- Cart 0
- English
Why Do Your Protein Samples Always "Run Off Track" During Electrophoresis?
March 18, 2026
Clicks:352
In routine protein research experiments, one of the most frustrating scenarios for researchers is when carefully prepared protein samples exhibit distorted bands, trailing smears, or gel backgrounds as dark as the night sky after electrophoresis. The root cause of these problems often lies not in the samples themselves, but in the selection and operational details of the electrophoresis system. A complete protein electrophoresis kit represents a systematic solution to address these critical pain points.
What Exactly Is a Protein Electrophoresis Kit?
A protein electrophoresis kit is a complete electrophoretic solution based on the Tris-glycine buffer system (containing SDS), specifically designed for SDS-polyacrylamide gel electrophoresis (SDS-PAGE). This technique represents the most fundamental and core method in current protein separation analysis, capable of high-resolution separation of complex protein samples according to molecular weight.
A standard protein electrophoresis kit typically comprises four synergistic modules:
- Electrophoresis Buffer System: 5× concentrated Tris-glycine-SDS buffer, diluted to 1× working solution for use. This buffer system creates the necessary conditions for maintaining protein negative charge and stabilizing the pH environment.
- Sample Processing System: 4× loading buffer containing SDS, β-mercaptoethanol, bromophenol blue, and glycerol. SDS denatures proteins and imparts uniform negative charge; β-mercaptoethanol cleaves disulfide bonds; bromophenol blue indicates the electrophoresis front; glycerol increases sample density for convenient loading.
- Gel Staining System: Coomassie Brilliant Blue staining solution, which forms stable blue complexes with proteins with sensitivity reaching the microgram level.
- Background Destaining System:配套 destaining solution for removing non-specific dye adsorption from the gel background, enabling clear visualization of protein bands.
These four modules constitute the complete "preparation-separation-visualization" workflow, each being indispensable.
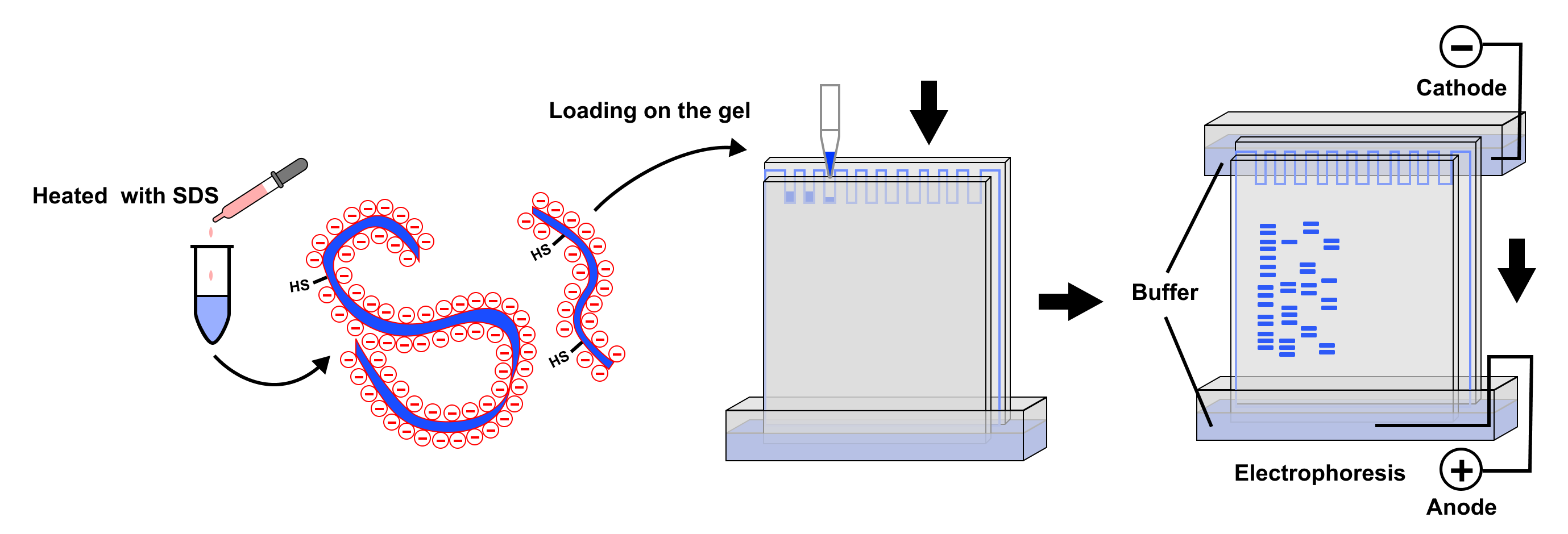
Figure 1: SDS-PAGE Working Principle. Protein samples treated with SDS and reducing agents form rod-shaped complexes with SDS, carrying uniform negative charges. Under electric field, proteins migrate toward the positive electrode, with migration rate inversely proportional to the logarithm of molecular weight, thereby achieving separation by molecular weight. After staining, proteins of different molecular weights appear as distinct bands.
