- Cart 0
- English
Powering Pancreatic Cancer Innovation! Absin's Copper Detection Kit Unlocks New Frontiers in Cuproptosis and Immunotherapy Research
March 12, 2026
Clicks:73
As a core supplier deeply rooted in the life sciences field, Absin consistently provides robust support to global researchers with high-quality products, empowering cutting-edge research exploration. Recently, a landmark study published in Molecular Cancer (DOI: 10.1186/s12943-025-02529-x) focused on cuproptosis resistance and anti-tumor immunotherapy response mechanisms in pancreatic cancer, utilizing Absin's core detection tools to complete critical experimental validation, providing novel targeted strategies and theoretical foundations for pancreatic cancer treatment, and demonstrating the reliable value of Absin products in life science research.
Title: Targeting SERPINB3–MAPK axis-mediated cuproptosis resistance enhances the response to antitumor immunotherapy
Journal: Molecular Cancer (IF=33.9)
DOI: https://doi.org/10.1186/s12943-025-02529-x
Absin Products Used: Copper Microplate Assay Kit (abs580140)


Focusing on Clinical Pain Points, Constructing Multi-dimensional Research Strategy
Pancreatic ductal adenocarcinoma (PDAC), as a highly malignant tumor with a 5-year survival rate of merely 13%, presents limited efficacy with existing therapeutic regimens, urgently necessitating the identification of novel therapeutic targets and strategies. Cuproptosis, as an emerging form of cell death closely associated with tumor metabolism and therapeutic response, has yet to have its regulatory mechanisms in pancreatic cancer resistance and relationship with immunotherapy fully elucidated.
The research team constructed a comprehensive investigative framework of "phenomenon discovery—mechanism elucidation—therapeutic development" around this core scientific question:
- 1. Initially, through clinical sample analysis and cellular experiments, the association between the "copper accumulation" phenotype and cuproptosis resistance in pancreatic cancer was validated;
