- Cart 0
- English
Landmark Study | Absin Organoid Culture Reagents Enable Discovery of TIMELESS-Mediated Regulation of Ferroptosis and Immune Microenvironment in Lung Adenocarcinoma
February 25, 2026
Clicks:71
Lung cancer remains the leading cause of cancer-related mortality worldwide, with lung adenocarcinoma (LUAD) representing a major histological subtype. The clinical challenge of immune checkpoint inhibitor resistance in LUAD treatment remains unresolved. Recently, a research team from the Air Force Medical University published a landmark study in Cancer Communications (2026, 46: Article 0009), revealing for the first time that the RNA-binding protein TIMELESS recruits CNOT3 to mediate degradation of transferrin (TF) mRNA, thereby suppressing transferrin-mediated ferroptosis and reprogramming the tumor microenvironment, ultimately leading to PD-1 immunotherapy resistance in LUAD. This discovery provides novel therapeutic targets and combination strategies for LUAD treatment. Absin products played a critical role throughout this study, supporting in vitro model construction, cellular functional validation, and mechanistic exploration.
Journal:Cancer Communications (IF=24.9)
DOI:https://doi.org/10.34133/cancomm.0009
Absin Products Used: Human Lung Cancer Organoid Culture Medium Kit (abs9443), GFR OrganoGel Phenol red free (abs9495)


I. Research Strategy: Decoding the Pro-Oncogenic Mechanisms of TIMELESS Through Systematic Investigation
This study focused on aberrantly expressed RNA-binding proteins (RBPs) in LUAD, employing a classical research framework of clinical data screening - cellular functional validation - molecular mechanism elucidation - in vivo model verification - clinical translation exploration to comprehensively decode the role and mechanisms of TIMELESS in LUAD:
▸Core Target Identification:Integration of TCGA-LUAD, GEO, and other public datasets identified significantly upregulated RBPs in LUAD; functional knockdown experiments established TIMELESS as a critical target affecting LUAD cell viability;
▸Cellular Function Characterization:Construction of TIMELESS-knockout LUAD cell lines and patient-derived organoids (PDOs), combined with ferroptosis detection assays, confirmed that TIMELESS primarily promotes LUAD cell proliferation through ferroptosis inhibition;
