- Cart 0
- English
Discovery of a New Inflammatory Checkpoint in KRAS-Mutant iCCA
December 03, 2024
Clicks:457
Intrahepatic cholangiocarcinoma (iCCA) is an epithelial adenocarcinoma originating from the second-order bile ducts within the liver and their branches. It accounts for 10% to 15% of primary liver malignancies and has seen a steady increase in both incidence and mortality over the past few decades. The primary treatment method remains surgical resection, similar to that for primary liver cancer, supplemented by immune checkpoint inhibitors (ICIs) and therapies targeting the tumor microenvironment (TME). However, there are currently no effective treatment measures for unresectable iCCA.
By the end of 2021, a team led by Academician Fan Jia from Zhongshan Hospital affiliated with Fudan University, in collaboration with Professor Gao Qiang, Researcher Zhou Hu from the Shanghai Institute of Materia Medica, Chinese Academy of Sciences, and Researcher Gao Daming from the Center for Excellence in Molecular Cell Science, Chinese Academy of Sciences, published a research paper titled "Proteogenomic characterization identifies clinically relevant subgroups of intrahepatic cholangiocarcinoma" in the journal Cancer Cell. In this study, Academician Fan Jia's team discovered that TP53, KRAS, FGFR2, IDH1/2, and BAP1 mutations (fusions) are the main driver gene alterations in intrahepatic cholangiocarcinoma.
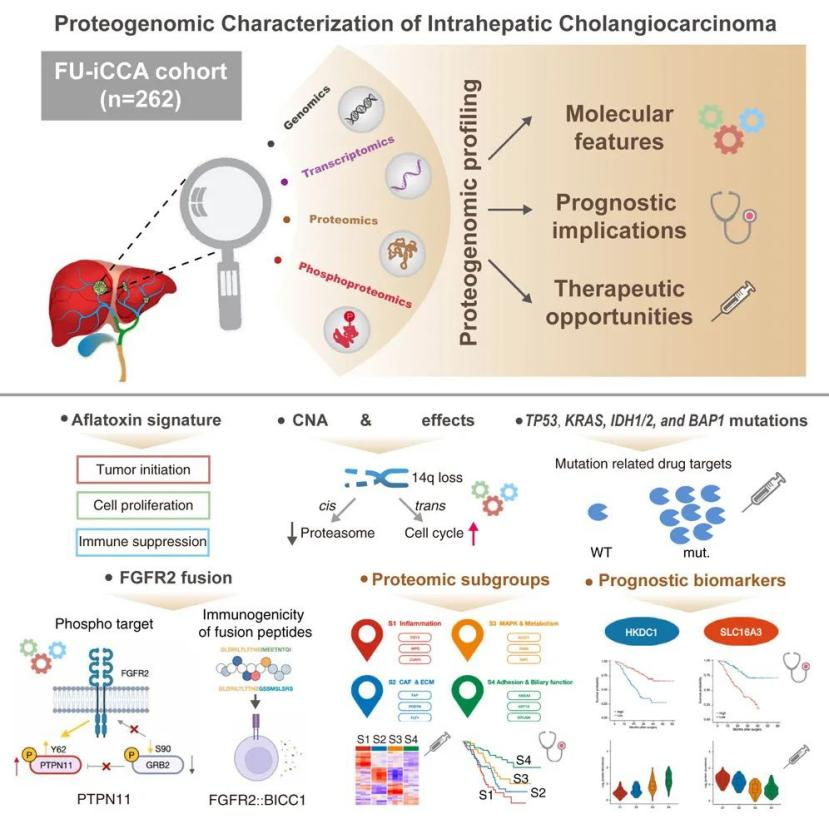
On July 24, 2023, Professor Gao Qiang and Dr. Lin Jian from the team of Academician Fan Jia at Zhongshan Hospital affiliated with Fudan University, in collaboration with Researcher Wei Wu's team from Lingang Lab, published a research paper titled "An inflammatory checkpoint generated by IL1RN splicing offers therapeutic opportunity for KRAS mutant intrahepatic cholangiocarcinoma" in the journal Cancer Discovery (IF=28.1). The study revealed a causal relationship between KRAS mutation and pre-tumor inflammation, which is considered a driving factor in tumorigenesis.
