- Cart 0The latest additions
0 items in total
Subtotal $0.00
- EnglishChinese
- Cart 0The latest additions
0 items in total
Subtotal $0.00
- EnglishChinese
Immunohistochemistry (IHC) Protocol — Paraffin Sections
March 23, 2026
Clicks:957
Immunohistochemistry (IHC) is a core technique based on specific antigen-antibody binding for qualitative, localization, and semi-quantitative analysis of target proteins in tissue sections, widely used in pathological diagnosis and basic research. According to tissue types, it is mainly divided into paraffin sections and frozen sections.
Experimental Protocol
Specimen Fixation
Cut fresh tissue into small pieces (approximately 1.5 cm × 1.5 cm × 0.3 cm), fix in 4% paraformaldehyde (or formalin) overnight. Generally, fixation time should not exceed 48 hours.
Purpose:
- Prevent specimen detachment from the slide;
- Remove lipids that interfere with antigen-antibody binding, facilitating optimal staining results of antigen-antibody complexes;
- Fixed specimens are easy to preserve.
4% paraformaldehyde is generally selected as the fixative, but for testicular tissue and ocular tissue, Bouin's solution or mDF solution may yield better results.
Dehydration, Paraffin Embedding and Sectioning
Dehydration:
To minimize rapid tissue shrinkage, proceed in ascending concentrations from low to high: 70%, 85%, 95%, to absolute ethanol (anhydrous ethanol), 1 hour for each step, ensuring complete dehydration. Fixed tissue specimens must be cleared of residual fixative and crystalline precipitates, otherwise subsequent staining results may be compromised.
Clearing:
The process of immersing specimens in clearing agent is termed "clearing." Xylene is commonly used as the clearing agent, which serves as a solvent for paraffin. Absolute ethanol is immiscible with paraffin; therefore, a solvent miscible with both ethanol and paraffin is required to replace ethanol in the tissue. Transfer dehydrated tissue blocks into xylene for approximately 20 minutes until the tissue appears translucent. Avoid over-clearing, as this will cause tissue brittleness and fragility.
Infiltration:
First immerse tissue blocks in a 1:1 mixture of molten paraffin and xylene for 1 hour, then transfer successively to two changes of molten paraffin for approximately 1 hour each.
Embedding:
Apply a small amount of hot paraffin wax to rapidly attach the bottom to the embedding cassette, then place on a cooling platform to allow the paraffin to solidify.
Sectioning:
The sharpness of the microtome knife and appropriate paraffin block hardness directly affect section quality. Paraffin block hardness can be modified by placing it in cold water. Typically, section thickness is 3-5 μm. Gently transfer sections with a brush and float them in a 37°C water bath for expansion.
Mounting:
A constant temperature water bath is generally used, controlled at approximately 37-40°C, which facilitates the expansion of paraffin sections.
Baking:
After tissue sections are fully expanded, retrieve slides and place in a 60°C oven for baking (baking time varies according to tissue type; generally 30 minutes for most tissues, but bone tissue requires extended baking up to 1 hour). Deparaffinization can then proceed.
Deparaffinization and Rehydration
Xylene (5 min) — Xylene (5 min) — Xylene (5 min) — 100% Ethanol (5 min) — 100% Ethanol (5 min) — 95% Ethanol (3 min) — 85% Ethanol (3 min) — 75% Ethanol (3 min) — Deionized Water (5 min).
Note: Incomplete deparaffinization and rehydration may result in focal reactions and uneven washing, leading to non-specific background staining.
Antigen Retrieval
Place deparaffinized sections into antigen retrieval solution (select sodium citrate or EDTA according to primary antibody datasheet requirements). Perform heat-induced epitope retrieval (HIER) in a pressure cooker: heat until full pressure is reached, then continue heating for 5 minutes. Turn off heat, wait 10 minutes before removing the staining container, and cool at room temperature for 30 minutes. Alternatively, perform microwave antigen retrieval: place deparaffinized and rehydrated slides in a retrieval chamber, immerse in 1× working solution of antigen retrieval buffer, and boil at high power in a microwave. Maintain at low power for 15 minutes (note: replenish liquid to prevent excessive evaporation causing dry sections). Remove and allow to cool naturally to room temperature.