- 2. Multi-omics analysis was employed to screen key regulatory factors, identifying SERPINB3 as a central player in cuproptosis resistance;
- 3. The molecular mechanism of SERPINB3-mediated cuproptosis resistance was deeply dissected, revealing its critical pathway through MAPK signaling regulation of FDX1 transcription;
- 4. MOF-based nanodelivery systems and combination therapeutic regimens were developed targeting this mechanism, with therapeutic efficacy validated in in vitro and in vivo models.
The entire research logic progressed systematically, departing from clinical phenomena and integrating multidisciplinary technologies including molecular biology, nanomaterials science, and immunology to provide systematic solutions for precision therapy in pancreatic cancer.
Breakthrough Research Findings Reshaping Pancreatic Cancer Treatment Paradigms
Through a series of rigorous experiments, this study achieved multiple milestone accomplishments:
- 1. First discovery of the paradoxical phenotype of "copper accumulation yet cuproptosis resistance" in pancreatic cancer, with copper content positively correlating with tumor malignancy and negatively correlating with patient prognosis (corresponding to Figs. 1B, 1F-H in original text);
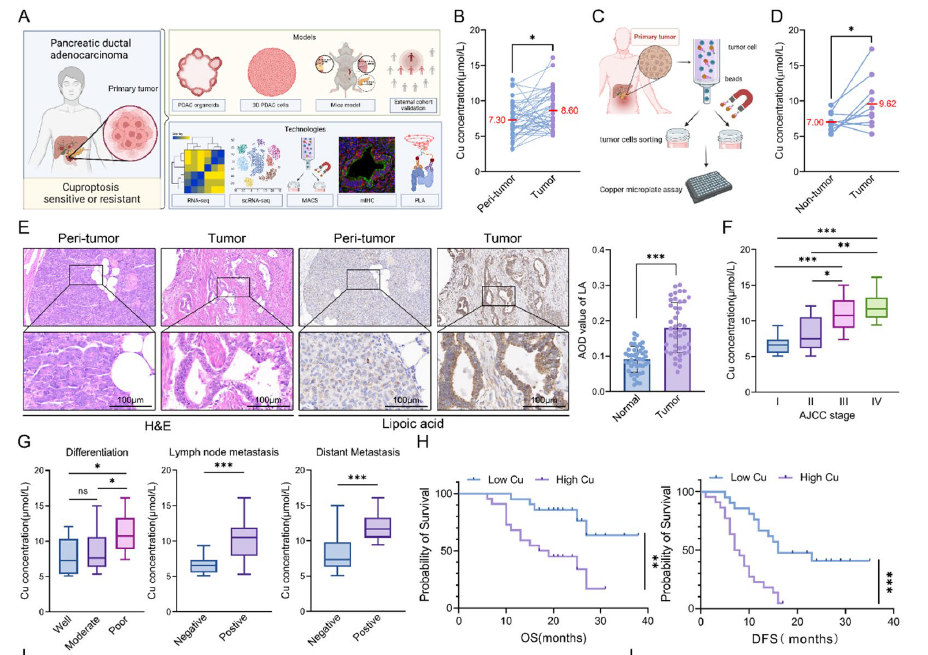
Fig. 1 Pancreatic cancer cells exhibit cuproptosis resistance. A Overall experimental scheme of the study. B Paired line scatter plot showing copper (Cu) concentrations in PDAC tissues (purple dots) (n = 43) and adjacent normal tissues (blue dots) (n = 43). C-D Schematic illustration of cancer cell sorting from cancer tissues of PDAC patients and Cu concentration determination (C). Paired line scatter plot showing the Cu concentrations in the different groups (D). E We used a lipoic acid-specific antibody to quantify the abundance of lipoylated proteins, and the representative images of lipoylated protein IHC in pancreatic cancer tissues (n = 43) and adjacent normal pancreatic tissues (n = 43) from representative patients with PDAC in the cohort from our center are shown (scale bar: 100 μm). Bar chart showing the