- Cart 0
- English
Absin Antibodies Power Breakthrough Osteoporotic Bone Repair Research: Micro-/Nanostructured Hydroxyapatite Scaffold Mechanism Study Published in Bioactive Materials
March 12, 2026
Clicks:73
Repair of osteoporotic bone defects remains a significant challenge in clinical practice—compromised osteogenesis, dysregulated angiogenesis, and poor scaffold integration have left countless patients facing non-union fractures and increased disability risk. Recently, a landmark study published in Bioactive Materials introduced a novel scaffold design strategy of "mechanical-bioactive decoupling," offering an innovative solution for osteoporotic bone repair. Absin's premium antibody products served as the core support for mechanistic validation in this research.
Article Title: Hierarchical micro-/nanostructured hydroxyapatite scaffolds promote osteoporotic bone regeneration via activation of hedgehog and HIF-1α signaling
Journal: Bioactive Materials (IF=20.3)
DOI: https://doi.org/10.1016/j.bioactmat.2026.01.049
Absin Products Used: Rabbit anti-PTCH1 Polyclonal Antibody (abs115174)

I. Innovative Research Strategy: "Customizing" Nanoscale Surfaces for Scaffold-Mediated Precision Activation of Repair Signaling
Traditional hydroxyapatite (HA) scaffolds, despite possessing osteoconductive properties, fail to simultaneously fulfill mechanical support and bioactivity requirements in osteoporotic microenvironments. The research team adopted an innovative approach by constructing hierarchical micro-/nanostructured hydroxyapatite (nwHA) scaffolds: utilizing whisker-reinforced hydroxyapatite (wHA) as the structural backbone to ensure mechanical integrity, followed by vacuum infiltration technology to coat five distinct nanohydroxyapatite (nHA) morphologies onto the surface, thereby achieving programmable regulation of nanoscale topographical architecture (Figure 1).