Why Has SDS-PAGE Become the "Gold Standard" for Protein Analysis?
To understand the irreplaceability of SDS-PAGE, one must delve into the elegance of its technical principles.
Charge Homogenization: Eliminating Native Charge Variations
The charge state of native proteins is determined by their amino acid composition and solution pH. Different proteins may carry varying positive or negative charges at the same pH, leading to unpredictable electrophoretic migration behaviors. SDS (sodium dodecyl sulfate) is an anionic detergent whose hydrophobic tail binds to the protein backbone while the hydrophilic head faces outward, coating all proteins with a uniform layer of negative charge. Approximately 1.4 grams of SDS bind per gram of protein, with charge density far exceeding the intrinsic charge differences of proteins themselves, thereby making migration rate dependent solely on molecular size.
Molecular Conformation Standardization: Breaking Spatial Structure Barriers
The native three-dimensional structure of proteins (primary, secondary, tertiary, and quaternary structures) significantly affects their migration behavior in gels. β-mercaptoethanol in the loading buffer acts as a reducing agent capable of cleaving disulfide bonds between cysteine residues; 100°C heat treatment further disrupts hydrogen bonds and hydrophobic interactions. This series of operations "flattens" all proteins into linear polypeptide chains, ensuring that separation reflects only molecular weight differences rather than conformational differences.
Molecular Sieving Effect: The Resolving Power of Polyacrylamide Gels
Polyacrylamide gels possess a three-dimensional network structure that acts as a molecular sieve, generating different frictional resistances for proteins of different sizes. Small proteins easily traverse gel pores and migrate rapidly; large proteins encounter severe resistance and migrate slowly. By adjusting acrylamide concentration (typically 8%-15%) and cross-linking degree, resolution for specific molecular weight ranges can be optimized.
Which Experimental Scenarios Cannot Do Without Protein Electrophoresis Kits?
Recombinant Protein Expression Verification
In molecular cloning and protein expression experiments, SDS-PAGE serves as the first checkpoint for verifying whether target proteins have been successfully expressed. By comparing whole-cell lysates before and after induction, as well as under different induction conditions, one can rapidly determine whether the target protein (typically bearing His-tag or GST-tag, with predictable molecular weight) has appeared. If the band position matches the theoretical molecular weight, this constitutes preliminary evidence of successful expression.
Protein Purification Process Monitoring
During chromatographic purification (such as affinity chromatography, ion exchange chromatography, and gel filtration chromatography), real-time monitoring of protein content and purity in collected fractions is essential. SDS-PAGE combined with Coomassie Brilliant Blue staining can determine which fractions contain target proteins and whether contaminant proteins have been effectively removed within tens of minutes. This immediate feedback is crucial for optimizing purification conditions.
Protein Sample Concentration Determination
Although Bradford or BCA assays are preferred for quantitative protein concentration determination, when samples contain detergents or reducing agents that interfere with these colorimetric methods, SDS-PAGE combined with standard protein gradients (such as BSA serial dilutions) can serve as semi-quantitative or even quantitative analytical means. By comparing band grayscale values, sample concentration can be estimated with reasonable accuracy.
Prerequisite Step for Immunoblotting (Western Blot)
Although Western Blot employs different transfer and detection systems, its separation phase relies entirely on SDS-PAGE. The buffer system and loading buffer provided by protein electrophoresis kits are prerequisites for successful Western Blot. The quality of band separation directly determines the specificity and signal-to-noise ratio of subsequent antibody hybridization.
