- Cart 0
- English
A Comprehensive Guide to Detecting Reactive Oxygen Species (ROS) in Cells
Reactive oxygen species (ROS) are by-products of cellular metabolism, including superoxide anion radicals, hydrogen peroxide, hydroxyl radicals, etc. They play roles in cellular signaling, but excessive production can lead to cellular damage and is associated with various diseases. Current methods for detecting ROS include fluorescent staining, chemiluminescence, electron paramagnetic resonance (EPR/ESR), electrochemical biosensors, chromatography, spectrophotometry, and methods based on fluorescent proteins (Table 1).
Table 1 Common Methods for ROS Detection
|
ROS Detection Method |
Method Description |
|
Fluorescent Staining |
Using specific fluorescent probes such as DCFH-DA, which can detect various ROS. The probe itself is non-fluorescent, enters cells and is hydrolyzed by esterases to generate DCFH, which is then oxidized by ROS to produce the fluorescent DCF. |
|
Chemiluminescence |
Utilizing the luminescent signals generated from the reaction of certain chemicals with ROS for quantitative detection of ROS. |
|
Electron Paramagnetic Resonance (EPR/ESR) |
A direct method for detecting free radicals, suitable for detecting various ROS including hydroxyl radicals. |
|
Electrochemical Biosensors |
Detection of ROS using electrochemical methods, characterized by high sensitivity and selectivity. |
|
Chromatography |
Detection of specific ROS or their metabolites through chromatographic separation techniques. |
|
Spectrophotometry |
Using specific spectrophotometers to detect ROS, such as detecting superoxide anion by monitoring the reduction of nitroblue tetrazolium (NBT). |
|
Fluorescent Protein-based Methods |
Genetic engineering techniques are used to fuse fluorescent proteins with ROS-sensitive peptides, and ROS are detected through changes in fluorescence. |
Detection of ROS levels is crucial for understanding cellular physiological functions and cell damage caused by environmental factors. Today, we will focus on the principles and specific operational steps of detecting intracellular ROS using fluorescent staining.
ROS Detection Guide
I. Selection of Appropriate Probes
DCFH-DA: This is a commonly used fluorescent probe that quantifies intracellular ROS levels by detecting the fluorescence intensity of its oxidation product DCF. DCFH-DA itself is non-fluorescent, can freely cross cell membranes, is hydrolyzed by esterases within cells to generate DCFH, and is then oxidized to produce DCF, with fluorescence intensity being proportional to ROS levels.
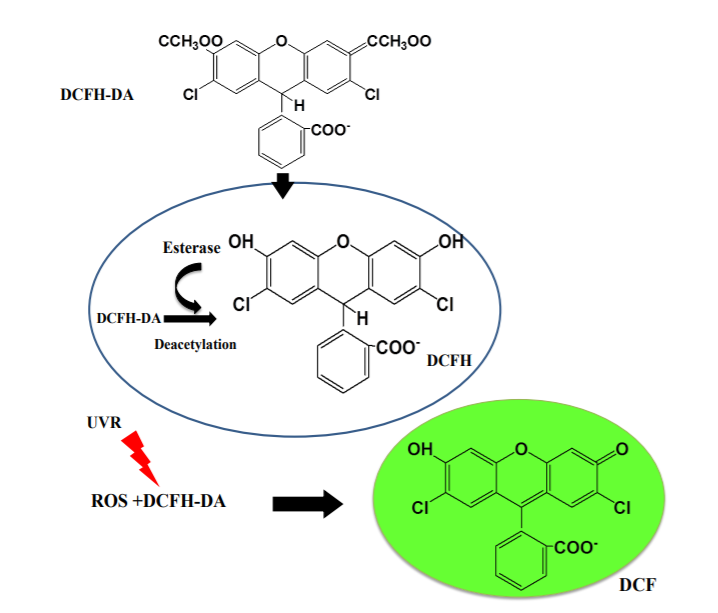
Figure 1 Mechanism of action of DCFH-DA probe in cells[1]
II. Experimental Steps (Taking abs580232, ROS Detection Kit as an example)
1. Loading ROS Probes
1) In situ Probe Loading (Only applicable to adherent cells)
① Cell Preparation: Plate cells the day before detection to ensure that the cell density is less than 5×105/mL at the time of detection.