difference in the average optical density (AOD) values of lipoylated proteins between PDAC tumor and peritumoral tissues. F Box plot showing the Cu concentrations between patients with different AJCC stages. G Statistical graphs of the correlations between Cu concentrations and pathological characteristics in PDAC patients. H Survival analysis of OS and DFS between PDAC patients with high and low Cu concentrations.
- 2. Identification of SERPINB3 as a key regulatory factor for cuproptosis resistance and immune evasion in pancreatic cancer, with its high expression closely associated with tumor progression and poor prognosis (corresponding to Figs. 1M-N, 3D-E in original text);
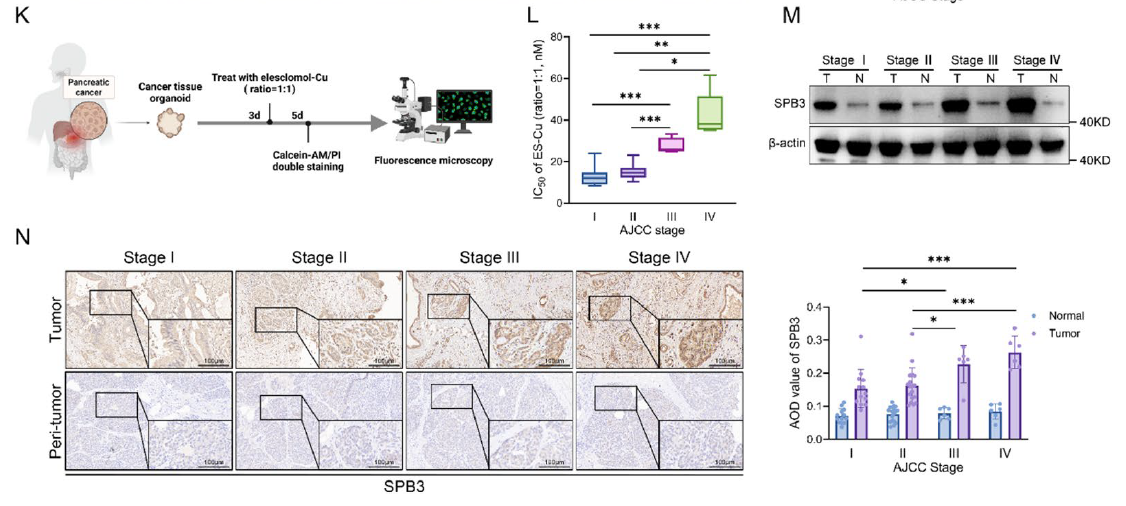
K Schematic illustration of the induction and quantification of cuproptosis in PDOs, which were successfully generated from 32 out of 43 pancreatic cancer specimens obtained from the clinical biobank. L IC50 values for ES-Cu in PDOs with different AJCC stages after 48 h of 2-hour pulsed treatment with different concentrations. M WB analysis showing the expression levels of SERPINB3 in pancreatic cancer tissues and adjacent normal pancreatic tissues from patients with different AJCC stages. N Representative images of SERPINB3 IHC in pancreatic cancer tissues (n = 43) and adjacent normal pancreatic tissues (n = 43) from representative PDAC patients with different AJCC stages in the cohort from our center (scale bar: 100 μm). Box plot showing the AOD values of SERPINB3 between patients with different AJCC stages
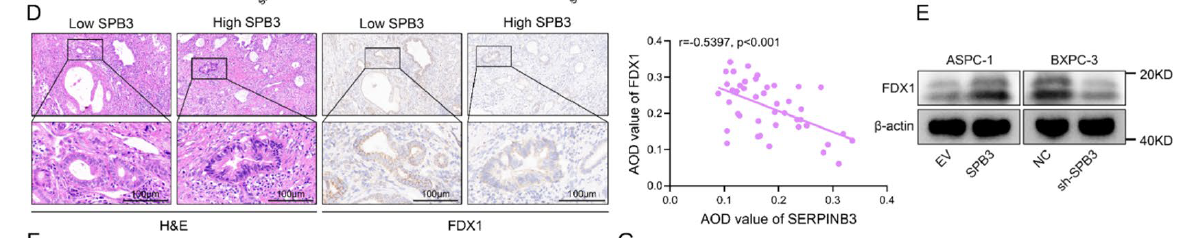
D Representative images of FDX1 immunohistochemical staining in samples with high SERPINB3 expression (SERPINB3hi, n = 22) and low SERPINB3 expression (SERPINB3low, n = 21) pancreatic cancer tissues from representative patients with PDAC in the cohort from our center (scale bar: 100 μm). Scatter plot showing correlation analysis of AOD values between SERPINB3 and FDX1 (right). E WB analysis of FDX1 expression in the indicated ASPC-1 (EV or OE-SERPINB3) and BXPC-3 (NC or sh-SERPINB3) cells.