Protein Complex Subunit Analysis
For multi-subunit protein complexes (such as ribosomes and proteasomes), SDS-PAGE can dissociate and separate individual subunits, allowing inference of complex composition through band number and molecular weight. Combined with mass spectrometry identification, this enables compositional analysis of unknown protein complexes.
Proteomics Differential Analysis
In comparative proteomics research, through two-dimensional electrophoresis (2D-PAGE) or differential gel electrophoresis (DIGE), systematic comparison of protein expression profile changes under different physiological or pathological states can be achieved. SDS-PAGE serves as the second-dimension separation technique, combined with isoelectric focusing (IEF) to achieve high-resolution two-dimensional separation of proteins.
How to Transform Your Electrophoresis Results from "Usable" to "Publication-Quality"?
Buffer Preparation Pitfalls
When diluting 5× buffer, high-purity deionized water or ultrapure water is recommended. Ordinary distilled water may contain ionic impurities that alter buffer ionic strength and conductivity, leading to abnormal electrophoresis speed or band distortion. The diluted 1× working buffer should be prepared fresh for immediate use; prolonged storage may cause pH drift due to CO₂ dissolution.
The Science of Pre-Loading Heat Treatment
The 100°C heating (typically 3-5 minutes) after mixing samples with loading buffer is critical for the denaturation step, but several details are often overlooked in practice:
- Avoid excessive heating duration: Overheating may cause protein aggregation or degradation, particularly for proline-rich proteins
- Centrifugation after heating is mandatory: Remove potential precipitates to prevent well clogging or band trailing
- Cool to room temperature before loading: High-temperature samples disrupt the buffer layer near gel wells, causing irregular bands
Gel Concentration Selection Strategy
Acrylamide concentration determines the separation range of the gel:
- 8% gel: Suitable for separating large molecular weight proteins (30-200 kDa)
- 10-12% gel: Suitable for separating medium molecular weight proteins (10-100 kDa), the most commonly used "universal gel"
- 15% gel: Suitable for separating small molecular weight proteins (10-50 kDa)
For samples with large molecular weight spans, gradient gels (such as 4-20%) are preferable, though at higher cost.
The Balancing Art of Staining and Destaining
Although Coomassie Brilliant Blue staining is classical, achieving low-background, high-contrast results requires mastering the principle of "moderation":
- Staining time: Overnight staining at room temperature on an orbital shaker usually yields optimal results, but when urgently needed, temperature can be raised to 37°C to shorten time
- Staining solution recycling: Coomassie Brilliant Blue staining solution can be reused 2-3 times, but sensitivity decreases with usage
- Destaining strategy: Change destaining solution 3-5 times; higher temperature can accelerate initial destaining, with subsequent lower temperature to preserve band signal. If background remains dark, extend destaining time overnight, but note that excessive destaining will weaken faint band signals.
From "Band Viewing" to "Data Interpretation": Deep Analysis of Electrophoresis Results
A high-quality SDS-PAGE gel is not merely an experimental record but contains rich information:
- Band position: Comparison with protein molecular weight markers to determine whether molecular weight matches expectations. Significant deviation may indicate post-translational modifications (such as glycosylation increasing apparent molecular weight), protein degradation, or alternative splicing.
- Band shape: Sharp, symmetrical bands indicate good protein homogeneity; trailing may result from overloading or high salt/detergent content in samples; diffuse bands may stem from incomplete protein denaturation or sample diffusion during electrophoresis.
- Background intensity: Uniform light background indicates proper staining-destaining balance; dark background with punctate particles suggests inadequate staining solution filtration or incomplete gel washing; inter-lane background differences may reflect uneven loading buffer mixing.
- Inter-lane comparison: Band intensity differences in parallel lanes directly reflect relative protein quantity changes, providing basis for semi-quantitative analysis.
Conclusion
As fundamental infrastructure in life science research, the value of protein electrophoresis kits lies not only in providing a standardized solution but also in translating complex biophysical principles into reproducible daily operations. From recombinant protein verification to proteomics exploration, from teaching laboratories to industrial quality control, SDS-PAGE technology remains an indispensable tool for protein researchers due to its reliability, economy, and universality. Mastering the essence of this technology means obtaining the key to unlock the world of proteins.
Absin Protein Electrophoresis Kit Recommendation:
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