By analyzing multi-omics data collected from a large number of patients, the research team also found that KRAS mutations are associated with specific selective mRNA splicing, which is related to myeloid inflammation in intrahepatic cholangiocarcinoma. Subsequently, the team identified a negative feedback mechanism where the interleukin-1 receptor antagonist (IL1RN)-201/203 is upregulated due to selective splicing, thereby playing a significant anti-inflammatory role in KRAS-mutant iCCA. In KRAS-mutated iCCA mice, upregulation of IL1RN-201/203 and treatment with Anakinra (an interleukin-1 receptor antagonist) both ignite a significant anti-tumor immune response by altering neutrophil recruitment and phenotype. Furthermore, Anakinra treatment synergistically enhances anti-PD-1 therapy, activating GZMB+CD8+ T cells within the tumors of KRAS-mutated iCCA mice. Clinically, the research team found that high levels of IL1RN-201/203 in patients with KRAS-mutated iCCA are closely associated with a good response to anti-PD-1 immunotherapy.

Figure: The overall design concept and experimental methods of this study.
The research team utilized a large amount of high-throughput data, including whole exome sequencing (WES), bulk RNA sequencing (RNA-seq), and BD single-cell sequencing (scRNA-seq) from iCCA cohorts and public databases. These analyses confirmed the causal link between KRAS mutations and tumors, indicating that promoting inflammation is a key driving factor in iCCA. The team identified a new inflammatory checkpoint generated by selective splicing of IL1RN mRNA, which can inhibit KRAS mutation-mediated inflammatory signaling cascades, tumor progression, and ICI resistance by regulating neutrophil recruitment and polarization. These findings suggest that selective splicing of IL1RN may be an effective therapeutic target for KRAS-mutant cancers.
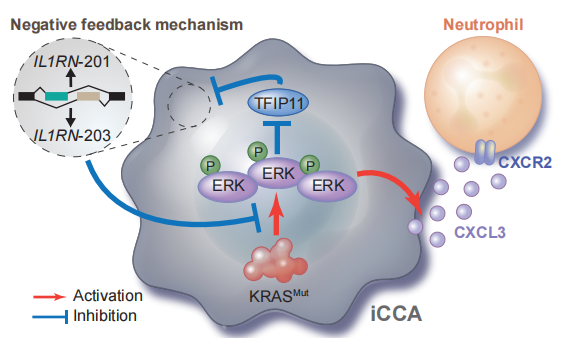
Figure: Schematic diagram of the negative feedback of IL1RN-201/203 transcripts and their downstream functions in iCCA.
In the study, the research team also applied Absin's multiplex fluorescence immunohistochemistry (mIHC) technology to perform some protein-level staining validations on the model mice.
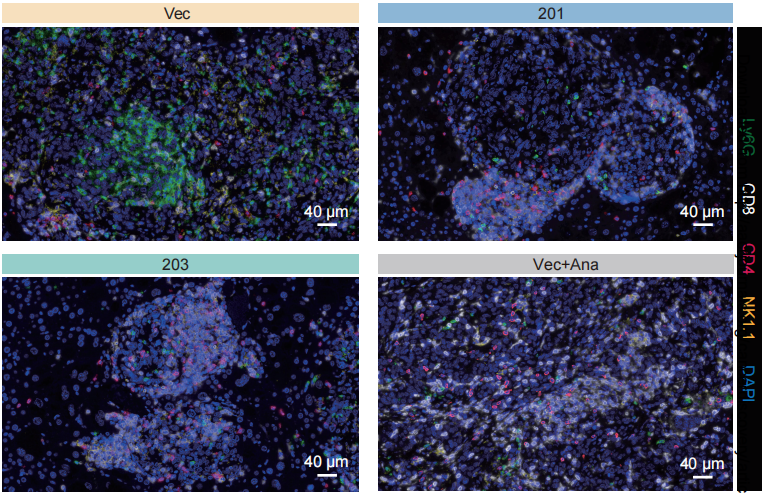
Figure: Significant increase in infiltration of CD8+ T cells in the IL1RN-201/203 or Anakinra treatment groups in Yak and KP mice.