- 3. Elucidation of molecular mechanisms: SERPINB3 inhibits chaperone-mediated autophagy (CMA)-mediated MEK1 degradation, constitutively activating the MAPK/ERK pathway, thereby suppressing FOXO3 nuclear localization and FDX1 transcription, ultimately leading to cuproptosis resistance; concurrently upregulating PD-L1 expression to promote immune evasion (corresponding to Figs. 4A-E, 5L-P, 6A-N in original text);
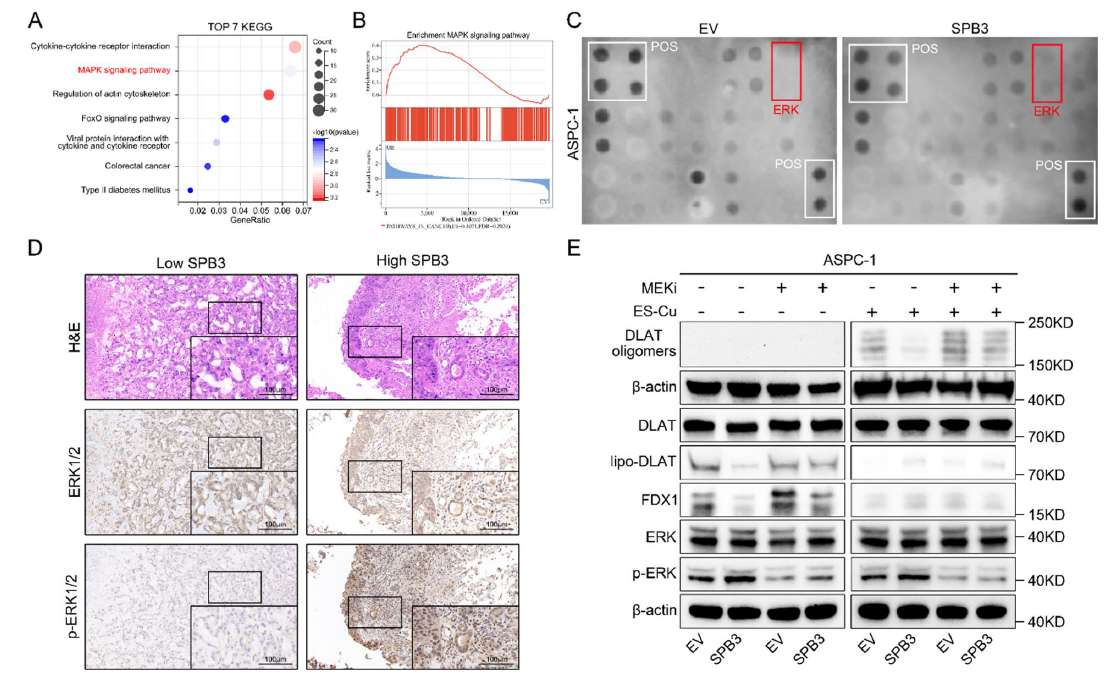
Fig. 4 SERPINB3 induces cuproptosis resistance by promoting the activation of the MAPK signaling pathway. A The top 7 KEGG terms enriched in genes upregulated after SERPINB3 overexpression in ASPC-1 cells (n = 3 biologically independent sequenced samples per group). B Gene set enrichment analysis of upregulated signatures after SERPINB3 overexpression in ASPC-1 cells (n = 3 biologically independent sequenced samples per group). P values were calculated via Kolmogorov‒Smirnov tests. NES, normalized enrichment score. C The activation status of MAPK signaling pathway-associated proteins was evaluated by a phosphorylation antibody array. D Representative images of ERK1/2 and phosphorylated ERK1/2 immunohistochemical staining in SERPINB3hi (n = 22) and SERPINB3low (n = 21) pancreatic cancer tissues from representative patients with PDAC in the cohort from our center (scale bar: 100 μm). E The ASPC-1-EV and ASPC-1-OE-SERPINB3 cells were treated with or without ES-Cu (40 nM, ratio = 1:1) or trametinib (40 nM) for 24 h, and cell lysates were collected for WB analysis.
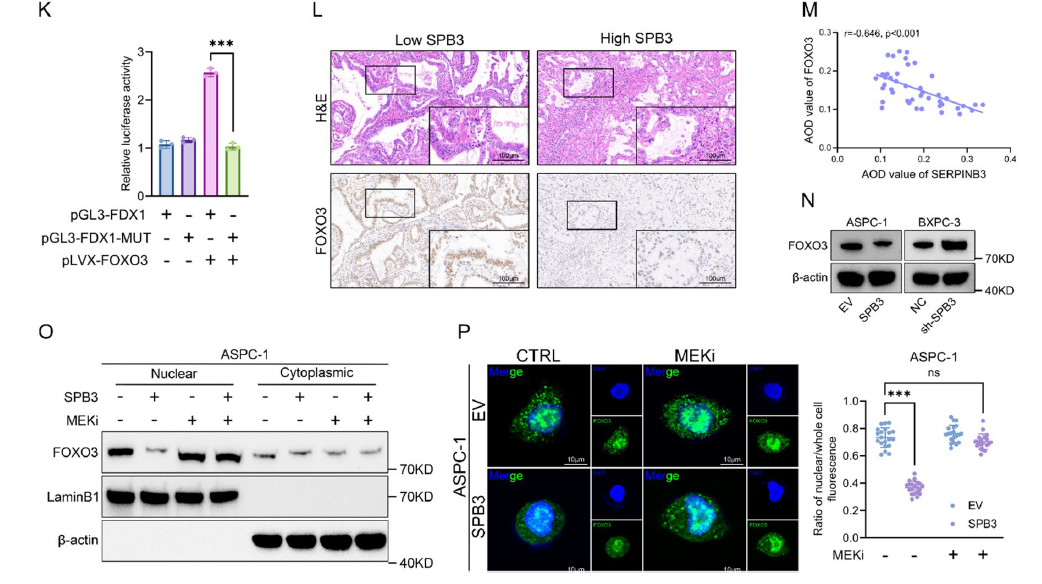
(12) . Correlation analysis of the AOD values for SERPINB3 and FOXO3 (M). N WB analysis of FOXO3 expression in the indicated cells (ASPC-1 [EV or OE-SERPINB3] and BXPC-3 cells [NC or sh-SERPINB3]). O WB analysis of nuclear and cytoplasmic fractions from whole-cell lysates of ASPC-1-EV and ASPC-1-OE-SERPINB3 cells treated with (+) or without (-) trametinib (10 nM) for 24 h. P ASPC-1-EV and ASPC-1-OE-SERPINB3 cells were treated with (+) or without (-) trametinib (100 nM) for 24 h, and representative images of immunofluorescence staining revealed that trametinib reversed the decrease in the expression of FOXO3 in the nucleus induced by the overexpression of SERPINB3 (left). Scatter plot showing the difference in the expression of FOXO3 in the nucleus between the different groups (right)