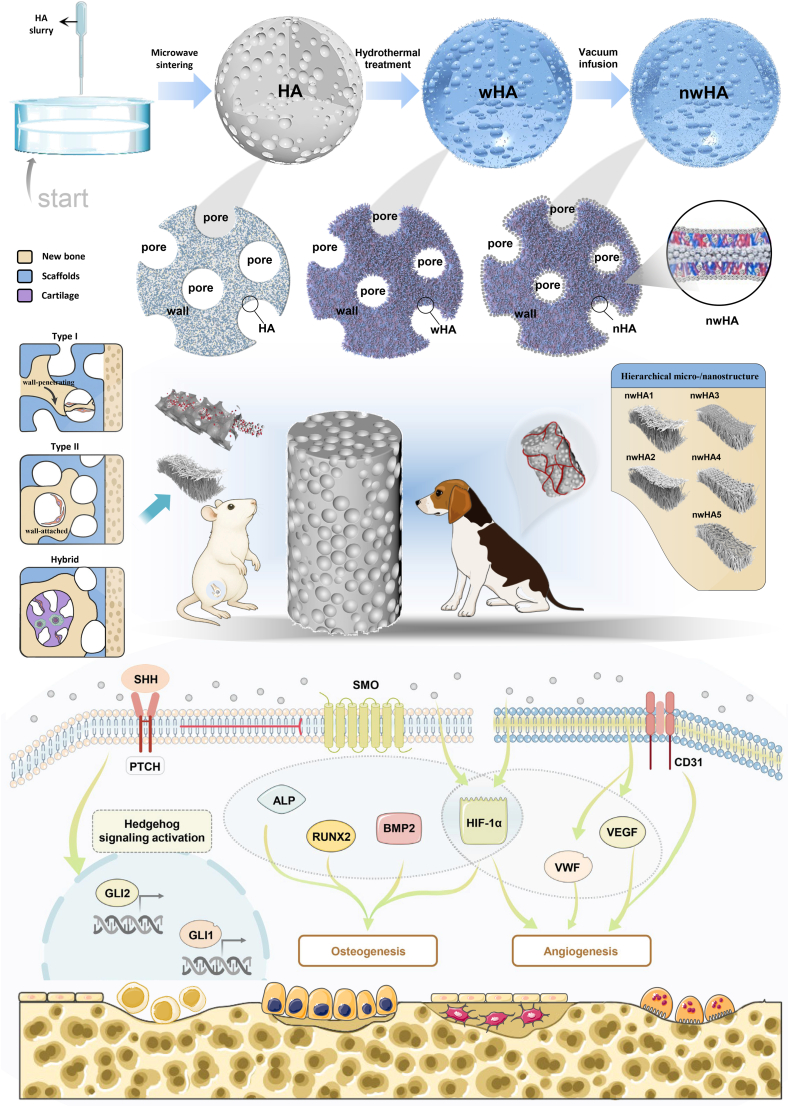 Diagram illustrating the preparation and proposed mechanism of action for nwHA scaffolds. Schematic showing how hierarchical micro-/nanostructured nwHA topographies enhance the osteogenic microenvironment in osteoporotic femoral defects. These topographies promote osteoprogenitor adhesion, vascular infiltration, and morphology-driven ossification modes—including wall-penetrating (type I), surface-appositional (type II), and hybrid patterns—thereby guiding context-specific intramembranous or endochondral bone regeneration.
Diagram illustrating the preparation and proposed mechanism of action for nwHA scaffolds. Schematic showing how hierarchical micro-/nanostructured nwHA topographies enhance the osteogenic microenvironment in osteoporotic femoral defects. These topographies promote osteoprogenitor adhesion, vascular infiltration, and morphology-driven ossification modes—including wall-penetrating (type I), surface-appositional (type II), and hybrid patterns—thereby guiding context-specific intramembranous or endochondral bone regeneration.
The core ingenuity of this design lies in decoupling the scaffold's "mechanical load-bearing" and "cellular regulation" functions. By screening for optimal nHA morphologies, the strategy specifically activates critical signaling pathways governing bone regeneration. The study focused on Hedgehog and HIF-1α signaling pathways, systematically investigating how nanoscale topography influences mesenchymal stem cell (MSC) osteogenic differentiation and human umbilical vein endothelial cell (HUVEC) angiogenesis, ultimately identifying a "precision-matched solution" for osteoporotic microenvironments.
II. Core Research Findings: Nanofiber-Coated Scaffold Emerges as Optimal Solution, Achieving Triple Breakthroughs in Repair Paradigms
Through in vitro cellular experiments and in vivo validation in osteoporotic rat models, the study achieved three key breakthroughs:
- 1. Morphology determines repair efficacy: Among five nHA morphologies, the nanofiber-coated scaffold (nwHA1) demonstrated the most prominent performance, significantly enhancing bone volume fraction, mineral apposition rate, mechanical strength, and neovascularization density. Its repair efficacy substantially surpassed that of uncoated wHA and other morphological scaffolds (Figures 6, 7).
