worldwide@absin.cn
- Cart 0
- English
Why Are Your Cell Culture Results Inconsistent and Irreproducible?
March 05, 2026
Clicks:78
In cell culture laboratories, there exists an "invisible killer" that frustrates researchers: cells appear normal with no turbidity, no floating debris, and no bacteria or fungi visible under the microscope, yet experimental results fluctuate unpredictably like "rolling dice." Western blot band intensities vary erratically, flow cytometry apoptosis rates show enormous batch-to-batch variation, and cell transfection efficiency declines inexplicably. More terrifyingly, such problems often persist for months before being discovered, wasting countless reagents, time, and samples during this period. This "invisible killer" is Mycoplasma contamination. The Mycoplasma Staining Detection Kit serves as the "magic mirror" that exposes this latent enemy.
What Exactly is a Mycoplasma Staining Detection Kit?
The Mycoplasma Staining Detection Kit is a DNA fluorescent dye-based cell culture contamination detection system specifically designed for in situ detection of Mycoplasma or other prokaryotic contamination in cultured cells. It utilizes Hoechst fluorescent dye that binds specifically to DNA, allowing observation of nuclear and extracellular DNA presence under fluorescence microscopy to determine Mycoplasma contamination status.
From a technical composition perspective, the complete kit contains three core components:
Fixative Solution (Component A): Contains glacial acetic acid fixative solution for rapidly killing cells while preserving cellular structural integrity, and enabling Mycoplasma and other prokaryotes to attach to the cell surface
Hoechst Staining Solution (Component B): Specific DNA-binding dye capable of penetrating cell membranes to bind to DNA minor grooves, emitting blue fluorescence under UV excitation
Antifade Mounting Medium (Component C): Glycerol buffer containing antifade reagents for mounting and preservation, preventing rapid fluorescence quenching to facilitate extended observation and documentation
Hoechst Staining Solution (Component B): Specific DNA-binding dye capable of penetrating cell membranes to bind to DNA minor grooves, emitting blue fluorescence under UV excitation
Antifade Mounting Medium (Component C): Glycerol buffer containing antifade reagents for mounting and preservation, preventing rapid fluorescence quenching to facilitate extended observation and documentation
This integrated "fixation-staining-mounting" design standardizes and ensures reproducibility of the detection process. One six-well plate sample detection kit can test at least 100 samples, suitable for routine laboratory screening.