▸Molecular Mechanism Elucidation:Utilization of PAR-CLIP-seq, RIP, and Co-IP technologies identified transferrin (TF) as a direct target of TIMELESS, revealing the molecular mechanism by which TIMELESS recruits CNOT3 to mediate TF mRNA degradation;
▸In Vivo Efficacy Validation:Establishment of LLC1 orthotopic lung cancer mouse models to validate the synergistic anti-tumor effects of TIMELESS knockdown combined with the ferroptosis inducer erastin and PD-1 inhibitors;
▸Clinical Correlation Analysis:Multiplex immunofluorescence analysis of LUAD tissue microarrays confirmed that high TIMELESS expression correlates with ferroptosis suppression, elevated PD-L1 expression, and poor prognosis in LUAD patients.
This rigorous research framework, progressing from clinical problems to clinical value, provides an exemplary model for translational research from basic science to clinical application.
II. Core Research Findings: TIMELESS/CNOT3/TF Axis as a Novel Therapeutic Target for LUAD
TIMELESS is significantly overexpressed in LUAD tumor tissues, with expression levels closely correlated with tumor size, AJCC stage, and T stage. LUAD patients with high TIMELESS expression exhibit significantly shortened overall survival (Fig. 2H), establishing TIMELESS as an independent prognostic indicator of poor outcome. Knockdown of TIMELESS significantly inhibits LUAD cell proliferation and induces cell death.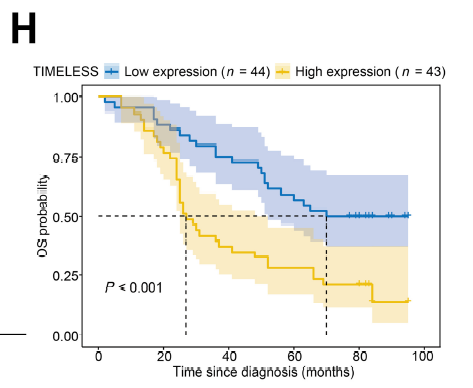
Transcriptomic sequencing revealed significant enrichment of ferroptosis pathways following TIMELESS knockdown (Fig. 2A); in A549, H1975 cells and PDO models, TIMELESS knockout significantly enhanced LUAD cell sensitivity to the ferroptosis inducer erastin, manifested by elevated ROS, lipid peroxidation, and intracellular iron levels, with mitochondria exhibiting characteristic ferroptosis-associated morphological changes (Fig. 3).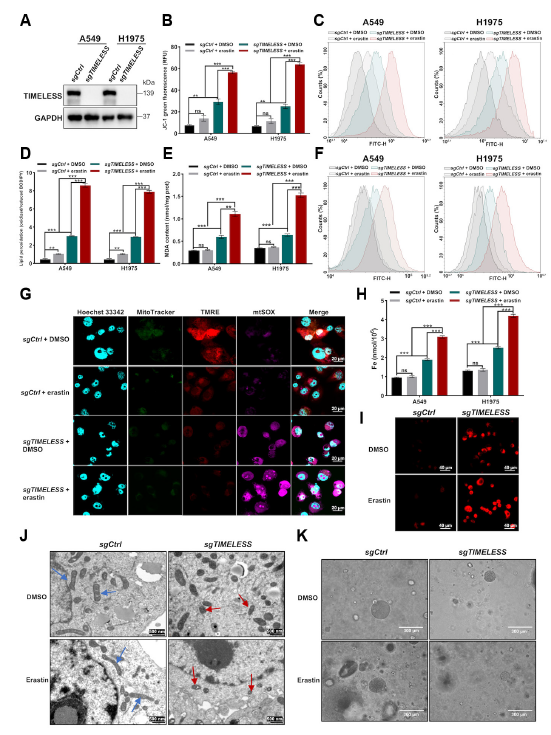
PAR-CLIP-seq identified transferrin (TF) as a direct target of TIMELESS, with binding occurring at the GAGATGG motif within the TF 3'UTR (Fig. 4D-E);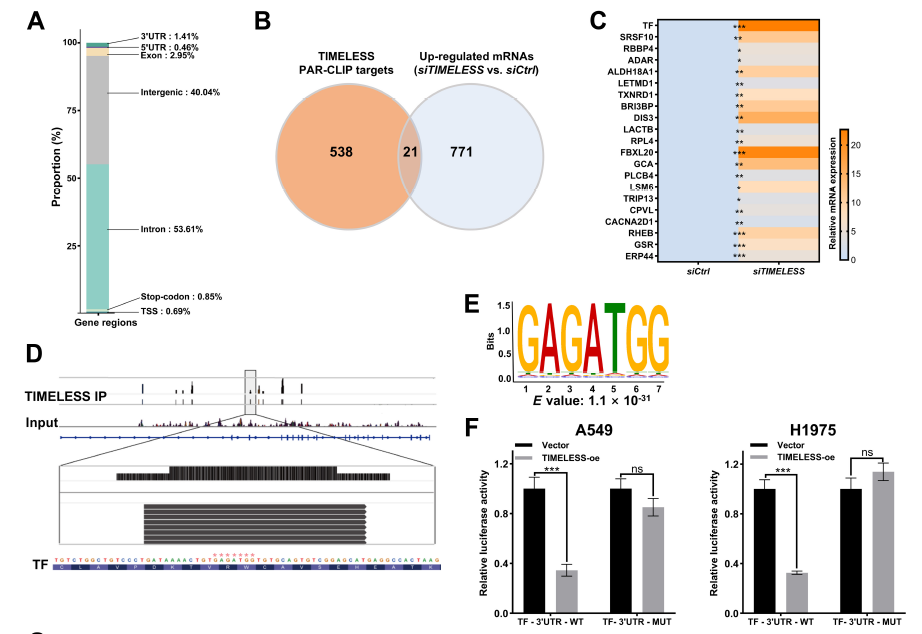
Further mechanistic studies revealed that TIMELESS, functioning as an RNA-binding protein, recruits CNOT3 (a core subunit of the Ccr4-Not complex) to accelerate TF mRNA deadenylation and degradation, thereby suppressing transferrin-mediated ferroptosis (Fig. 6). TF knockdown reversed ferroptosis induced by TIMELESS knockout, confirming TF as the critical downstream target mediating TIMELESS-regulated ferroptosis.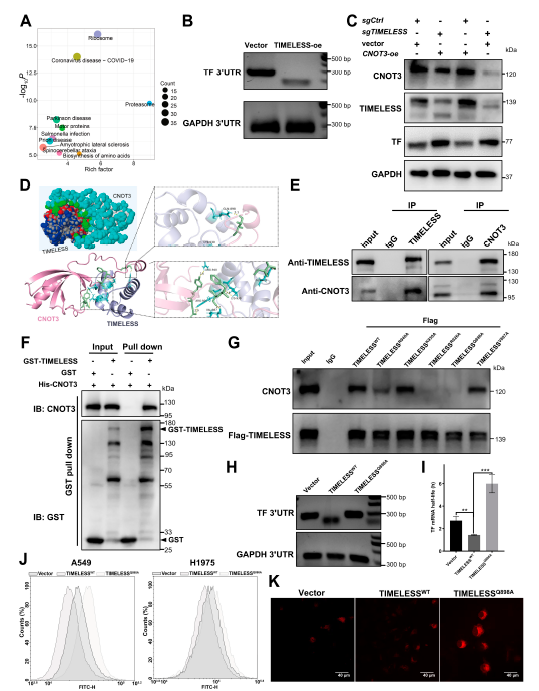