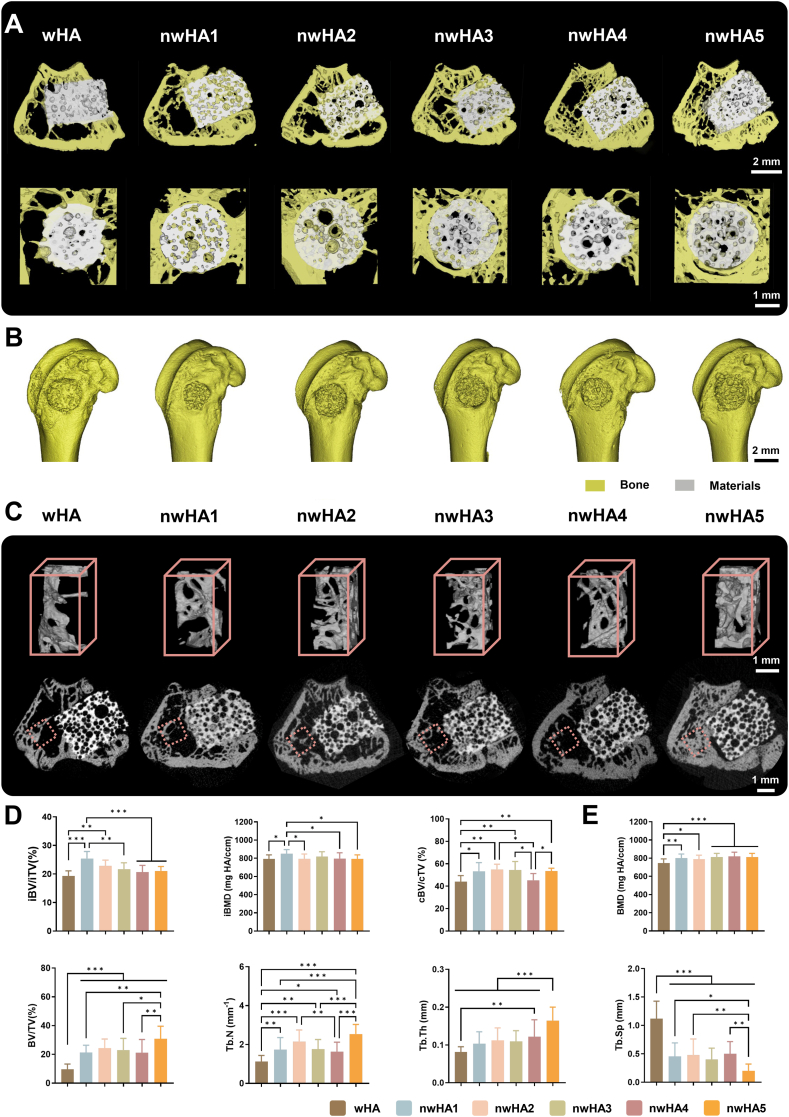
Micro-CT assessment of in vivo bone regeneration at 8 weeks. (A) 3D micro-CT reconstructions showing new bone formation and bone–scaffold interface (white: scaffold; yellow: bone); scale bar: 1 mm. (B) Whole femoral metaphysis reconstruction; scale bar: 2 mm. (C) Top: 3D renderings of trabecular ROI (red dotted box); bottom: corresponding 2D cross-sections at implant–bone interface; scale bar: 1 mm. (D) Quantification of intra-defect bone volume fraction (iBV/iTV), bone mineral density (iBMD), and concentric bone volume (cBV/cTV) within a 100 μm annular ROI; data are mean ± SD (n = 8; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001). (E) Histomorphometric analysis of trabecular microarchitecture parameters including BMD, BV/TV, BS/BV, Conn.Dens, Tb.Th, Tb.Sp, and SMI; data as mean ± SD (n = 8; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001).
