- Cart 0
- English
Why is it that you always find out that the Western Blot transfer failed only at the last step?
March 18, 2026
Clicks:75
In the lengthy workflow of Western Blot, one of the most frustrating moments for researchers occurs after completing electrophoresis, transfer, blocking, primary and secondary antibody incubation, and chemiluminescent exposure—only to find a blank film or bizarrely shaped bands on the autoradiograph. Tracing the root cause at this stage often requires repeating the entire experiment. The crux of the problem likely originated during the transfer step—proteins failed to efficiently transfer to the membrane or transferred unevenly. Ponceau S staining solution serves precisely as the "early warning system" to resolve this dilemma.
What Exactly Is Ponceau S Staining Solution?
Ponceau S staining solution is a water-soluble anionic azo dye, chemically named 3-hydroxy-4-[2-sulfo-4-(sulfophenylazo)phenylazo]-2,7-naphthalenedisulfonic acid tetrasodium salt, with molecular formula C₂₂H₁₂N₄Na₄O₁₃S₄ and molecular weight 760.6. This red dye is applied post-transfer and pre-immunodetection, capable of visualizing total protein on PVDF or nitrocellulose (NC) membranes within minutes, presenting red bands against a pink background.
Unlike gel staining methods such as Coomassie Brilliant Blue or silver staining, Ponceau S staining possesses unique reversibility—stained membranes can be completely destained through simple washing, restoring the membrane to its original state without affecting subsequent immunoblotting detection. This characteristic establishes it as the "gold standard" for Western Blot quality control.
The standard working concentration is 0.1% (w/v) dissolved in 5% acetic acid, with acidic pH. This formulation ensures both dye solubility and staining sensitivity while causing no significant impact on membrane protein antigenicity.
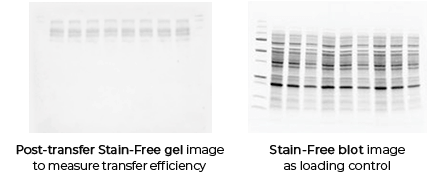
Figure 1: Detection of transfer efficiency in Western Blot. Left panel shows PVDF membrane after Ponceau S staining, displaying clear and uniformly transferred protein bands; right panel shows immunoblot detection results after washing the same membrane. Through Ponceau S prestaining, transfer quality can be confirmed before antibody incubation, preventing failed experiments.
Why Has Ponceau S Become the "First Line of Defense" for Transfer Quality Control?
Immediate Feedback: Identifying Problems Before Antibody Investment
In Western Blot reagent costs, primary antibodies typically constitute the largest proportion, especially imported branded antibodies. Proceeding with subsequent steps after transfer failure wastes expensive antibodies and valuable time. Ponceau S staining completes within 5 minutes post-transfer, enabling researchers to assess before antibody investment:
- Have proteins successfully transferred from gel to membrane?
- Is transfer uniform, without bubbles or "smiling" bands?
- Does the molecular weight distribution match expectations (roughly assessable by band position)?
This immediate feedback mechanism controls experimental failure risk at the earliest possible stage.
Reversibility: Multiple Detections from a Single Membrane
Ponceau S binds to proteins through electrostatic interactions (negatively charged dye binding to positively charged amino acid residues) and hydrophobic interactions (binding to non-polar regions of proteins). These binding forces are relatively weak. Simple rinsing with distilled water, PBS, or other buffers 2-3 times for 3-5 minutes each completely removes the dye without affecting protein antigenicity or binding sites on the membrane.
This reversibility enables two important applications:
- Multiple uses of the same membrane: First perform Ponceau S staining to confirm transfer efficiency, wash, then proceed with Western Blot; or complete detection of one target protein, wash, stain with Ponceau S to confirm total protein amount, then perform second detection (such as ECL, Dot-ELISA, etc.).
- Result traceability and publication quality control: During manuscript submission, editors or reviewers may request total protein loading controls. Ponceau S staining images serve as original evidence for total protein quantification, with immunoblot bands derived from the same membrane, providing irrefutable data consistency.
Compatibility and Sensitivity
Ponceau S is compatible with the two most commonly used solid-phase supports in Western Blot:
- PVDF membrane (polyvinylidene fluoride): Highly hydrophobic, high protein binding capacity (approximately 170-200 μg/cm²), preferred for low-abundance protein detection. Ponceau S displays clear coloration on PVDF with low background.
- Nitrocellulose membrane (NC membrane): Hydrophilic, extremely low background, though relatively brittle. Ponceau S performs equally excellently on NC membranes.
Important Exception: Ponceau S is not suitable for nylon membranes. Nylon membranes carry positive charges that strongly electrostatically adsorb the anionic Ponceau S dye, resulting in excessively high background that is difficult to remove.
Regarding sensitivity, Ponceau S can detect proteins above 250 ng, which is more than sufficient for routine loading amounts (10-50 μg total protein per well). Although less sensitive than silver staining or certain fluorescent dyes, it is completely adequate for transfer quality control and total protein quantification.
Which Experimental Scenarios Cannot Do Without Ponceau S Staining Solution?