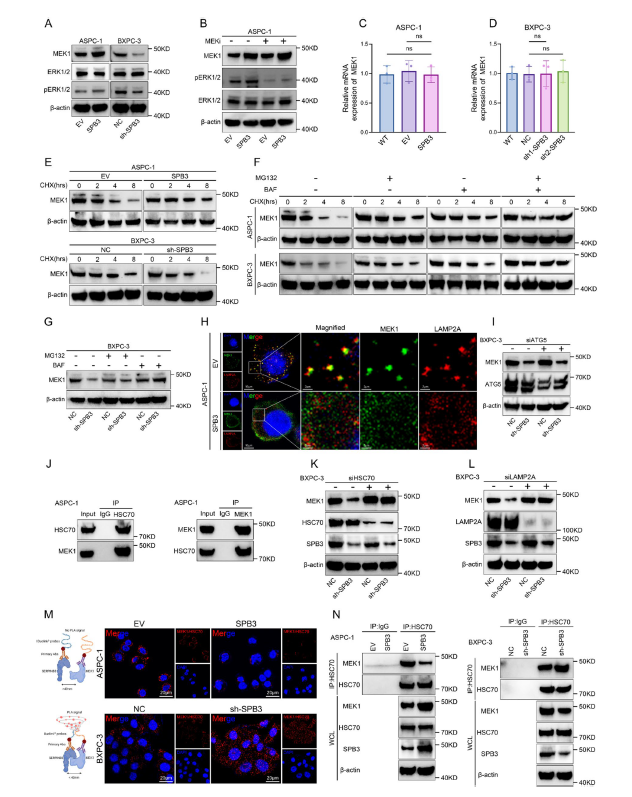
Fig. 6 SERPINB3 inhibits MEK1 degradation through the CMA pathway. A WB analysis of MEK1, ERK1/2 and phosphorylated ERK1/2 expression in the indicated ASPC-1 (EV or OE-SERPINB3) and BXPC-3 cells (NC or sh-SERPINB3). B ASPC-1-EV and ASPC-1-OE-SERPINB3 cells were treated with (+) or without (-) trametinib (100 nM) for 24 h, and cell lysates were collected for WB analysis. C qRT-PCR analysis of MEK1 expression in ASPC-1-EV and ASPC-1-OE-SERPINB3 cells. The data are presented as the means ± SEMs. D qRT-PCR analysis of MEK1 expression in BXPC-3-NC and BXPC-3-sh-SERPINB3 cells. The data are presented as the means ± SEMs. E The indicated ASPC-1 (EV or OE-SERPINB3) and BXPC-3 cells (NC or sh-SERPINB3) were treated with cycloheximide (CHX, 100 μg/ml) for the indicated times, and the resulting cell lysates were collected for WB analysis. F The indicated ASPC-1 (EV or OE-SERPINB3) and BXPC-3 cells (NC or sh-SERPINB3) were treated with CHX (100 μg/ml), MG-132 (10 μM), or BAF (100 nM) for the indicated times, and the resulting cell lysates were collected for WB analysis. G The BXPC-3-NC and BXPC-3-sh-SERPINB3 cells were treated with CHX (100 μg/ml), MG-132 (10 μM), or BAF (100 nM) for 8 h, and cell lysates were collected for WB analysis. H ASPC-1-EV and ASPC-1-OE-SERPINB3 cells were treated with BAF (100 nM) for 3 h, and colocalization between MEK1 (green) and a lysosomal marker (LAMP2A, red) was examined by confocal immunofluorescence staining (scale bar: 10 μm, 2 μm). I BXPC- 3-NC and BXPC-3-sh-SERPINB3 cells were transfected with control siRNA or ATG siRNA for 48 h, and cell lysates were collected for WB analysis. J Wild-type ASPC-1 cells were treated with BAF (100 nM) for 3 h, and cell lysates were collected to detect the interaction between MEK1 and HSC70 