- Cart 0
- English
Breakthrough Discovery: KDM6B-Pdk1-ZEB2 Lactylation Axis Regulates Cementum Regeneration—Mechanistic Insights Enabled by Absin
March 12, 2026
Clicks:75
Periodontitis, a highly prevalent chronic inflammatory disease worldwide, ultimately leads to cementum destruction and irreversible tooth loss, while cementum regeneration represents the core component of periodontal tissue repair. Recently, a landmark study published in International Journal of Oral Science has, for the first time, elucidated the critical mechanism by which epigenetic-metabolic crosstalk regulates cellular cementum formation, providing novel therapeutic targets for periodontal regenerative therapy. Notably, the Absin CoIP Kit played a pivotal role in the core mechanistic validation of this study, enabling the research team to overcome technical bottlenecks.
Journal: International Journal of Oral Science (IF=12.2)
DOI: https://doi.org/10.1038/s41368-025-00420-5
Absin Products Used: Immunoprecipitation (IP/CoIP) Kit (abs955)


I. Research Strategy: Systematic Dissection of the Molecular Code Underlying Cementum Regeneration
The research team established a logically rigorous investigative framework centered on "core regulatory mechanisms of cementogenesis":
- Target Screening: Through Gene Ontology (GO) enrichment analysis, the histone lysine demethylase family (KDMs) was identified as significantly enriched during cementoblast mineralization. Combined with prior research foundations, KDM6B was selected as the key candidate molecule;
- Functional Validation: Through in vivo and in vitro inhibition/knockdown experiments, the positive regulatory role of KDM6B in cellular cementum formation and cementoblast mineralization was definitively established;
- Mechanistic Elucidation: Focusing on the association between KDM6B and glucose metabolism, the glycolytic key enzyme Pdk1 was identified as a direct target gene through Seahorse metabolic assays and ChIP-seq technologies;
- Downstream Extension: Investigating the role of the glycolytic metabolite lactate, it was discovered that lactate promotes the expression of mineralization-related genes through mediating lactylation modification of the transcription factor ZEB2.