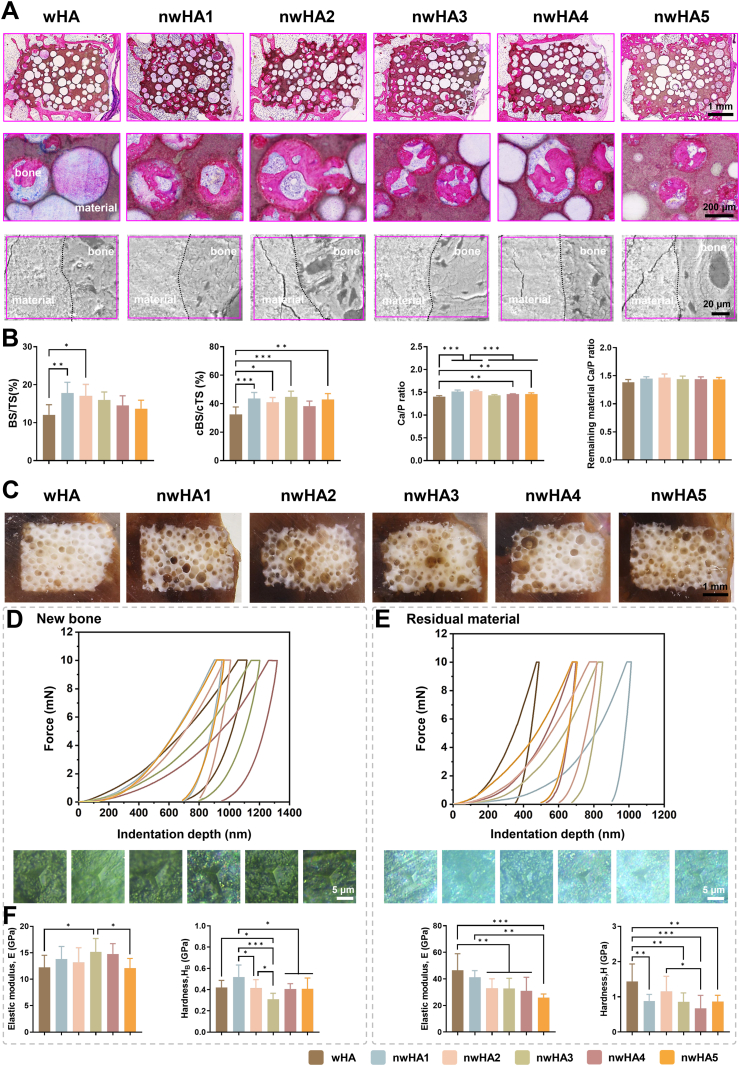
Histological and mechanical characterization of regenerated bone. (A) H&E-stained sections at 4 × and 20 × magnification, and SEM images showing bone–scaffold interfaces; scale bars: 1 mm (4 × ), 200 μm (20 × ), 20 μm (SEM). (B) Quantification of bone surface (BS/TS), contact surface (cBS/cTS) within 100 μm interface zone, and Ca/P ratios (EDS) in newly formed bone vs. residual scaffold; data as mean ± SD (n ≥ 8). (C) Optical imaging of PMMA-embedded undecalcified sections (B: bone, brown; M: material, white); scale bar: 1 mm. (D) Nanoindentation curves, microscopy images, and quantification of elastic modulus and hardness in newly formed bone; scale bar: 5 μm. (E) Corresponding mechanical analysis of undegraded material; all data as mean ± SD (n ≥ 8; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001).
- 2. Unlocking three ossification modes: Through histological analysis, the study first definitively established that nHA morphology and microenvironment co-regulate three distinct ossification patterns—Type I (wall-penetrating), Type II (surface-appositional), and hybrid endochondral-intramembranous ossification modes. Notably, nwHA1 more readily induced efficient Type I ossification (Figure 8).