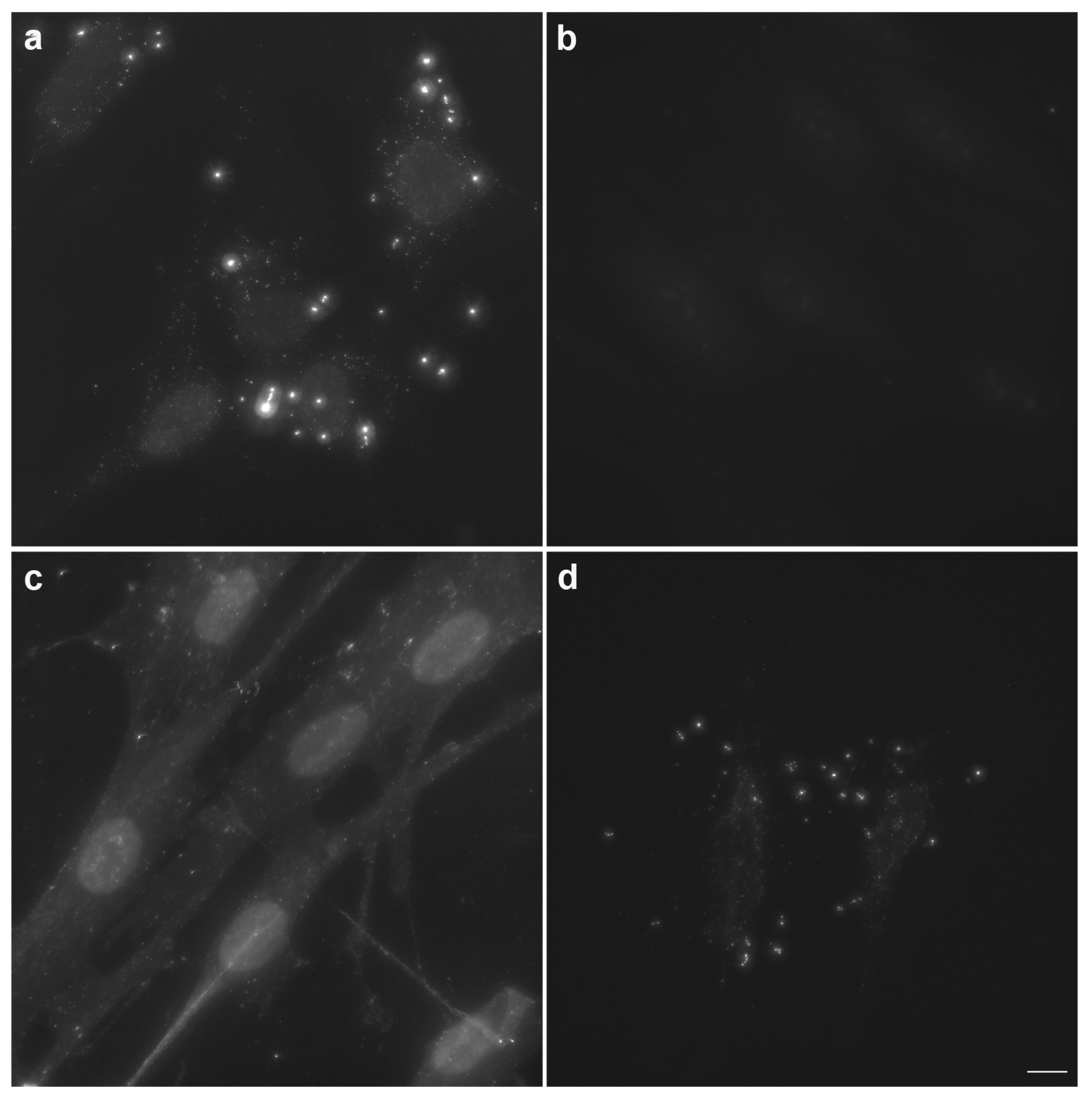
Figure 1: Typical results of Hoechst staining for Mycoplasma detection. Normal cells (a) show only nuclear blue fluorescence; Mycoplasma-contaminated cells (b-d) exhibit numerous punctate or filamentous blue fluorescent particles surrounding the nucleus, representing Mycoplasma DNA signals. Higher contamination levels correlate with increased density of extracellular fluorescent particles.
Why is Mycoplasma Called the "Invisible Killer" of Cell Culture?
The "Invisible Man" Under Optical Microscopy
Unlike bacteria, yeast, or mold, Mycoplasma:
- Cannot be observed by conventional microscopy: Size is below the optical microscopy resolution limit (~0.2 μm) and does not cause culture medium turbidity
- Does not form colonies: Grows slowly on solid medium (2-4 weeks) without forming visible colonies to the naked eye
- Does not cause pH changes: Slow metabolism does not significantly alter medium color (unlike bacterial contamination causing yellow color change)
This "invisible" characteristic means contamination may persist undetected for months until systematic bias appears in experimental results.
Widespread Interference with Cellular Functions
Although Mycoplasma does not produce acute cytotoxicity, its chronic presence severely interferes with cellular physiology:
- Nutrient consumption: Competes with cells for amino acids, nucleotides, and other nutrients in culture medium
- Metabolic alteration: Induces cytokine expression and alters cell surface receptor expression profiles
- Signal transduction interference: Affects key signaling pathways including MAPK and NF-κB
- Cell cycle alteration: Causes slowed cell growth and abnormal apoptosis rates
- Chromosomal abnormality induction: Causes chromosomal breakage, aneuploidy, and other genetic instabilities
These effects are often "dose-dependent" and "time-dependent"; mild contamination may not immediately manifest, but cellular functions gradually deviate from normal states with increasing passage numbers.
Systematic Bias in Experimental Results
Mycoplasma contamination affects specific experiments as follows:
- Western Blot: Elevated background, band smearing, inaccurate quantification
- Flow Cytometry: Enhanced cellular autofluorescence, false positives in apoptosis detection
- Transfection Experiments: Decreased transfection efficiency, abnormal gene expression levels
- Virus Packaging: Reduced viral titers, compromised packaging efficiency
- Drug Screening: Altered cellular drug sensitivity, increased false negatives or false positives
How Do Dedicated Kits Solve Detection Challenges?