The entire study establishes a complete regulatory cascade of "epigenetic regulator (KDM6B) → metabolic pathway (glycolysis) → post-translational protein modification (lactylation) → functional phenotype (cementum formation)", progressively revealing the core mechanism layer by layer.
II. Core Findings: Unlocking the Key Regulatory Axis of Cementum Regeneration
1. KDM6B is an Essential Activator of Cellular Cementum Formation
In vivo experiments demonstrated that following KDM6B inhibitor GSK-J4 treatment, mice exhibited thinning of the dentin-cementum complex and significant reduction in cellular cementum area (Fig. 2c-d), with decreased expression of mineralization markers OSX and OCN (Fig. 2e-f); in vitro experiments confirmed that KDM6B knockdown significantly impaired cementoblast mineralization capacity.
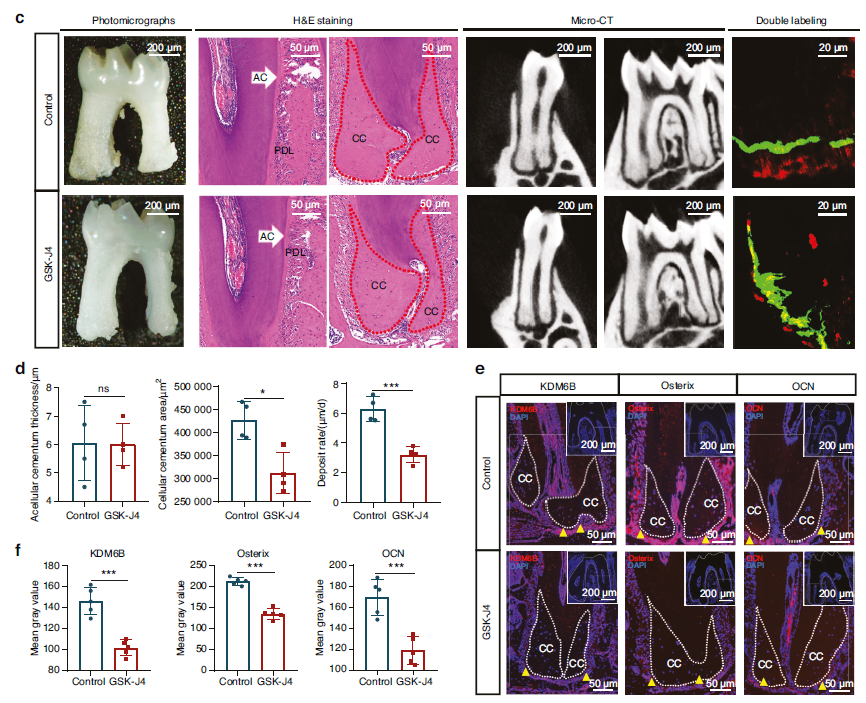
Fig. 2 KDM6B positively governs cellular cementum formation and cementoblast mineralization. a, b GO enrichment and heatmap results of cementogenesis-related gene expression from RNA sequencing analysis of cementoblasts following KDM6B inhibition. c, d Microphotography, hematoxylin and eosin staining (H&E) staining (n = 4), micro-CT and double labeling (n = 4) results of apical cementum in control and GSK-J4 groups of mice. CC, cellular cementum; AC, acellular cementum; PDL, periodontal ligament. (The area of AC and CC displayed in H&E staining originates from the same specimen within each group.) e, f Immunofluorescence (IF) analysis of KDM6B, OSX, and OCN expression levels in cementoblasts of control and GSK-J4 groups (n = 5). CC cellular cementum. Data are presented as mean ± SD. **P < 0.01. ***P < 0.001. ****P < 0.000 1
2. The KDM6B-Pdk1 Axis Drives Glycolytic Reprogramming in Cementoblasts
ChIP-seq validation confirmed that KDM6B directly activates Pdk1 transcription through H3K27me3 demethylation (Fig. 4a-b); following KDM6B knockdown, expression of glycolytic key markers LDHA and PDK1 decreased, with cellular metabolic patterns shifting from aerobic glycolysis toward oxidative phosphorylation (Fig. 3a-e).
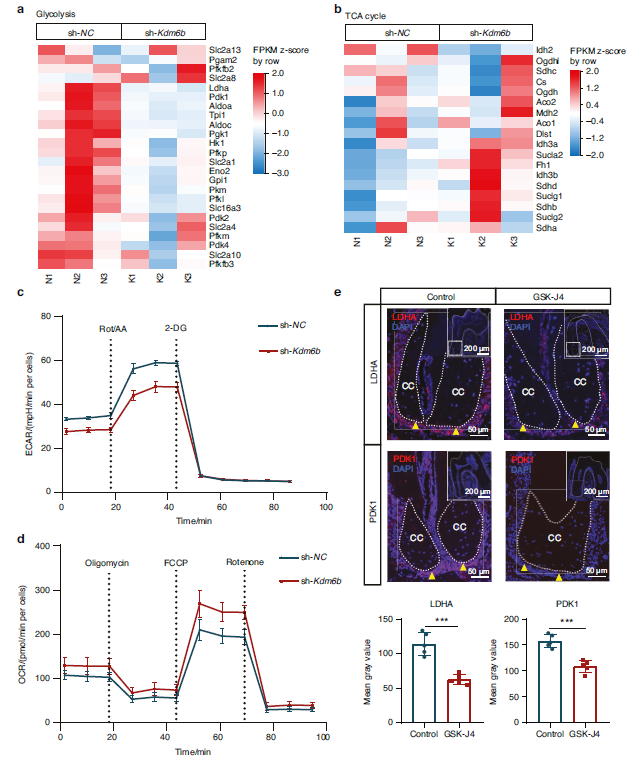
Fig. 3 KDM6B positively governs the glycolytic process of cementoblasts. a, b Heatmap of glycolysis and TCA cycle-related genes expression of cementoblasts following KDM6B inhibition. c Seahorse examinations of extracellular acidification rate (ECAR). d Seahorse examinations of oxygen consumption rate (OCR). e IF analysis of LDHA and PDK1 expression levels in cementoblasts of control and GSK-J4 groups (n = 5). Data are presented as mean ± SD. *P < 0.05. **P < 0.01. ***P < 0.001. ****P < 0.000 1