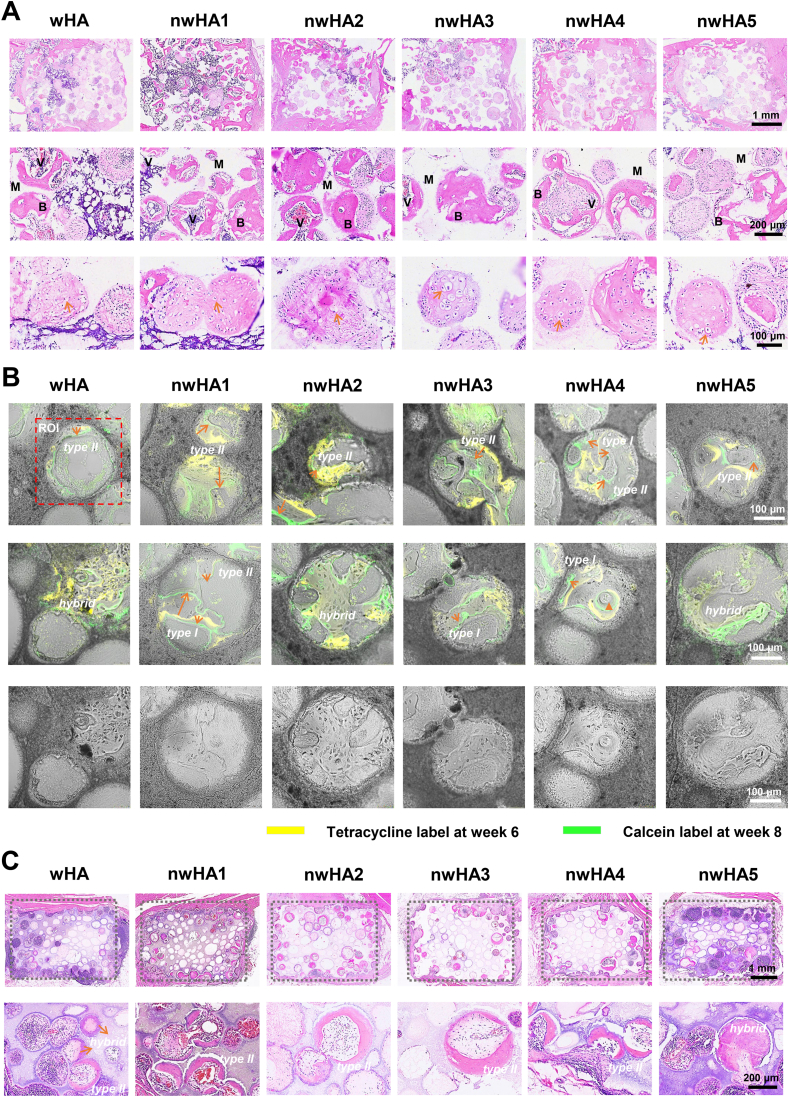
Ossification modes and mineralization dynamics in orthotopic and ectopic models. (A) Decalcified H&E-stained sections at 8 weeks, showing chondrocyte-like clusters at 4 × , 20 × , and 40 × magnification; scale bars: 1 mm (4 × ), 200 μm (20 × ), 100 μm (40 × ). (B) Sequential fluorochrome labeling (tetracycline: red, 6 weeks; calcein: green, 8 weeks) showing type I (wall-penetrating, bridging), type II (surface-appositional, concentric), and hybrid ossification with cartilage-like matrix features. The red dashed rectangle marks the regions of interest (ROI) used for mineral apposition rate (MAR) calculation, which evaluates the kinetics of mineralization by measuring the distance between the two fluorescent labels and dividing by the 14-day interval between the two injections. Scale bar: 100 μm. (C) H&E-stained sections of beagle dorsal muscle implants (12 weeks), demonstrating predominantly type II and hybrid ossification at 4 × and 20 × magnification; scale bars: 1 mm (4 × ), 200 μm (20 × ).
- 3. Definitive signaling pathway mechanisms: nwHA1 activates the canonical Hedgehog signaling pathway (SHH-PTCH1-GLI1 axis) and upregulates HIF-1α expression, simultaneously promoting osteogenesis and angiogenesis in MSCs and HUVECs, with bidirectional cross-regulation between the two pathways (Figures 3, 5).