by Co-IP and WB analysis. K BXPC-3-NC and BXPC-3-sh-SERPINB3 cells were transfected with control siRNA or HSC70 siRNA for 48 h, and cell lysates were collected for WB analysis. L BXPC-3-NC and BXPC-3-sh-SERPINB3 cells were transfected with control siRNA or LAMP2A siRNA for 48 h, and cell lysates were collected for WB analysis. M Schematic of the PLA for MEK1 and HSC70 (left) and representative fluorescence images (right) of the PLA probe-bound fluorophore in the indicated ASPC-1 cells (top row) and BXPC-3 cells (bottom row) (scale bar: 20 μm). N The indicated ASPC-1 cells (left) and BXPC-3 cells (right) were treated with BAF (100 nM) for 3 h. Cell lysates were collected to detect the interaction between MEK1 and HSC70 via Co-IP and WB analysis. WCL: whole-cell lysate
- 4. Development of MOF-M-CuSx nanodelivery system achieving targeted co-delivery of copper ions and MEK inhibitors, with combined anti-PD-1 therapy significantly inhibiting tumor growth and prolonging survival in SERPINB3-high expressing pancreatic cancer models (corresponding to Figs. 7J-K, 9B-E, 9M in original text).
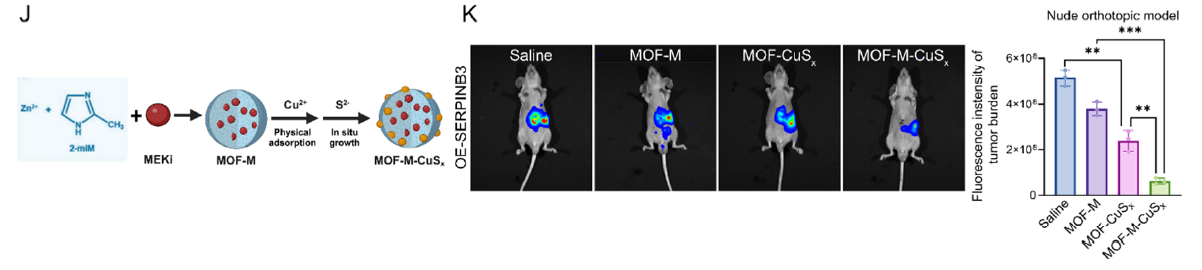
J Schematic illustration of MOF-M-CuSx synthesis. K A nude mouse orthotopic model was generated via the injection of ASPC-1-OE-SERPINB3 cells. Bioluminescence of orthotopic tumors from the mice in different groups treated for three weeks with saline, MOF-M, MOF-CuSx, or MOF-M-CuSx (200 μg/mL, 100 μL, via tail vein) on day 7 after the injection of one million ASPC-1-OE-SERPINB3 cells into the pancreas. Images were collected four weeks after injection. Bar chart showing the fluorescence intensity of the tumor burden in different groups
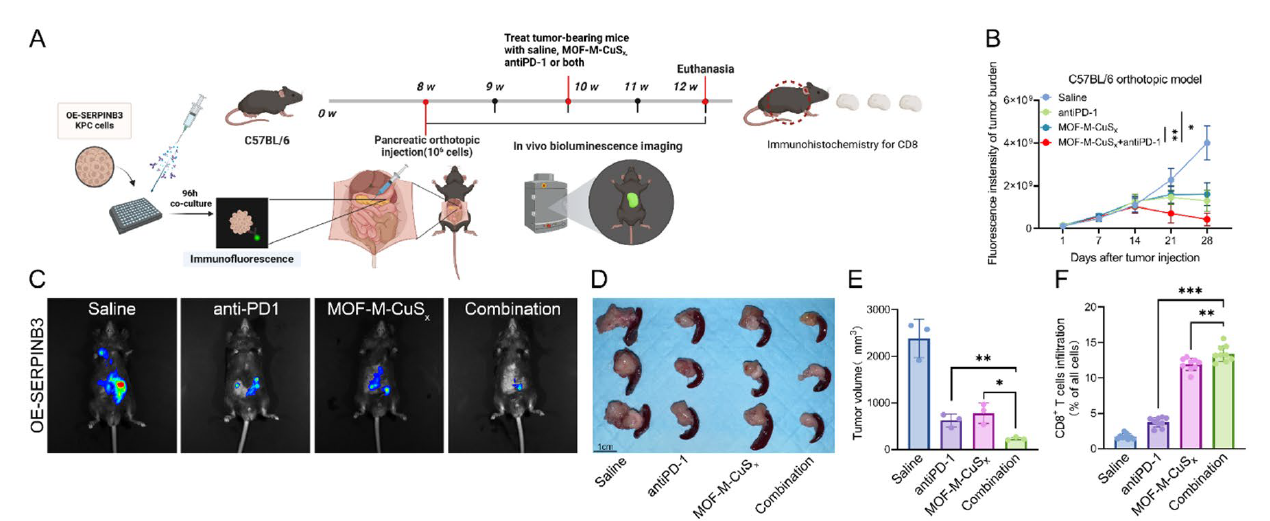
Fig. 9 Induction of cuproptosis in combination with anti-PD-1 therapy can further inhibit the progression of SERPINB3hi PDAC. A The workflow employed to establish C57BL/6 orthotopic tumor models and subsequent treatment. B-C Different groups of C57BL/6 mice received saline, anti-mouse PD-1 monoclonal antibodies (Bio X Cell, 100 μg, i.p., every three days), MOF-M-CuSx (200 μg/mL, 100 μL, via tail vein), or a combination of both for two weeks on day 14 following the orthotopic pancreatic injection of one million KPC-OE-SERPINB3 cells. Each mouse underwent in vivo bioluminescence imaging once a week from day 1 to day 28 postinjection, and the fluorescence intensities representing the tumor burden in each group were plotted individually (B). Representative bioluminescence images of the different groups on day 28 are presented (C). D-E Different groups of C57BL/6 mice received saline, anti-mouse PD-1 monoclonal antibodies (Bio X Cell, 100 μg, i.p., every three days), MOF-M-CuSx (200 μg/mL, 100 μL, via tail vein), or a combination of both for two weeks on day 14 following the orthotopic pancreatic injection of one million KPC-OE-SERPINB3 cells. Tumors were harvested at 4 weeks after pancreatic orthotopic injection
These findings not only reveal the core mechanisms of cuproptosis resistance in pancreatic cancer but also provide a novel "cuproptosis sensitization + immune awakening" combination therapeutic strategy, laying a solid foundation for clinical translation.
Absin Core Product Support: Building Solid Foundations for Experimental Data
In the critical experimental segments of this study, the Absin Copper Microplate Assay Kit (Catalog No.: abs580140) served as a core detection tool, providing comprehensive support for copper ion concentration quantitative analysis throughout the research, becoming an important guarantee for reliable output of research conclusions.
This product employs highly specific detection principles with simple operation and high sensitivity, capable of precisely quantifying copper ion concentrations in cellular and tissue samples, meeting multi-scenario detection requirements for both clinical samples and experimental models. Its stable detection performance and precise quantitative results fully matched the stringent requirements for copper ion concentration analysis in this study, providing direct experimental evidence for validation of the "pancreatic cancer copper accumulation phenotype."