In LLC1 orthotopic lung cancer mouse models, TIMELESS knockdown combined with the ferroptosis inducer erastin and PD-1 inhibitors significantly inhibited tumor growth and prolonged mouse survival (Fig. 7B,C); this combination effectively remodeled the tumor immune microenvironment, increasing infiltration of CD4+, CD8+ T cells and M1 macrophages while reducing M2 macrophage infiltration (Fig. 7E-G), achieving dual effects of "ferroptosis induction + immune microenvironment reprogramming."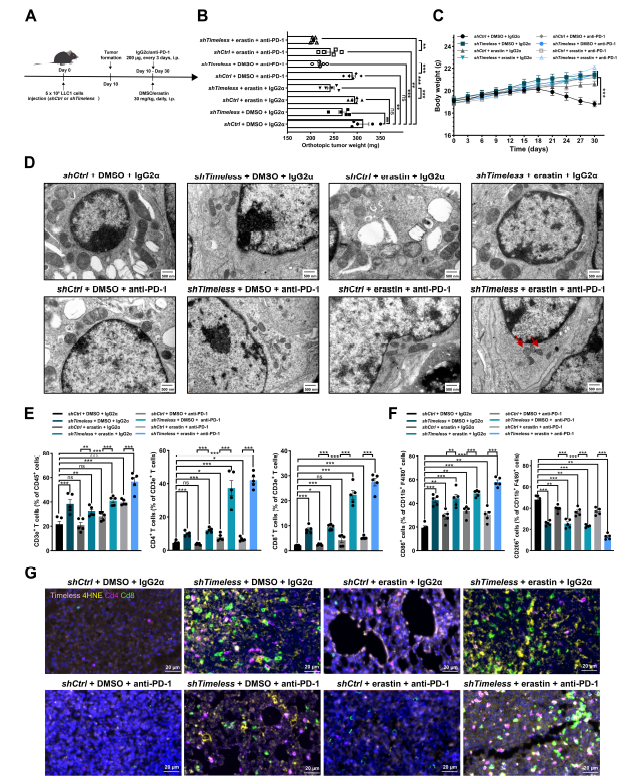
Clinical tissue microarray multiplex immunofluorescence analysis revealed that LUAD tissues with high TIMELESS expression exhibited reduced ferroptosis marker 4HNE expression, elevated PD-L1 expression, and concomitant reduction in CD4+ and CD8+ T cell infiltration with M2 macrophage enrichment (Fig. 8), confirming that TIMELESS drives PD-1 immunotherapy resistance through dual mechanisms of "ferroptosis suppression + immunosuppressive microenvironment remodeling."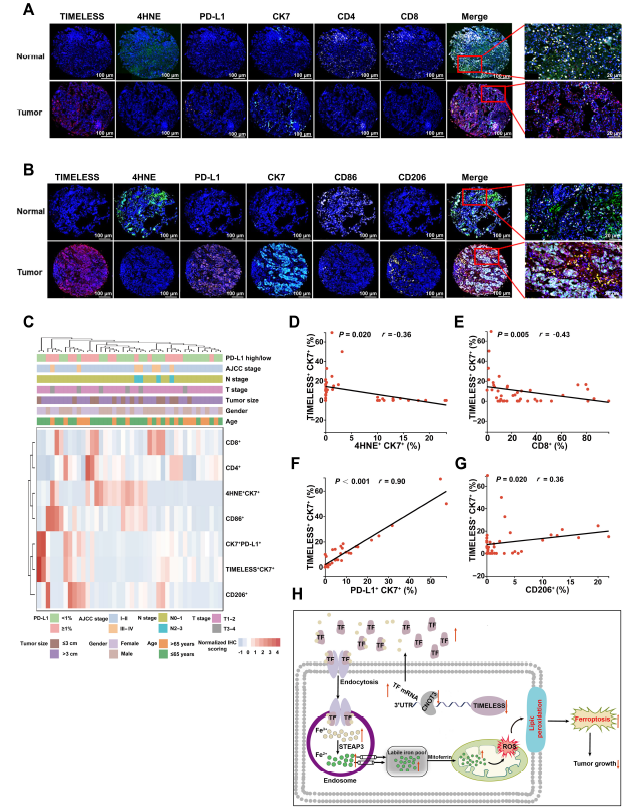
III. Absin Products Powering Critical Research Steps, Providing Robust Experimental Support
In this study, Absin human lung cancer organoid culture kits, Matrigel matrix, and other core products were applied to patient-derived organoid construction, in vitro cell culture, and organoid viability assays and other critical experimental procedures. With stable product performance, they provided essential guarantees for the reliability of experimental results. Specific products and applications are detailed below:
Application Scenario: Construction of LUAD patient-derived organoid (PDO) models (Methods section "Patient-derived organoids culture")
Research Contribution: As a preclinical model that highly simulates in vivo tumor characteristics, PDOs serve as a critical bridge between cell-based experiments and animal studies. The research team utilized this kit to successfully culture PDOs from fresh LUAD patient tumor tissues and achieved TIMELESS knockout in PDOs via CRISPR-Cas9 technology, validating that TIMELESS knockout enhances PDO sensitivity to erastin (Fig. 3K), providing a clinically relevant preclinical model for subsequent in vivo experiments.