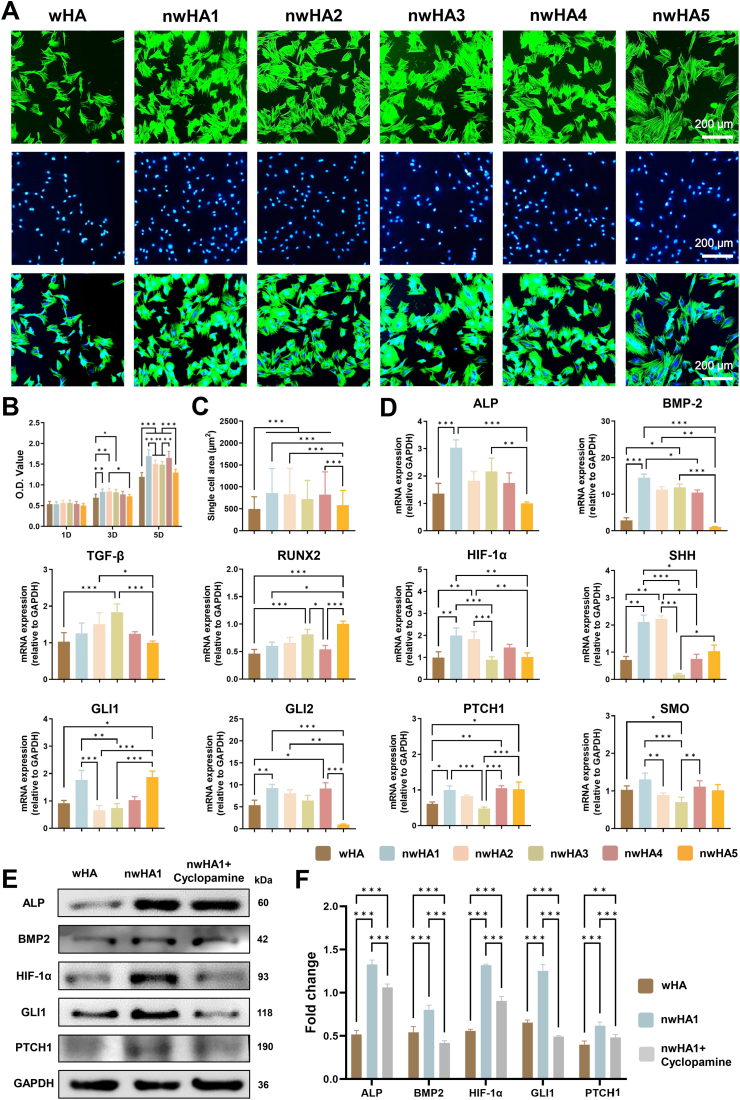
In vitro evaluation of biocompatibility and osteogenic activity of wHA and nwHA scaffolds using MSCs. (A) Fluorescence staining for F-actin (phalloidin-TRITC, green) and nuclei (DAPI, blue), showing enhanced cell spreading. (B) CCK-8 proliferation assay; data presented as mean ± SD (n = 8, ∗p < 0.05, one-way ANOVA with Tukey's post hoc test). (C) Quantification of single-cell area; data presented as mean ± SD (n ≥ 10, ∗p < 0.05, one-way ANOVA with Tukey's post hoc test). (D) qRT-PCR of osteogenic and Hedgehog-related markers (ALP, BMP2, Runx2, HIF-1α, SHH, PTCH1, SMO, GLI1, GLI2) at day 2; data presented as mean ± SD (n = 6, ∗p < 0.05, Kruskal–Wallis with Dunn's post hoc test, when applicable). (E) Western blot and (F) corresponding densitometric quantification of ALP, BMP2, HIF-1α, and GLI1 expression (n > 6, ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001, one-way ANOVA with Tukey's post hoc test).