② Drug Induction: Remove the cell culture medium, add drug treatment diluted in serum-free medium, and incubate in the cell culture incubator at 37℃ in the dark. The actual induction time is determined by the drug characteristics and cell type.
③ (Optional) Positive Control: Dilute the positive control (abs580232, Rosup, 100mM) with serum-free medium to a commonly used working concentration of 100μM, add to cells, and incubate at 37℃ in the dark for 30min-4h to increase ROS levels. Different cell types may vary. For example: HeLa cells require 30-60min of incubation, while MRC5 human embryonic fibroblasts require 90min.
④ ROS Probe Preparation: Dilute DCFH-DA 1:1000 with serum-free culture medium before probe loading to achieve a final concentration of 10μM.
⑤ ROS Probe Loading: Remove the drug treatment, add the appropriate volume of the diluted DCFH-DA working solution. The volume added should fully cover the cells. For example: for a 6-well plate, at least 1mL is usually required, and for a 96-well plate, at least 100μL is usually required. Incubate at 37℃ in the cell culture incubator in the dark for 30min.
⑥ Cell Washing: Wash the cells 1-2 times with serum-free culture medium to thoroughly remove DCFH-DA that has not entered the cells.
2) Probe Loading After Cell Collection (Applicable to both adherent and suspension cells)
① Cell Preparation: Culture cells according to standard methods, ensuring the cell condition for detection. Collect a sufficient amount of cells using appropriate methods.
② Drug Induction: Suspend the collected cells in an appropriate volume of diluted drug and incubate in the cell culture incubator at 37℃ in the dark. The actual induction time is determined by the drug characteristics and cell type.
③ (Optional) Positive Control: Dilute the positive control (abs580232, Rosup, 100mM) with serum-free medium to a commonly used working concentration of 100μM, add to cells, and incubate at 37℃ in the dark for 30min-4h to increase ROS levels. Different cell types may vary. For example: HeLa cells require 30-60min of incubation, while MRC5 human embryonic fibroblasts require 90min.
④ ROS Probe Preparation: Dilute DCFH-DA 1:1000 with serum-free culture medium before probe loading to achieve a final concentration of 10μM.
⑤ Probe Loading: Remove the drug from the cells, collect the cells by centrifugation, and add the diluted probe to achieve a cell density of 1.0×106-2.0×107. Note: The cell density should be adjusted according to the subsequent detection system, detection method, and total detection volume. For example: for flow cytometry, the number of cells per tube should be no less than 104 and not more than 106.
⑥ Cell Washing: Wash the cells 1-2 times with serum-free cell culture medium to thoroughly remove DCFH-DA that has not entered the cells.
2. Fluorescence Microscopy Detection
1) For adherent cells or live tissues, direct observation under the fluorescence microscope is possible; for suspension cells, take 25-50μL of cell suspension and drop it onto a microscope slide, then cover with a cover slip.
2) Under the fluorescence microscope, use the FITC filter to observe fluorescence and remove background to observe changes in fluorescence.
3. Flow Cytometry Analysis
1) For adherent cells, digest with trypsin to prepare a single-cell suspension; for suspension cells, collect the cells directly. Resuspend the cells with 0.5-1mL of PBS (0.5-1×105/mL).
2) Select the FL1 or BL1 channel of the flow cytometer, excite at 488nm, and measure the emission at 530nm. Cells should be divided into two subpopulations: ROS-negative cells with very low fluorescence intensity and ROS-positive cells with strong green fluorescence.
Application of DCFH-DA in ROS Detection
Gao[2] et al. used the standard fluorescent probe 2′,7′-dichlorodihydrofluorescein diacetate to measure ROS in 4T1 cells after liposome treatment. The specific procedure is as follows:
1) 4T1 cells were seeded in 35mm dishes at a density of 8×104 cells/well.