Western Blot Transfer Efficiency Verification
This is the core application scenario. Whether using wet transfer, semi-dry transfer, or rapid transfer systems, transfer efficiency is influenced by multiple factors: electric field strength, transfer time, buffer ionic strength, gel concentration, and protein molecular weight. Ponceau S staining can visually reveal:
- High molecular weight proteins (>100 kDa): Often remain in gel due to gel pore size limitations or insufficient transfer time; Ponceau S staining can reveal absent or faint bands on membrane
- Low molecular weight proteins (<20 kDa): May pass through membrane due to over-transfer; Ponceau S staining can reveal faint membrane bands with deep bands on filter paper
- Uneven transfer: Bubbles or poor gel-membrane contact causing localized white "voids"
Protein Loading Normalization
Traditional Western Blot quantification relies on "housekeeping proteins" (such as GAPDH, β-actin, Tubulin), but housekeeping protein expression may be affected by experimental treatments (cell cycle, drug treatment, differentiation status), leading to normalization bias. In recent years, Total Protein Normalization (TPN) has been recognized as a more reliable method.
Ponceau S staining of total protein bands, analyzed through imaging software for total lane grayscale values or grayscale values within specific molecular weight ranges, serves as the basis for loading normalization. This method is unaffected by housekeeping protein expression fluctuations and complies with MIWE (Minimum Information About Western Blots) standards for quantitative accuracy.
Quality Control for Multicolor Fluorescent Western Blot
When performing multicolor fluorescent Western Blot (such as LI-COR Odyssey systems), multiple target proteins must be detected on the same membrane. Ponceau S staining can be performed before fluorescent detection to confirm uniform protein loading and transfer efficiency across all lanes, providing a foundation for subsequent quantitative comparison of fluorescent signals.
Recombinant Protein Purification Process Monitoring
At various stages of protein purification (lysate supernatant, eluate, post-purification samples), trace amounts of samples can be spotted onto membranes via Dot blot. Ponceau S staining rapidly determines protein presence and approximate concentration, faster than BCA or Bradford assays, and unaffected by detergent interference.
Nucleic Acid-Protein Interaction Studies
In Electrophoretic Mobility Shift Assay (EMSA) or Southwestern blot experiments, confirmation of successful protein transfer to membrane and binding with nucleic acid probes is required. Ponceau S staining serves as a pilot experiment to optimize transfer conditions, ensuring success of subsequent radioactive or non-radioactive labeling experiments.
How to Correctly Use Ponceau S Staining Solution?
Staining Protocol Essentials
- Stain immediately after transfer: After transfer completion, immerse membrane in Ponceau S staining solution (0.1% in 5% acetic acid), shake on orbital shaker at room temperature for 3-5 minutes. If bands are weak, extend to 10 minutes, but prolonged staining increases background.
- Observation and documentation: Membrane can be removed during staining and observed against white background. Once clear bands appear, immediately terminate staining with distilled water or PBS. Photograph using digital camera or gel documentation system, ensuring uniform lighting and avoiding reflections.
- Thorough washing: Rinse with distilled water, PBS, or TBST 2-3 times, 3-5 minutes each, until background appears light pink or colorless. Residual dye may interfere with subsequent antibody binding or ECL background.
Common Usage Misconceptions
Misconception 1: Excessive pursuit of deep red staining
Ponceau S staining bands should appear red; excessive staining causes overly high background and increases washing difficulty. Stop staining as soon as bands are clearly visible.
Misconception 2: Neglecting thorough washing
Insufficient washing may cause residual dye to continue releasing during blocking or antibody incubation, elevating background. After washing, lightly press membrane with white paper to observe whether red traces exude.
Misconception 3: Use on nylon membranes
As previously mentioned, Ponceau S strongly binds to positive charges on nylon membranes, causing irreversible high background. For nylon membranes, use alternative quality control methods such as Coomassie Brilliant Blue gel staining or amido black membrane staining.
Misconception 4: Long-term storage after staining
Ponceau S-stained membranes should be photographed and washed promptly; prolonged storage (>24 hours) may cause dye-protein binding to become too firm for complete removal.
From "Empiricism" to "Data-Driven": The Value of Ponceau S Staining in Quantitative Western Blot
Traditional Western Blot is often considered a "semi-quantitative" technique, primarily due to difficulties in loading control and signal normalization. Ponceau S staining combined with total protein normalization strategies is driving Western Blot toward greater rigor:
- Linear range validation: Ponceau S-stained total protein shows good linear relationship with loading amount (typically within 5-50 μg range), usable for standard curve generation.
- Dual normalization with housekeeping and total protein: Simultaneously detect housekeeping proteins and Ponceau S total protein, comparing consistency between two normalization methods to improve data reliability.
- Publication standard compliance: Increasingly, journals require total protein loading evidence; Ponceau S staining images provide a convenient means to meet this requirement.
Conclusion
Though Ponceau S staining solution is a "simple" dye, it plays an irreplaceable role in the Western Blot workflow. It serves as the "diagnostic report" for experiments, controlling transfer quality before investing expensive reagents and valuable time. In today's research environment where data reliability receives increasing attention, this reversible, simple, and intuitive quality control method is evolving from an "optional step" to "standard operating procedure." Mastering the correct use of Ponceau S staining means adding insurance to Western Blot experiments and assurance to data quality.
Absin Ponceau S Staining Solution Recommendation:
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
