 7.1-fold enhancement in EGFP expression and 6.8-fold enhancement in luciferase expression compared to clinical LNPs;
- Cell viability remained >80% across all formulations, indicating negligible cytotoxicity;
- Molecular simulation confirmed that FC12 enhances membrane rigidity and fluidity, balancing stability with release efficiency.
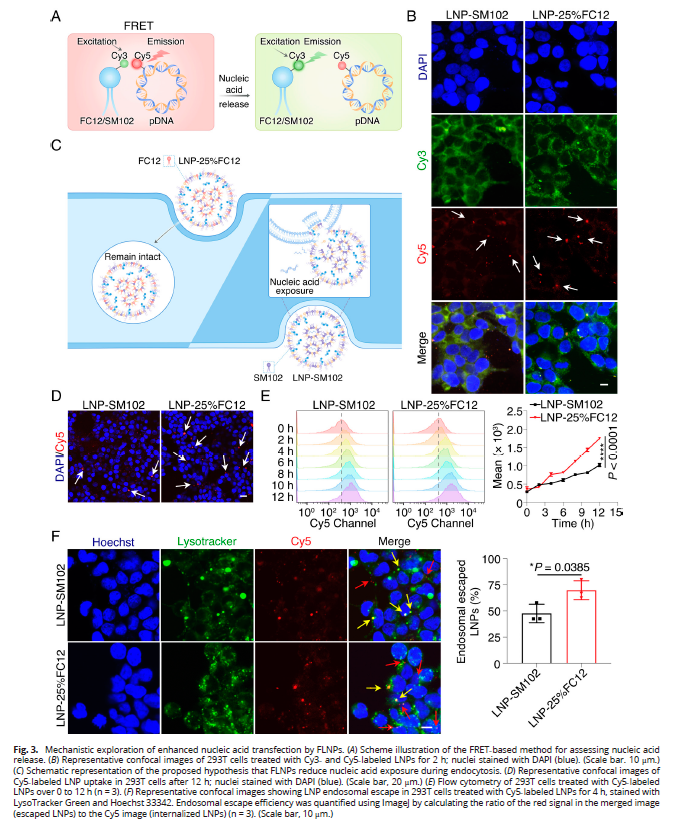
3. In-depth Mechanistic Analysis of Delivery (Fig. 3)
- FRET assays confirmed electrostatic interactions between lipids and nucleic acids, with signal disappearance upon release;
- LNP-25% FC12 exhibited 1.7-fold enhanced cellular uptake and 1.5-fold improved endosomal escape efficiency;
- Reduced nucleic acid exposure during endocytosis, preventing degradation and enhancing transfection efficiency.
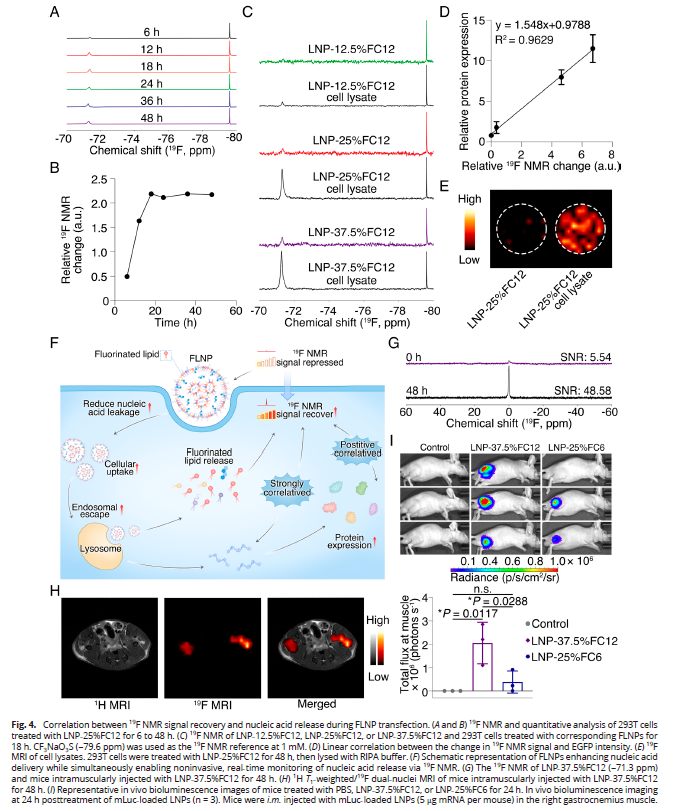
4. Quantitative Correlation Between 19F Signal and Delivery Efficiency (Fig. 4)
- 19F NMR signal recovery over time demonstrated positive correlation with nucleic acid release and protein expression (R2=0.9629);
- Signal recovery serves as an early predictive biomarker for non-invasive assessment of delivery efficacy;
- In vivo MRI clearly visualized injection site signals, enabling real-time tracking in living organisms.