During formalin or paraformaldehyde fixation, some tissue antigens undergo protein cross-linking and aldehyde blocking, resulting in loss of antigenicity. Antigen retrieval re-exposes cellular antigenic determinants, improving positive detection rates. Common retrieval methods, from strong to weak, are generally classified as pressure cooker retrieval, microwave retrieval, and enzymatic retrieval (trypsin digestion).
Cell Permeabilization (Required for Cytoplasmic and Nuclear Targets)
For paraffin sections approximately 4 μm thick, permeabilization may be omitted as cells have already been sectioned. For thick sections >10 μm, use 0.1% Triton X-100 for permeabilization for 5-10 minutes. The purpose is to enable antibodies to fully enter cells for binding reactions.
Inactivation of Endogenous Peroxidase and Biotin
Draw a hydrophobic barrier around tissue sections, wash slides 3 times in PBST for 3 minutes each. Add 100 μL 3% H2O2 (volume should cover the sample area, adjustable as needed) per section, incubate at room temperature for 10 minutes. Wash with PBST for 3 minutes × 3 times. The purpose is to eliminate endogenous peroxidase activity.
Serum Blocking
Add 100 μL 5% normal goat serum blocking solution (volume should cover the sample area, adjustable as needed) per slide, incubate at room temperature for 30 minutes, then remove blocking solution. The purpose is to bind non-specific proteins, providing blocking action to reduce non-specific staining.
Antibody Incubation
Incubate with 100 μL primary antibody at room temperature in a humidified chamber for 1 hour or overnight at 4°C. Wash with PBST for 3 minutes, 3 times. Discard PBST and add 100 μL secondary antibody, incubate for 30 minutes to 1 hour. Wash with PBST for 3 minutes, 3 times.
Notes on Section Washing (Immersion Washing, Rinsing and Floating Washing):
- Wash individually to prevent cross-contamination from cross-reactions.
- Wash gently to prevent section detachment; immersion washing is generally used.
- Ensure sufficient washing time to completely remove bound substances.
DAB Chromogenic Detection
Add 100 μL DAB chromogenic substrate solution, develop for 5 minutes, terminate the chromogenic reaction with tap water. Both background intensity and specific staining intensity can be controlled by DAB incubation conditions. DAB development time is not fixed and is primarily controlled under the microscope; wash when specific staining is strong and background staining is minimal.
If deep brown color appears within seconds or tens of seconds: This likely indicates excessively high antibody concentration or prolonged antibody incubation time; reduce antibody concentration or shorten incubation time.
Additionally, if deep background appears very quickly, non-specific protein blocking may have been incomplete; extend blocking time.
If positive staining only appears after extended development (e.g., over ten minutes): On one hand, this may indicate insufficient antibody concentration or short incubation time (preferably incubate primary antibody overnight at 4°C); on the other hand, blocking time may have been too long.
Counterstaining
Counterstain with Harris hematoxylin for 3 minutes (1 minute for tumor tissues only), rinse with running tap water with agitation, differentiate in 1% acid alcohol for 1 second, briefly wash with tap water, blue in running tap water for 5 minutes (or blue with 2% ammonia water for 1-2 seconds). The purpose is to create cellular outlines for better localization of target proteins; Harris hematoxylin (nuclear stain) is commonly used.
Dehydration, Clearing and Mounting
For long-term preservation, neutral mounting medium is generally used for coverslipping, avoiding bubble formation. The method is to directly apply one drop of mounting medium onto the tissue on the slide, then cover with a coverslip. After mounting, bake at 60°C for 20 minutes.
Representative Experimental Results
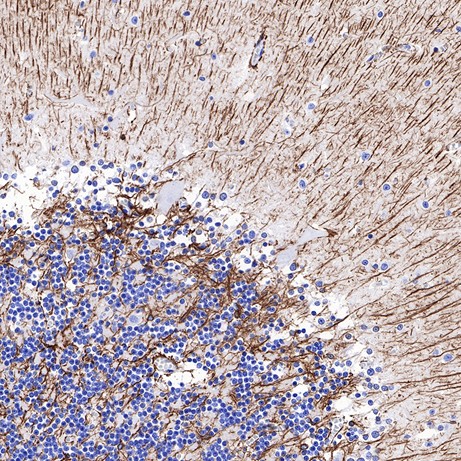
Positive GFAP staining in paraffin-embedded human cerebellum
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