Specific Recognition by Hoechst DNA Dye
Hoechst 33258 is a bisbenzimidazole dye capable of:
- Penetrating intact cell membranes: Can stain live cell DNA without fixation, though signal is stronger after fixation
- High-affinity DNA binding: Intercalates into DNA minor grooves, binding to AT-rich regions with fluorescence intensity proportional to DNA quantity
- Distinguishing DNA from RNA: Affinity for double-stranded DNA is significantly higher than for RNA
Although Mycoplasma is small, its genomic DNA (~0.5-1 Mb) is sufficient for Hoechst staining, appearing as punctate or filamentous fluorescent particles surrounding the nucleus under fluorescence microscopy.
Spatial Information from In Situ Staining
Unlike PCR methods, staining detection provides spatial localization information:
- Normal cells: Show uniform blue fluorescence only in the nucleus; mitochondrial DNA (mtDNA) is not stained (low copy number and encapsulated within mitochondria)
- Mycoplasma contamination: Numerous tiny fluorescent particles appear surrounding the nucleus, distributed uniformly or aggregated into filamentous patterns
- Bacterial/fungal contamination: Visible rod-shaped, spherical, or filamentous large particles, significantly larger than Mycoplasma, often arranged in chains or clusters
These morphological characteristics allow researchers to preliminarily distinguish Mycoplasma from other microbial contamination.
Optimization of Fixation Technology
The glacial acetic acid component in the fixative solution can:
- Rapidly penetrate cells: Instantly halt cellular metabolism while preserving cell morphology
- Enable Mycoplasma attachment: Alters Mycoplasma surface charges, causing firm adhesion to cell surfaces to prevent loss during washing
- Enhance DNA accessibility: Moderately denatures DNA to improve Hoechst binding efficiency
Which Experimental Scenarios Require Mandatory Mycoplasma Testing?
Cell Line Banking and Authentication
This is the most fundamental application scenario for Mycoplasma testing. Whether establishing new cell lines, purchasing commercial cell strains, or receiving cells from collaborating laboratories, Mycoplasma testing is mandatory:
- New cell line establishment: Primary culture or immortalization processes are highly susceptible to Mycoplasma introduction
- Commercial cell strains: Although suppliers claim testing compliance, contamination may occur during transportation
- Inter-laboratory transfer: Cells circulating among multiple laboratories carry extremely high cross-contamination risk
It is recommended that all cell lines undergo testing before liquid nitrogen preservation, with establishment of "Mycoplasma-free certificates" documentation.
Routine Cell Culture Monitoring
Mycoplasma contamination can occur at any stage:
- Incubator environment: Humidified incubators are breeding grounds for Mycoplasma
- Operator transmission: Humans are natural hosts for Mycoplasma (e.g., Mycoplasma orale)
- Reagent contamination: Animal-derived reagents such as fetal bovine serum and trypsin may carry Mycoplasma
It is recommended to perform regular screening of actively used cell lines every 1-2 months, or immediate testing when experimental results show abnormalities.
Troubleshooting Abnormal Experimental Data
When the following situations occur in experiments, Mycoplasma contamination should be considered first:
- Slow cell growth: Extended doubling time with normal morphology
- Elevated background signals: Turbid backgrounds in Western blot, immunofluorescence, and other experiments
- Decreased transfection efficiency: Sudden reduction in plasmid transfection or viral infection efficiency
- Altered drug sensitivity: Cellular responses to chemotherapeutic agents inconsistent with literature reports
- Abnormal cell behavior: Unstable results in migration, invasion, apoptosis, and other functional assays
Cell Therapy and Biologics Manufacturing
In GMP-grade cell therapy product manufacturing, Mycoplasma testing is a mandatory safety testing item:
- CAR-T cell therapy: Mycoplasma contamination in patient-derived T cells may lead to severe adverse reactions
- Stem cell therapy: Mesenchymal stem cell culture involves long periods with high contamination risk
- Biologics: Cell banks used for vaccine and antibody production must be Mycoplasma-negative
Although staining detection is less sensitive than PCR methods, as a rapid screening tool it can detect early contamination promptly, avoiding large-scale production losses.
Quality Control Before Manuscript Submission
An increasing number of journals require cell line authentication and Mycoplasma testing reports. Conducting Mycoplasma testing before submission:
- Meets journal requirements: Top-tier journals including Cancer Research and Nature series have such requirements
- Ensures data credibility: Eliminates experimental bias caused by Mycoplasma contamination
- Avoids retraction risk: Paper retraction cases due to cell contamination are increasingly common
How to Correctly Use the Mycoplasma Staining Detection Kit?