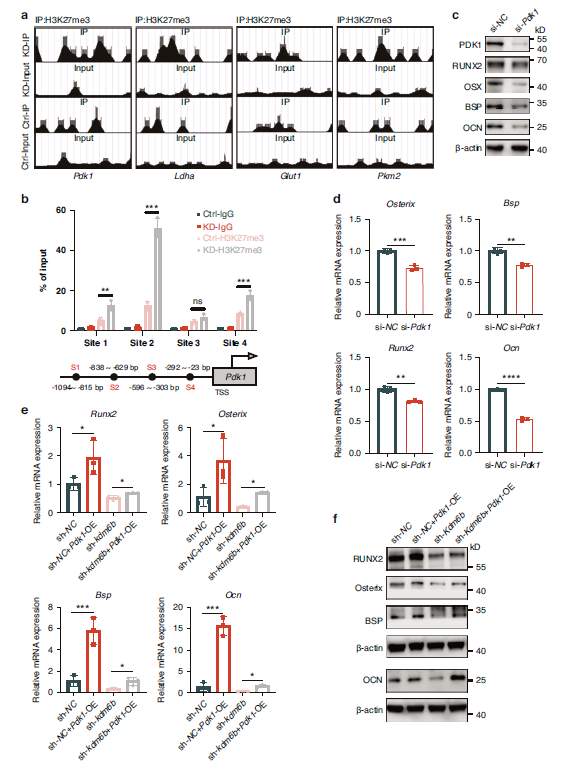
Fig. 4 KDM6B regulates cementoblast mineralization by targeting Pdk1. a Chromatin Immunoprecipitation (ChIP) sequencing detection of H3K27me3 peaks at 1 000 bp upstream of the transcription start sites of glycolysis-related markers under KDM6B inhibition. b By dividing Pdk1 upstream into 4 sites, ChIP-qPCR shows that inhibiting Kdm6b could upregulate H3K27me3 in these 4 sites (n = 3). c, d QRT-PCR and WB results indicate reduced mineralization levels after transient transfection with si-Pdk1 (n = 3). e, f QRT-PCR and WB assays reveal rescue of the mineralization phenotype following Pdk1 overexpression under Kdm6b inhibition. Data are presented as mean ± SD. *P < 0.05. **P < 0.01. ***P < 0.001. ****P < 0.000 1
3. ZEB2 Lactylation Represents the Core Downstream Event in Mineralization Regulation
Through lactylome profiling, three lactylation sites on ZEB2 were identified (K485, K762, K993), with the K485 site being critical for mineralization regulation (Fig. 6a-b); PDK1 enhances ZEB2 lactylation through promoting lactate generation, thereby upregulating the expression of mineralization genes including RUNX2 and BSP (Fig. 6c-e).
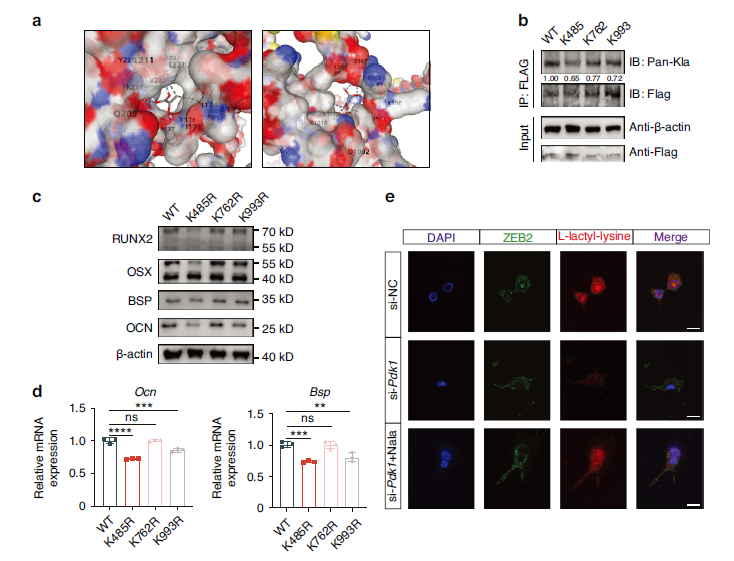
Fig. 6 PDK1-ZEB2 lactylation enhances cementoblast mineralization. a Docking Results of ZEB2 and L-Lactate. b Co-immunoprecipitation (Co- IP) confirms the occurrence of lactylation modifications. c, d Mineralization levels detected by qRT-PCR (n = 3) and WB after mutating lysine (K) to arginine (R). e IF results reveal that ZEB2 lactylation levels were rescued by lactate sodium addition under Pdk1 inhibition. Nala, lactate sodium. Data are presented as mean ± SD. Scale bars: 30 μm, **P < 0.01. ***P < 0.001. ****P < 0.000 1
4. Sodium Lactate Supplementation Rescues Mineralization Defects Caused by KDM6B Inhibition
In vivo ectopic mineralization models demonstrated that sodium lactate supplementation significantly restored mineralized nodule formation and collagen fiber synthesis in the KDM6B knockdown group (Fig. 7c), and elevated tissue lactylation levels (Fig. 7d), providing potential strategies for clinical translation.