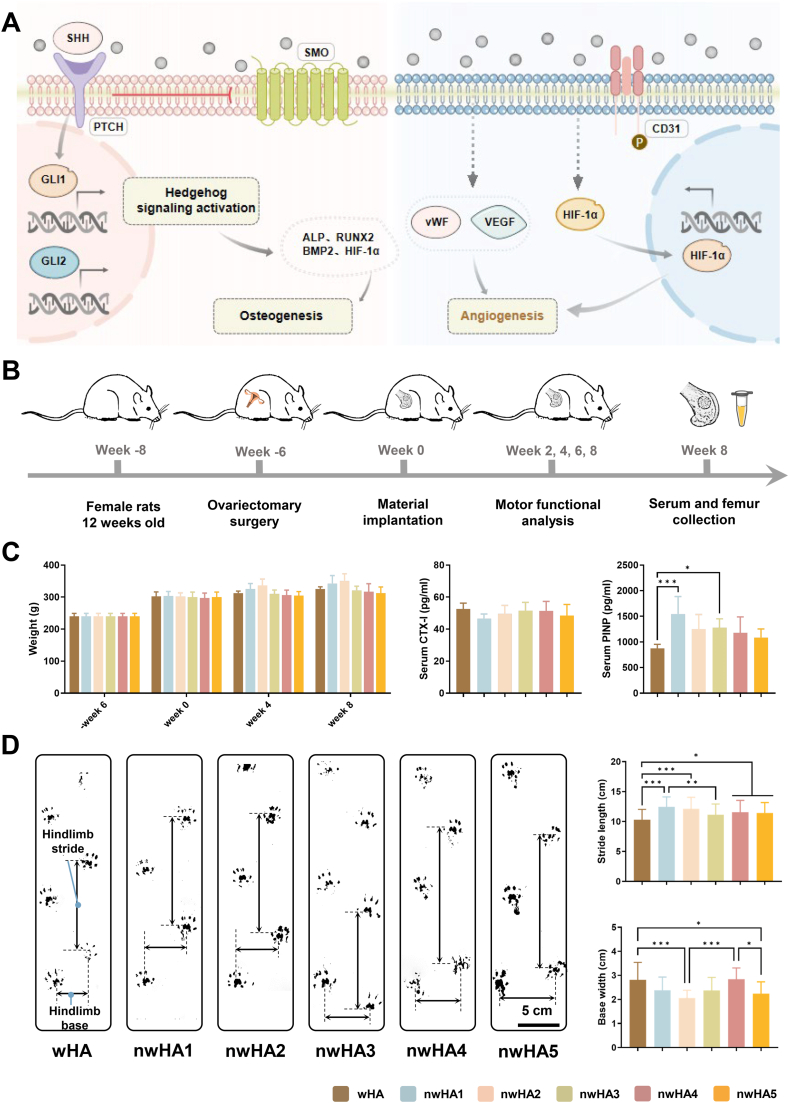
In vivo bone regeneration and functional recovery in osteoporotic rat femoral defects. (A) Schematic illustrating nwHA1-induced osteogenesis via Hedgehog activation in MSCs and pro-angiogenic effects in HUVECs through HIF-1α upregulation. (B) Surgical implantation of wHA and nwHA scaffolds (nwHA1–nwHA5) into distal femoral defects; evaluation at 8 weeks. (C) Body weight and serum markers (CTX-I, PINP) at 8 weeks; data shown as mean ± SD (n = 5; ∗p < 0.05, ∗∗∗p < 0.001, Kruskal–Wallis with Dunn's post hoc test). (D) Gait analysis quantifying stride length and base width as indicators of functional recovery; data shown as mean ± SD (n ≥ 8; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001; one-way ANOVA with Tukey's post hoc test).
III. Absin Product Empowerment: PTCH1 Antibody Enables Precise Validation of Critical Signaling Pathway Components
In the core mechanistic validation experiments—Western blot analysis—the research team utilized Absin's PTCH1 antibody (Cat. No.: abs115174) to successfully complete protein-level validation of Hedgehog signaling pathway activation.
Core Contributions of Absin Products in This Research:
- 1. Corroboration of signaling pathway activation: By detecting PTCH1 protein expression changes, the study definitively demonstrated that nwHA1 significantly upregulates key Hedgehog pathway molecules (including PTCH1, GLI1, etc.), directly proving the activating effect of nanofiber topography on the pathway (Figures 3E, 3F).