Product Advantages: The kit contains complete reagents including specialized digestion solution, culture medium, and matrix gel, with formulations optimized for lung cancer tissue characteristics, enabling efficient tumor tissue digestion and long-term passaging of organoids while maintaining original molecular features and pathological characteristics.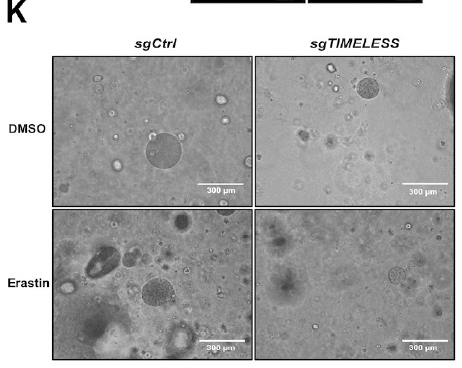
Application Scenario: Matrix support for PDO culture, cell embedding for orthotopic tumor model construction in mice (Methods section "Patient-derived organoids culture" and "Lewis lung carcinoma mouse model and therapeutic protocols")
Research Contribution:
① In PDO culture, Matrigel matrix provides a biomimetic extracellular matrix environment for three-dimensional organoid growth, ensuring normal morphology and proliferation;
② In LLC1 cell orthotopic lung injection experiments, mixing cells with Matrigel improved cell colonization efficiency in lung tissue, successfully establishing stable orthotopic lung cancer models and providing reliable in vivo models for combination therapy efficacy validation.
Product Advantages: Serum-free, mycoplasma-free, with stable gel structure and excellent biocompatibility, efficiently supporting three-dimensional culture of tumor organoids and normal tissue organoids, while being suitable for cell transplantation in vivo and tumor formation experiments.
IV. Research Significance and Clinical Translation Value
This study is the first to reveal the dual function of TIMELESS in regulating ferroptosis and immune microenvironment in LUAD, establishing the TIMELESS/CNOT3/TF axis as a novel therapeutic target for LUAD, and proposing a "TIMELESS knockdown/inhibition + ferroptosis inducer + PD-1 inhibitor" triple combination therapy strategy, providing new insights for addressing immunotherapy resistance in LUAD.
From a basic research perspective, this study enriches the molecular mechanisms of RBP-mediated regulation of tumor ferroptosis and expands the functional understanding of circadian genes in tumor immune microenvironment remodeling; from a clinical translation perspective, TIMELESS may serve as a biomarker for prognostic assessment in LUAD patients, while providing a potential target for precision therapy, with the combination of ferroptosis inducers and immune checkpoint inhibitors representing a promising new therapeutic regimen for LUAD.
V. Conclusion
This research represents a significant breakthrough for LUAD basic research and clinical treatment. As a professional brand in the research reagent field, Absin supported this study throughout with high-quality organoid culture products, Matrigel matrix, and supporting reagents, fully demonstrating the core supporting role of Absin products in tumor organoid model construction, cellular functional validation, and in vitro/in vivo experiments.
Moving forward, Absin will continue to focus on the frontiers of life science research, continuously optimizing our product portfolio to provide superior and more comprehensive research tools for oncology, immunology, cell biology, and related fields, empowering more researchers to overcome scientific challenges and advance the translation from basic research to clinical application!
This content is based on the original publication in Cancer Communications (DOI: 10.34133/cancomm.0009); all images, data, and intellectual property rights referenced herein belong to the original journal and research team. Should any infringement occur, please contact us promptly for removal, and we will cooperate fully to resolve such matters.
|
Item NO. |
Product Name |
Size |
| abs9443 | Human Lung Cancer Organoid Culture Medium Kit | 1kit |
| abs9495 | GFR OrganoGel Phenol red free | 1.5mL×4/1.5mL×8 |
Contact Absin
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
| Absin Bioscience Inc. worldwide@absin.cn |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