2) After 24h of cell culture with a confluence of 70–80%, liposomes (1mL, 1mg/mL) were added to the culture medium. After 6h of treatment, cells were washed three times with PBS.
3) Cells were stained with DCFH-DA (10μM), incubated for 30min, washed three times with PBS, and then imaged using confocal laser scanning microscopy (CLSM).

Figure 2 ROS Imaging in 4T1 Cells[2]
Regardless of the liposome type, fucosylated liposomes generated more ROS in 4T1 cells than non-targeted liposomes (Figure 2), mainly due to enhanced cell uptake. Due to DAPC peroxidation, DAPC-Fuc was more effective in ROS generation than DOPC-Fuc.
References:
[1] Rajneesh, Pathak J, Chatterjee A, et al. Detection of Reactive Oxygen Species (ROS) in Cyanobacteria Using the Oxidant-sensing Probe 2',7'-Dichlorodihydrofluorescein Diacetate (DCFH-DA)[J]. Bio Protoc. 2017;7(17):e2545.
[2] Gao Z, Zhang J, Hou Y, et al. Boosting the synergism between cancer ferroptosis and immunotherapy via targeted stimuli-responsive liposomes[J]. Biomaterials.2024;305:122442.
Recommended ROS Detection Products:
|
Catalog Number |
Product Name |
Specifications |
|
Reactive Oxygen Species (ROS) Detection Kit (Green Fluorescence) |
1000T |
|
|
Reactive Oxygen Species (ROS) Detection Kit (Red Fluorescence) |
100-500T |
Recommended Oxidative Stress Related Products:
|
Detection Item |
Catalog Number-Specifications |
Product Name |
Detection Method/Range |
|
Lipid Peroxidation Products |
Malondialdehyde (MDA) Detection Kit |
Colorimetric; 0.01mmol/L-1mmol/L |
|
|
Antioxidant Enzymes |
Superoxide Dismutase (SOD) Detection Kit |
Colorimetric; 1U/mL-30U/mL |
|
|
Glutathione Peroxidase Detection Kit |
Glutathione Peroxidase Detection Kit |
Colorimetric; 0.01mmol/L-5mmol/L |
|
|
Glutathione Reductase Detection Kit |
Glutathione Reductase Detection Kit |
Colorimetric; 4umol/L-400umol/L |
|
|
Glutathione S-Transferase (GST) Detection Kit |
Glutathione S-Transferase (GST) Detection Kit |
Colorimetric |
|
|
Non-enzymatic Antioxidants |
Glutathione Detection Kit |
Colorimetric; 0.01mmol/L-0.5mmol/L |
|
|
Vitamin E Detection Kit |
Vitamin E Detection Kit |
Colorimetric; 50umol/L-2000umol/L |
|
|
Vitamin C Detection Kit |
Vitamin C Detection Kit |
Colorimetric; 0.02mmol/L-2mmol/L |
|
|
Coenzyme Q10 Detection Kit |
Coenzyme Q10 Detection Kit |
Colorimetric; 0.1mmol/L-10mmol/L |
|
|
Total Antioxidant Capacity Detection Kit |
Total Antioxidant Capacity Detection Kit |
Colorimetric; 0.04mmol/L-4mmol/L |
Recommended Mitochondrial Oxidative Damage Detection Products:
|
Catalog Number |
Product Name |
Specifications |
|
Mitochondrial Respiratory Chain Complex I Activity Detection Kit (Micro Method) |
96T |
|
|
Mitochondrial Respiratory Chain Complex II Activity Detection Kit (Micro Method) |
96T |
|
|
Mitochondrial Respiratory Chain Complex III Activity Detection Kit (Micro Method) |
96T |
|
|
Mitochondrial Respiratory Chain Complex IV Activity Detection Kit (Micro Method) |
96T |
|
|
Mitochondrial Respiratory Chain Complex V Activity Detection Kit (Micro Method) |
96T |
|
|
Mitochondrial Permeability Transition Pore (MPTP) Detection Kit |
50T |
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
|
Absin Bioscience Inc. |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
April 15, 2025
Clicks:458