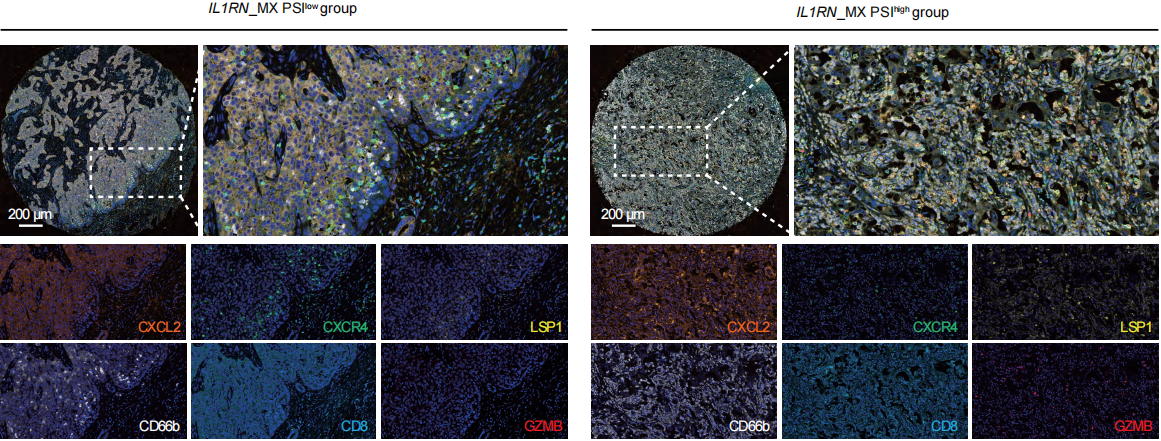
Figure: Compared to IL1RN_MX PSIlow KRAS-mutated samples, the density of CXCR4+ and LSP1+ neutrophils in IL1RN_MX PSIlow KRAS-mutated samples slightly decreases, while the density of CXCL2+ neutrophils and GZMB+CD8+ T cells increases.
This study provides a comprehensive overview of selective mRNA splicing and identifies a novel inflammatory checkpoint generated by IL1RN alternative splicing, offering an effective option for the treatment of KRAS-mutated iCCA. More broadly, in-depth research on this type of negative feedback loop will help to uncover the unique biology of KRAS-mutated cancers, thereby promoting the exploration of targeted strategies for these affected patients.
References
[2] Zhang M, Huang Y, Pan J, Sang C, Lin Y, Dong L, Shen X, Wu Y, Song G, Ji S, Liu F, Wang M, Zheng Y, Zhang S, Wang Z, Ren J, Gao D, Zhou J, Fan J, Wei W, Lin J, Gao Q. An inflammatory checkpoint generated by IL1RN splicing offers therapeutic opportunity for KRAS mutant intrahepatic cholangiocarcinoma. Cancer Discov. 2023 Jul 24:CD-23-0282. doi: 10.1158/2159-8290.CD-23-0282. Epub ahead of print. PMID: 37486241.
|
Item NO. |
Product Name |
Size |
|
Absin 4-Color IHC Kit (Anti-Rabbit and Mouse Secondary Antibody) |
20T/100T |
|
|
Absin 4-Color IHC Kit(Anti-Rabbit Secondary Antibody) |
20T/100T |
|
|
Absin 5-Color IHC Kit (Anti-Rabbit and Mouse Secondary Antibody) |
20T/100T |
|
|
Absin 5-Color IHC Kit (Anti-Rabbit Secondary Antibody) |
20T/100T |
|
|
Absin 6-Color IHC Kit (Anti-Rabbit and Mouse Secondary Antibody) |
20T/100T |
|
|
Absin 6-Color IHC Kit (Anti-Rabbit Secondary Antibody) |
20T/100T |
|
|
Absin 7-Color IHC Kit (Anti-Rabbit and Mouse Secondary Antibody) |
20T/100T |
|
|
Absin 7-Color IHC Kit(Anti-Rabbit Secondary Antibody) |
20T/100T |
|
|
Antibody eluent (for mIHC) |
30ml |
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.
|
Absin Bioscience Inc. |
 Follow us on Facebook: Absin Bio Follow us on Facebook: Absin Bio |