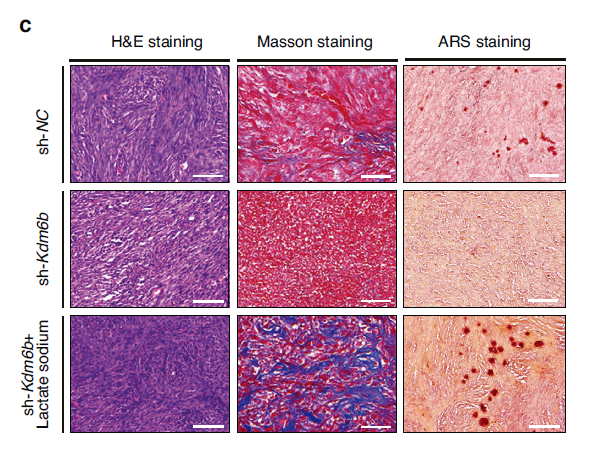
Fig. 7 Elevated lactylation recovers KDM6B inhibition-mediated cementogenesis downregulation in vivo. c H&E, Masson and ARS staining results for different groups.
III. Absin Products: Critical Tool Support for Mechanistic Validation
In this high-impact study, the Absin Co-Immunoprecipitation (Co-IP) Kit (abs955) served as the core tool for validating ZEB2 lactylation modifications, providing essential technical support for mechanistic breakthroughs.
Product Application: Precise Capture of Lactylation Modification Complexes
To confirm the presence of lactylation modifications on ZEB2, the research team utilized the Absin Co-IP Kit to specifically enrich ZEB2 protein complexes through Flag-tag antibody immunoprecipitation, followed by lactylation antibody detection, definitively establishing the authenticity of ZEB2 lactylation modifications (Fig. 6b). Furthermore, this kit facilitated validation of the effects of K485, K762, and K993 site mutations on ZEB2 lactylation levels, providing direct experimental evidence for identifying critical regulatory sites.
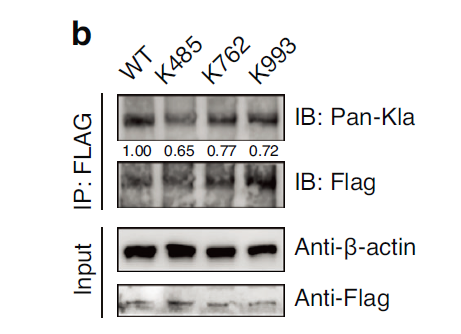
Fig. 6 PDK1-ZEB2 lactylation enhances cementoblast mineralization. b Co-immunoprecipitation (Co- IP) confirms the occurrence of lactylation modifications.
Product Advantages: Empowering Precision Mechanistic Analysis
The Absin Co-IP Kit, with its high-specificity antibody binding efficiency and low background interference characteristics, efficiently enriches target proteins and their modification complexes, avoiding result deviations caused by non-specific binding. In this study, the product successfully captured low-abundance ZEB2 lactylation complexes, laying a solid foundation for subsequent Western blot validation and functional analysis, serving as the critical experimental tool bridging "metabolic products" and "functional protein modifications".
IV. Research Significance and Future Perspectives
This study represents the first demonstration of protein lactylation modification in cementum formation, providing multiple potential therapeutic targets including KDM6B, Pdk1, and ZEB2 for periodontal regenerative therapy, while the intervention strategy of sodium lactate supplementation also offers novel insights for clinical application. Furthermore, the study establishes an innovative research paradigm of epigenetic-metabolic crosstalk for the bone regeneration field, possessing broad interdisciplinary reference value.
Absin remains committed to providing high-quality tools for life science research, comprehensively empowering scientific innovation from protein-protein interactions, signal pathway analysis to post-translational modification detection. Moving forward, Absin will continue to deepen its engagement with research needs, providing more comprehensive experimental solutions to facilitate breakthroughs in basic research and clinical translation.
This content is based on the original publication in International Journal of Oral Science (DOI: 10.1038/s41368-025-00420-5), interpreted and compiled by AI; all original figures, data, and intellectual property rights belong to the original journal and research team. Should any infringement occur, please contact us promptly for removal, and we will actively cooperate in addressing such matters.
|
Item NO. |
Product Name |
Size |
| abs955 | Immunoprecipitation (IP/CoIP) kit | 50T |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