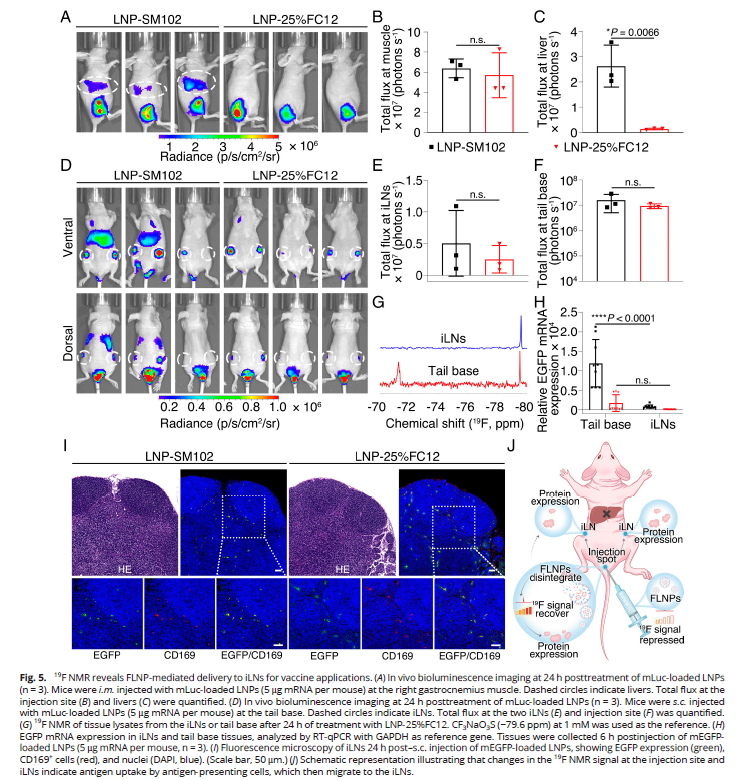
5. In Vivo mRNA Delivery and Immunological Mechanisms (Fig. 5)
- FLNPs achieved efficient expression at injection sites with 94.6% reduction in hepatic accumulation, substantially improving biosafety;
- Antigen-presenting cells (APCs) captured antigens at injection sites and migrated to draining lymph nodes, initiating localized immune responses;
- Only expressed proteins migrated to lymph nodes, with minimal systemic dissemination of nanoparticles and mRNA, resulting in negligible systemic exposure.
III. Robust Support from Absin Products Empowering High-Impact Research
In this study, high-quality mRNA raw materials provided by Absin served as critical reagents ensuring experimental success. Specific applications and contributions are detailed below:
IV. Research Significance and Translational Prospects
- Technological Breakthrough: FLNPs integrate efficient delivery with non-invasive tracking; 19F signals enable quantitative assessment of delivery efficacy, addressing the challenge of in vivo LNP monitoring;
- Mechanistic Innovation: First demonstration of the core immunological pathway involving local mRNA expression → APC uptake → lymph node migration, providing theoretical foundations for vaccine design;
- Clinical Value: Dramatically reduced hepatic accumulation enhances biosafety, applicable to precision delivery scenarios including infectious disease vaccines and cancer vaccines.
Conclusion
From LNPs to FLNPs, mRNA delivery technology has achieved transformative advancement. Absin continues to empower cutting-edge life science research through high-quality reagents. Moving forward, Absin remains committed to the nucleic acid delivery and vaccine development sectors, introducing more cost-effective products to assist researchers in overcoming technical bottlenecks and advancing mRNA technology from bench to bedside!
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
