- Cart 0
- English
Interpreting the Correlation Between Gut Microbiota and Autism
March 03, 2025
Clicks:600
The microbiome refers to the entire community of microorganisms and their genetic information in a specific environment or ecosystem. These microorganisms mainly include bacteria, archaea, protozoa, fungi, and viruses. Microorganisms are widely distributed in our living environment and exhibit ecosystem functions through complex interactions. Currently, microbiome research is highly active, primarily involving studies of microorganisms in marine, river, and soil environments, as well as in the human oral cavity, gut, skin, and reproductive tract. In recent years, the close relationship between human commensal microorganisms and host health and disease has gained significant attention, particularly the associations between microbiota and cardiovascular diseases, immune disorders, metabolic diseases, depression, autism, Alzheimer's disease, and cancer.

Today, we share a research article titled Human Gut Microbiota from Autism Spectrum Disorder Promote Behavioral Symptoms in Mice, published in Cell by Gil Sharon et al. from the California Institute of Technology, with an impact factor of 31.398. This study isolated functional bacteria from the feces of children with autism and typically developing children, transplanted them into the gut of mice to construct an animal model with a stable human-like gut microenvironment, and revealed that gut bacteria directly promote the occurrence of autistic behaviors in mice, pointing to a potential new direction for treating autism and other neurodevelopmental disorders.

Research Background:
Autism Spectrum Disorder (ASD) is characterized by complex changes in human behavior, including social communication deficits and restricted, repetitive behaviors. In addition to genetic differences, gut microbiota also vary between typically developing (TD) individuals and those with ASD. However, it remains unclear whether the microbiota are associated with symptoms.
Research Materials:
Germ-free (GF) male and female mice; ASD symptom mice; BTBR T+ tf/J (BTBR) mouse model; fecal samples from typically developing (TD) children and children with autism (ASD).
Research Techniques:
16S rDNA analysis, non-targeted metabolomics, and transcriptomics studies
Highlights:
1) Mice harboring microbiota from ASD, but not TD, individuals exhibited ASD-like behaviors;
2) ASD and TD microbiota produced distinct metabolomic profiles in mice;
3) Mice with ASD microbiota exhibited widespread alternative splicing of risk genes in the brain, and BTBR mice treated with 5AV or taurine showed improved repetitive and social behaviors.
Research Results:
1. Gut microbiota from ASD individuals are sufficient to alter mouse behavior
The study first tested whether human microbiota functionally promote ASD-like behaviors in mice. Fecal samples from human TD and ASD donors were transplanted into the gut of GF wild-type mice, and behavioral analyses were performed on these mice and their offspring. The results showed that mice transplanted with feces from ASD patients exhibited increased repetitive behaviors, reduced locomotion, decreased social interaction, and clear autistic symptoms.

Mice transplanted with ASD microbiota recapitulate human autistic behaviors
2. Different microbial populations in TD and ASD transplanted mice lead to behavioral differences
A systematic analysis of the gut microbiota in ASD patients, ASD mice, and their offspring revealed significant differences compared to controls, and these differences could be vertically transmitted to offspring mice. Further studies identified specific types of gut bacteria (Bacteroides, Parabacteroides, Eisenbergiella tayi) as key contributors to ASD.
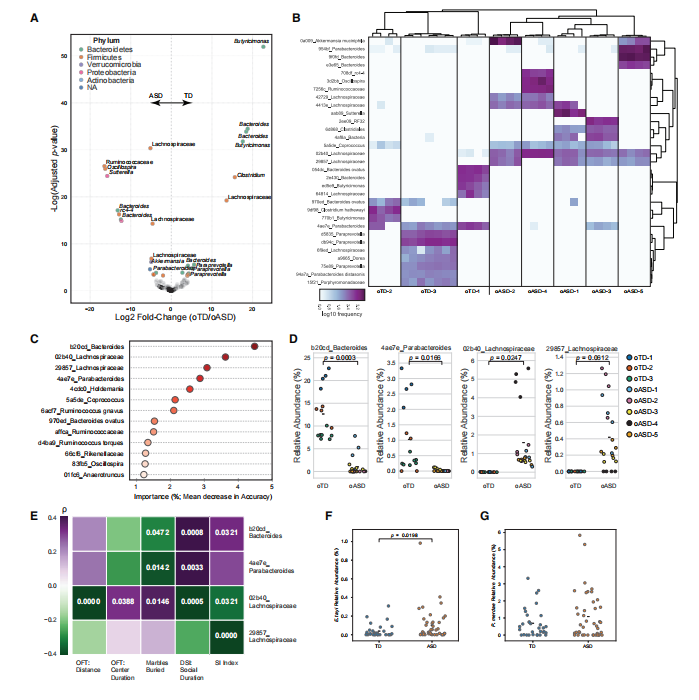
Differences in the abundance of Lachnospiraceae, Bacteroidaceae, and Parabacteroidaceae in the microbiota of TD and ASD offspring
3. ASD microbiota promote widespread alternative splicing of ASD-related genes in the brain
Social behaviors are mediated by multiple brain regions, including the prefrontal cortex (PFC) and striatum (STR), and synaptic dysfunction and abnormal developmental trajectories may lead to increased repetitive behaviors. Through large-scale analysis, researchers compared the differential expression of various genes and non-coding RNAs in the brains of TD and ASD individuals. The results showed few differentially expressed genes. Further investigation of whether ASD brains exhibited differential alternative splicing (AS) patterns (a characteristic found in human ASD brains) revealed that 569 genes exhibited significant changes in alternative splicing (highly expressed in neurons), including 52 ASD-related genes.
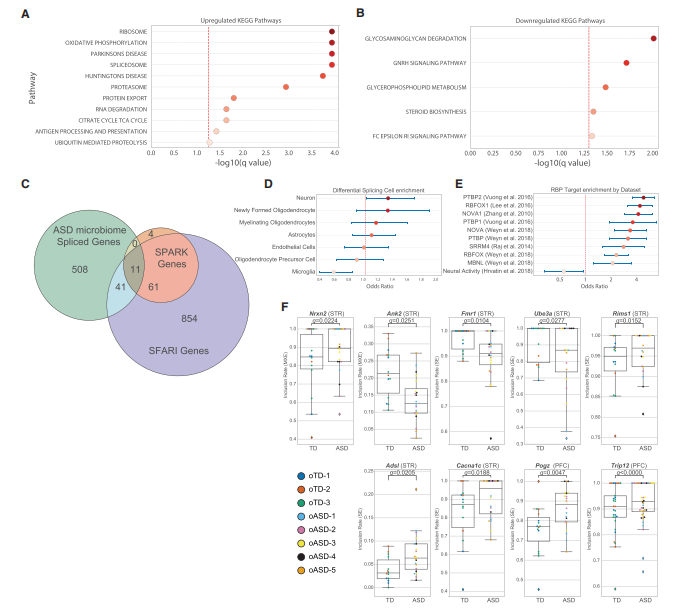
Microbiota influence the expression and alternative splicing of ASD genes in the mouse brain
4. Microbes from TD and ASD transplanted mice produce metabolites
Gene expression, splicing, and neuronal function in the brain can be regulated by small-molecule metabolites. Metabolomic changes are associated with Autism Spectrum Disorder. Therefore, non-targeted metabolomic analysis was performed on the colonic contents and corresponding serum of oTD and oASD mice. The results showed that 27 metabolites in the colonic contents of oASD mice were significantly different from those of oTD mice, with several agonists and antagonists of GABA and glycine receptors exhibiting differences. Specifically, 5-aminovaleric acid (5AV) and taurine were significantly reduced in oASD mice. Subsequent functional prediction analysis of the metabolic process revealed that L-proline and/or trans-4-hydroxy-L-proline in the gut of control mice ultimately produced 5AV, while glutamate was generated in the gut of ASD mice.