Critical Details for Sample Preparation
Cell Density Control:
- Culture adherent cells to 50%-80% confluence, avoid over-confluence
- Overly dense cells make intercellular regions difficult to observe, easily missing mild contamination
- Collect suspension cells by centrifugation, smear on slides, and air dry thoroughly
Pretreatment Steps:
- Culture in antibiotic-free medium for 2-3 passages
- Antibiotics (such as penicillin) may inhibit Mycoplasma growth, causing false negatives
- Gently wash 1-2 times with PBS to remove serum and antibiotic residues
Staining Operation Procedure
Hoechst Working Solution Preparation:
- Dilute Hoechst staining solution 1:10 with PBS (1 part stain + 9 parts PBS)
- Use diluted stain within 24 hours, store protected from light
- If precipitation occurs, sonicate at 37°C or prepare fresh solution
Fixation Step:
- Add appropriate amount of fixative (1 mL per well for six-well plates) to ensure complete cell coverage
- Fix at room temperature for 10-20 minutes
- Critical: Do not wash cells before fixation to avoid washing away Mycoplasma attached to cell surfaces
Staining Step:
- Remove fixative and air dry (approximately 5-10 minutes)
- Add 10-fold diluted Hoechst staining solution (1 mL per well for six-well plates)
- Stain at room temperature protected from light for 10-30 minutes (wrap with aluminum foil for light protection)
- Adjust staining time according to cell type; extend appropriately for difficult-to-stain cells
Washing and Mounting:
- Remove staining solution and air dry
- Add antifade mounting medium and cover with coverslip
- Observe under fluorescence microscopy after mounting, or store at 4°C protected from light and observe within 24 hours
Microscopic Observation and Result Interpretation
Observation Conditions:
- Use fluorescence microscope with UV excitation (350-370 nm)
- Observe blue fluorescence emission (450-480 nm)
- Magnification: 400× or 1000× (oil immersion); low magnification cannot resolve Mycoplasma
Result Interpretation Criteria:
Differential Diagnosis:
- Mycoplasma: Tiny punctate particles (<0.5 μm), uniformly distributed, mainly around nucleus
- Bacteria: Rod-shaped or spherical (1-2 μm), often in chains or clusters, visible under optical microscopy
- Fungi/Yeast: Large spherical forms (5-10 μm) with budding phenomena, visible under optical microscopy
- Cell debris: Irregular shapes, no specific distribution pattern, distinguishable by DAPI/Hoechst double staining
Handling Positive Results
Emergency Measures:
- Immediately cease all experiments with the contaminated cell line
- Isolate contaminated cells to prevent cross-contamination of other cells
- Thoroughly disinfect incubators, work surfaces, centrifuges, and other equipment
Cell Salvage (Optional):
- Treat with Mycoplasma elimination reagents (such as Plasmocin) for 2-4 weeks
- Retest after treatment; resume experiments only after confirmation of negative status
- Simultaneously cryopreserve backup copies of important cell lines
Complete Disposal:
- General recommendation is to directly discard contaminated cells to avoid long-term risks of "incomplete elimination"
- Re-obtain cells from reliable sources and re-establish cell banks
From "Passive Detection" to "Active Prevention": Quality Management in Cell Culture
Mycoplasma staining detection should not only serve as a "diagnostic tool" but also become an integral component of cell culture quality management systems:
- Mandatory testing for new cell banking: Establish "Mycoplasma-free" admission protocols
- Regular screening system: Test active cell lines every 1-2 months
- Experimental abnormality investigation: First exclude Mycoplasma contamination when data anomalies occur
- Personnel training: Enhance awareness of Mycoplasma contamination symptoms
Conclusion
The Mycoplasma Staining Detection Kit represents a fundamental tool for cell culture quality control. In today's increasingly sophisticated cell biology research, eliminating interference from "invisible killers" like Mycoplasma is a prerequisite for obtaining reliable, reproducible experimental data. From routine cell culture monitoring to clinical-grade cell therapy product release, from basic research to translational applications, rapid, economical, and intuitive staining detection methods remain the first line of defense in Mycoplasma prevention and control. Mastering the correct use of this technology means establishing fundamental assurance for the reliability of cell experiments.
【Disclaimer】This article is generated from publicly available online information by AI. If it inadvertently involves infringement, please contact us promptly, and we will cooperate in handling it immediately without assuming any legal liability.
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