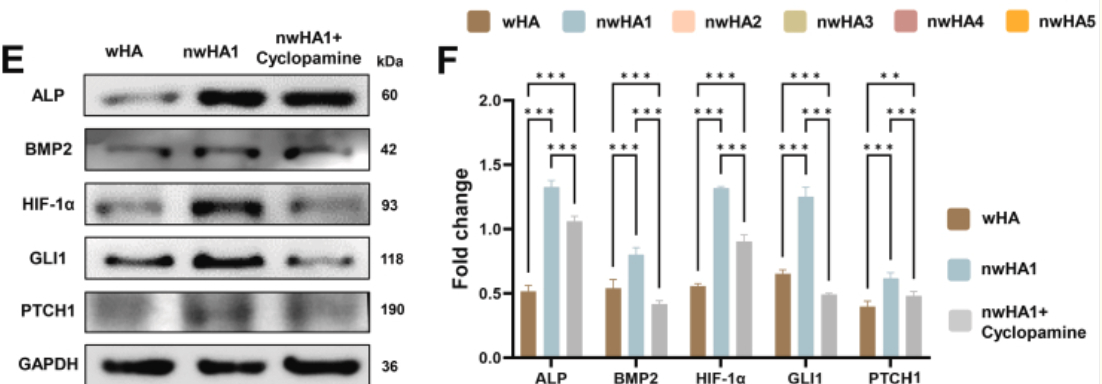
In vitro evaluation of biocompatibility and osteogenic activity of wHA and nwHA scaffolds using MSCs. (E) Western blot and (F) corresponding densitometric quantification of ALP, BMP2, HIF-1α, and GLI1 expression (n > 6, ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001, one-way ANOVA with Tukey's post hoc test).
- 2. Mechanistic specificity validation: In cyclopamine (Hedgehog pathway inhibitor) intervention experiments, Absin PTCH1 antibody clearly demonstrated that inhibitor treatment significantly downregulated PTCH1 protein expression, concomitantly reducing expression of osteogenic markers (ALP, BMP2) and HIF-1α, confirming that the Hedgehog pathway serves as the core mediator of scaffold-mediated repair effects.
- 3. Data reliability assurance: The antibody's high specificity and sensitivity ensured clear resolution and quantitative accuracy of protein bands in Western blot experiments, providing robust data support for the causal relationship between "pathway activation—osteogenesis/angiogenesis."
In Figure 3E, clear differences in PTCH1 protein expression among wHA, nwHA1, and nwHA1 + cyclopamine groups are presented. The precise recognition capability of Absin antibody renders pathway regulatory effects immediately apparent, serving as critical visual evidence for mechanistic argumentation.
IV. Industry Significance and Absin Commitment: Advancing Bone Repair Research Through Premium Research Tools
The publication of this study not only establishes nanoscale topography as a core "programmable regulatory factor" for osteoporotic bone repair but also provides clear direction for clinical translation of bioceramic scaffolds. As a reliable partner in life science research, Absin remains committed to the mission of "providing high-quality research tools." Its antibody products, distinguished by high specificity and sensitivity advantages, have been widely applied in critical experimental scenarios including signaling pathway validation and protein expression analysis.
From fundamental research to clinical translation, Absin will continue focusing on research needs in bone regeneration, osteoporosis, and related fields, providing one-stop solutions encompassing antibodies, reagents, and consumables. We are dedicated to assisting more research teams in overcoming technical bottlenecks, advancing life science research toward clinical application, and injecting robust momentum into human health initiatives.
This article is based on the original publication in Bioactive Materials (DOI: 10.1016/j.bioactmat.2026.01.049), interpreted and organized by AI; all original figures, data, and intellectual property rights belong to the original journal and research team. Should any infringement occur, please contact us promptly for removal, and we will actively cooperate in handling such matters.
|
Item NO. |
Product Name |
Size |
| abs115174 | Rabbit anti-PTCH1 Polyclonal Antibody | 50ul |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