Effects of oTD and oASD microbiota on the colonic and serum metabolome of mice
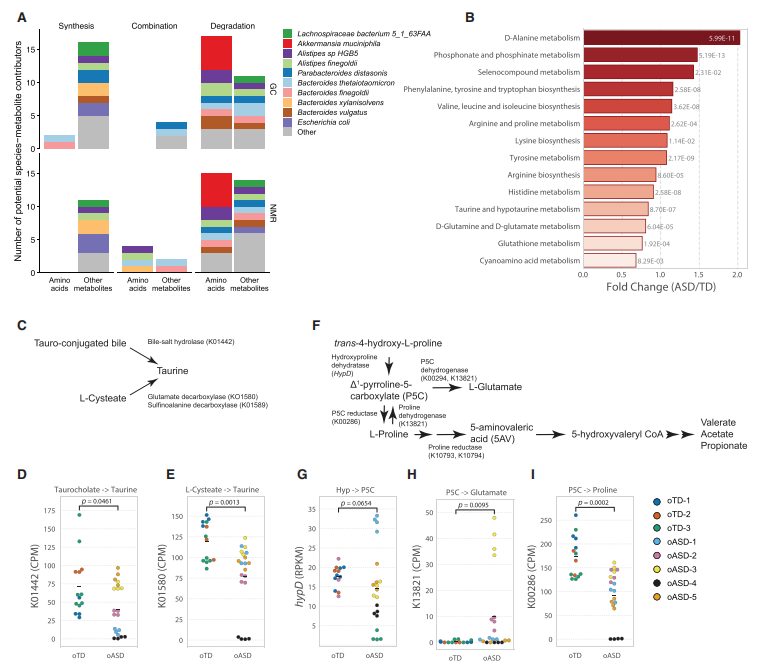
Metagenomic analysis confirms a lack of amino acid metabolism in oASD mice
5. Modulating ASD mouse metabolites improves ASD-like behaviors
Microbial metabolites in the gut can influence the prognosis of the nervous system. To test whether 5AV and taurine affect mouse behavior, Spearman correlation analysis revealed that several metabolites were correlated with behavioral abnormalities in the mouse model, including taurine and 5AV, which may protect mice from ASD-like behaviors. Subsequently, using the BTBR T+ tf/J (BTBR) mouse model, oral administration of taurine or 5AV from gestation to adulthood significantly reduced repetitive behaviors and increased social interactions. Electrophysiological and calcium imaging experiments further revealed that 5AV reduced neuronal excitability in the prefrontal cortex of BTBR mice.

Microbial metabolites affect behavior and brain physiology in the BTBR mouse model
|
Product Code |
Product Name |
Specification |
|
Rabbit anti-Integrin β3 Polyclonal Antibody |
50uL |
|
|
Mouse anti-GAPDH Monoclonal Antibody |
50uL |
|
|
Rabbit anti-ICAM1 Polyclonal Antibody |
50uL |
|
|
Mouse anti-HA Tag Monoclonal Antibody |
100ug |
|
|
Mouse anti-LC3A/B Monoclonal Antibody(3E9) |
100uL |
|
|
Click to find more antibody products: https://www.absin.net/antibodies.html |
||
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us
|
Absin Bioscience Inc. |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