Key Roles Throughout the Entire Process, Empowering Core Research Segments
The Absin Copper Microplate Assay Kit (abs580140) played an irreplaceable supporting role throughout the research, permeating multiple core experimental segments:
- 1. Clinical Sample Validation Segment: Precisely detected copper ion concentrations in 43 paired pancreatic cancer tissues and adjacent normal tissues, confirming significantly elevated copper content in pancreatic cancer tissues, providing critical data for the "copper accumulation phenotype" (corresponding to Fig. 1B in original text);
- 2. Tumor Cell Sorting Validation Segment: Through MACS sorting technology, EpCAM⁺ tumor cells and non-tumor cells were separated from pancreatic cancer tissues, with detection results clearly demonstrating significantly higher copper ion concentrations in tumor cells compared to non-tumor cells, further validating the cell specificity of copper accumulation (corresponding to Fig. 1D in original text);
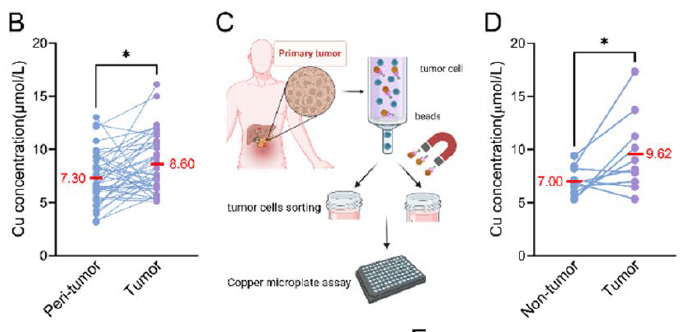
B Paired line scatter plot showing copper (Cu) concentrations in PDAC tissues (purple dots) (n = 43) and adjacent normal tissues (blue dots) (n = 43). C-D Schematic illustration of cancer cell sorting from cancer tissues of PDAC patients and Cu concentration determination
- 3. Therapeutic Efficacy Assessment Segment: In orthotopic pancreatic tumor models, quantified the impact of different treatment regimens (ES-Cu, MOF-CuSx, etc.) on tumor tissue copper ion concentrations, confirming that MOF nanodelivery systems can effectively enhance intratumoral copper enrichment, providing direct evidence for the superiority of treatment protocols (corresponding to Fig. 7I in original text).
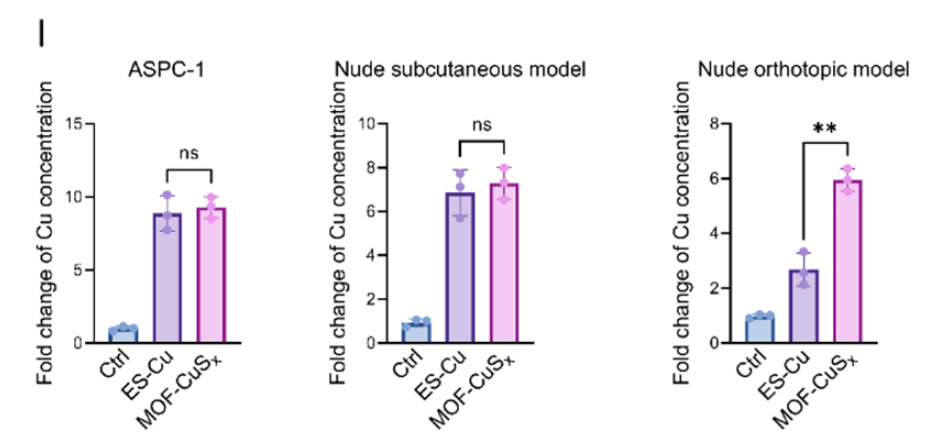
I Bar charts showing the fold change in copper concentration in tumor cells following different drug treatments in pancreatic cancer cells in vitro and in subcutaneous tumors and orthotopic tumors in nude mice.
The high precision and stability of this product ensured the reliability of copper ion concentration data across all segments, providing solid data support for the research team to reveal the association between copper accumulation and cuproptosis resistance, and to validate the efficacy of nanodelivery systems, serving as an important foundation for the smooth advancement of the research.
Continuously Deepening Research Needs, Empowering Breakthroughs in More Fields
The publication of these research findings once again confirms the core value of Absin products in cutting-edge life science research. As a supplier dedicated to research needs, Absin consistently adheres to the philosophy of "empowering innovative research," continuously optimizing product performance, enriching product categories, and providing comprehensive experimental solutions for multiple fields including oncology, molecular biology, and immunology.
This content is based on the original publication in Molecular Cancer (DOI: 10.1186/s12943-025-02529-x), interpreted and compiled by AI; all original figures, data, and intellectual property rights belong to the original journal and research team. Should any infringement occur, please contact us promptly for removal, and we will actively cooperate in addressing such matters.
|
Item NO. |
Product Name |
Size |
| abs580140 | Copper Microplate Assay Kit | 96T |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
